The compound of xenon with zero dipole moment is:
A. \[XeO{F_4}\]
B. \[Xe{O_2}\]
C. \[Xe{O_3}\]
D. \[Xe{F_4}\]
Answer
267.6k+ views
Hint: The zero dipole moment of a compound has symmetrical geometry and also has similar atoms with less distance between the charge separation.
Complete Step by Step Solution:
1. The dipole moment is the product of the magnitude of charge and the distance between the centre of the positive and negative charge.
2. It is expressed as:
\[\mu = Q \times r\]
Where, \[\mu \] =dipole moment
\[Q\] =charge
\[r\] =separation distance
3. The dipole moment is a vector quantity that is depicted on a Lewis structure as a crossed arrow.
4. A cross appears on the positive end while an arrowhead appears on the negative end.
5. The valence electrons of the xenon are eight and the valence electrons of the Fluoride are seven so the total valence electrons of the compound are thirty-six.
6. There are a total of two lone pairs on the xenon and it is attached to the four Fluoride atoms through the bond pairs.
7. The geometry of the xenon tetrafluoride is square planar with a symmetrical structure with an angle of \[{90^ \circ }\].
8. The zero dipole moment of the compound is defined as the two equal bond dipoles that point in the opposite directions and cancel the effect of each other.
9. The individual bond dipoles cancel out and leave the molecule as a nonpolar so there is no net dipole moment.
10. The Lewis dot structure of the xenon tetrafluoride is represented below:
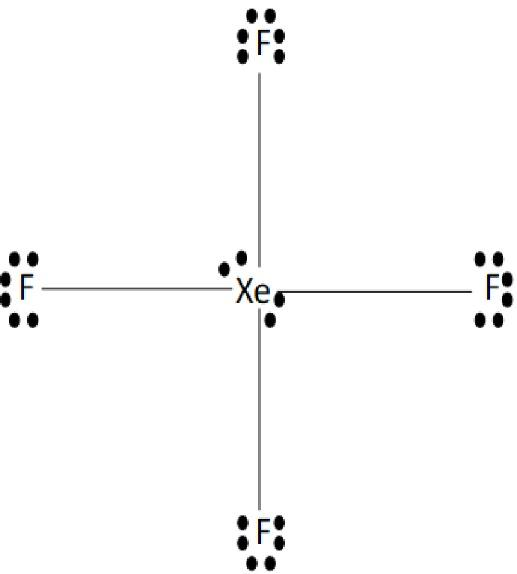
Image: Xenon tetrafluoride
Option (D) is correct.
Additional information:
1. When the same forces of energy are applied at an angle of \[{90^ \circ }\], there is no movement.
2. The direction of the dipole moment is from a less electronegative atom to a more electronegative atom.
Note: The dipole moment arrow represents the direction of the shift of electron density in the molecule with the direction of the crossed arrow opposite the conventional direction of the dipole moment vector.
Complete Step by Step Solution:
1. The dipole moment is the product of the magnitude of charge and the distance between the centre of the positive and negative charge.
2. It is expressed as:
\[\mu = Q \times r\]
Where, \[\mu \] =dipole moment
\[Q\] =charge
\[r\] =separation distance
3. The dipole moment is a vector quantity that is depicted on a Lewis structure as a crossed arrow.
4. A cross appears on the positive end while an arrowhead appears on the negative end.
5. The valence electrons of the xenon are eight and the valence electrons of the Fluoride are seven so the total valence electrons of the compound are thirty-six.
6. There are a total of two lone pairs on the xenon and it is attached to the four Fluoride atoms through the bond pairs.
7. The geometry of the xenon tetrafluoride is square planar with a symmetrical structure with an angle of \[{90^ \circ }\].
8. The zero dipole moment of the compound is defined as the two equal bond dipoles that point in the opposite directions and cancel the effect of each other.
9. The individual bond dipoles cancel out and leave the molecule as a nonpolar so there is no net dipole moment.
10. The Lewis dot structure of the xenon tetrafluoride is represented below:

Image: Xenon tetrafluoride
Option (D) is correct.
Additional information:
1. When the same forces of energy are applied at an angle of \[{90^ \circ }\], there is no movement.
2. The direction of the dipole moment is from a less electronegative atom to a more electronegative atom.
Note: The dipole moment arrow represents the direction of the shift of electron density in the molecule with the direction of the crossed arrow opposite the conventional direction of the dipole moment vector.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




