The chlorobenzene is generally obtained from a corresponding diazonium salt by reacting it with
A. \[C{u_2}C{l_2}\]
B. \[CuS{O_4}\]
C. \[Cu\]
D. \[Cu(N{H_3})_4^{2 + }\]
Answer
249.9k+ views
Hint: As we know that the equivalent haloarene is produced when a diazonium salt is treated with copper \[(I)\] chloride or copper \[(I)\] bromide. The Sandmeyer reaction states the name of this process. The benzene ring can then have a chloro or bromo group added to it.
Complete step-by-step answer:We must explain how the Sandmeyer reaction will be used to produce chlorobenzene from benzene diazonium chloride in order to answer the question.
As a result, aryl halides are produced from aryl diazonium salts using the Sandmeyer reaction, a chemical process. Traugott Sandmeyer, a Swiss chemist, is honored by the name of this reaction. The Sandmeyer reaction, which is frequently catalysed by copper \[(I)\] salts that is a procedure for the substituting an aromatic amino group by preparing its diazonium salt which is followed by its displacement. It is typically catalysed by copper salts. The nucleophile might be anything from cyanide to thiols to water to halide anions.
When cuprous chloride or cuprous bromide are applied to diazonium salt, respectively, chlorobenzene or bromobenzene is generated.
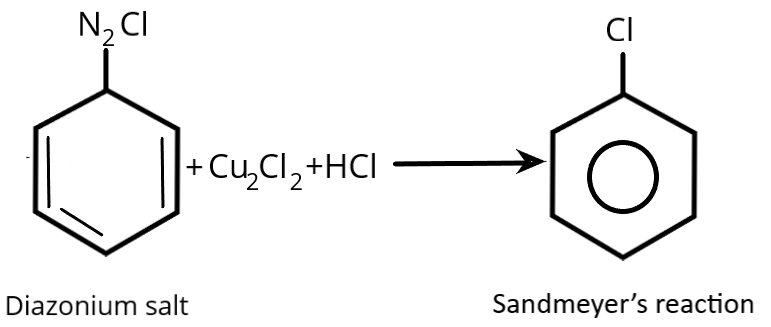
Option ‘A’ is correct
Note: It should be noted that an aromatic primary amine is processed with \[NaN{O_2}\] and dilute\[HCl\] at low temperature to produce benzene diazonium salt. The action is referred to as diazotization. The chemicals called diazonium salts are very reactive. A great deal of arene derivatives are prepared using them. With the aid of the Sandmeyer reaction, we are able to accomplish special transformations on benzene, such as halogenation, cyanation, and hydroxylation.
Complete step-by-step answer:We must explain how the Sandmeyer reaction will be used to produce chlorobenzene from benzene diazonium chloride in order to answer the question.
As a result, aryl halides are produced from aryl diazonium salts using the Sandmeyer reaction, a chemical process. Traugott Sandmeyer, a Swiss chemist, is honored by the name of this reaction. The Sandmeyer reaction, which is frequently catalysed by copper \[(I)\] salts that is a procedure for the substituting an aromatic amino group by preparing its diazonium salt which is followed by its displacement. It is typically catalysed by copper salts. The nucleophile might be anything from cyanide to thiols to water to halide anions.
When cuprous chloride or cuprous bromide are applied to diazonium salt, respectively, chlorobenzene or bromobenzene is generated.

Option ‘A’ is correct
Note: It should be noted that an aromatic primary amine is processed with \[NaN{O_2}\] and dilute\[HCl\] at low temperature to produce benzene diazonium salt. The action is referred to as diazotization. The chemicals called diazonium salts are very reactive. A great deal of arene derivatives are prepared using them. With the aid of the Sandmeyer reaction, we are able to accomplish special transformations on benzene, such as halogenation, cyanation, and hydroxylation.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




