Reaction of ethyl formate with an excess of \[C{H_3}MgI\] followed by hydrolysis gives:
A. N-propyl alcohol
B. Isopropyl alcohol
C. Acetaldehyde
D. Acetone
Answer
266.4k+ views
Hint: Ethyl formate is an ester compound with chemical formula \[HCOO{C_2}{H_5}\]. Hydrolysis is a reaction which involves the addition of water.
Complete Step by Step Solution:
Ethyl formate is an ester compound produced by the reaction of alcohol and formic acid.
A Grignard reagent is a chemical compound with a general formula of \[RMgX\]. Here, R is the alkyl group and X is the halide group. It is an organomagnesium compound. The grignard reagent is formed by the reaction of aryl or alkyl halide with magnesium.
When Grignard reagent reacts with ethyl formate an intermediate product is formed which on hydrolysis leads to isopropyl alcohol.
First, the negative charge of the methyl group of Grignard reagent attacks the central carbon of ethyl formate and the ethyl group is removed, again the negative charge methyl group attack the carbon and an intermediate compound magnesium iodide propan-2-olate. The intermediate compound then undergoes hydrolysis which means the addition of a water molecule which results in the formation of the main product i.e, Isopropyl alcohol.
The reaction of ethyl formate with an excess of \[C{H_3}MgI\] followed by hydrolysis gives Isopropyl alcohol. Therefore, option B is correct.
The reaction mechanism is shown below.
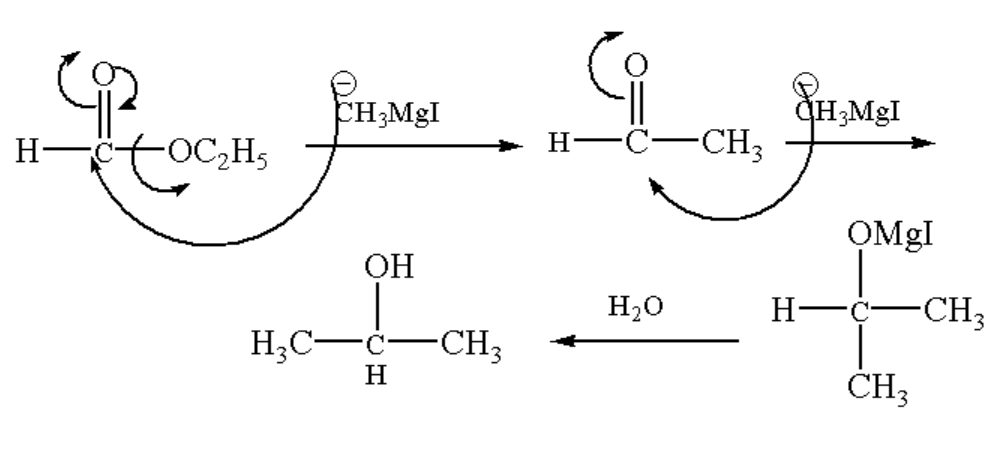
Image: Formation of Isopropyl alcohol
Note: It should be noted that the ester compound needs two equivalent of a Grignard reagent to produce tertiary alcohol. On the other hand aldehyde and ketone requires one equivalent of Grignard reagent to form secondary alcohol and tertiary alcohol respectively.
Complete Step by Step Solution:
Ethyl formate is an ester compound produced by the reaction of alcohol and formic acid.
A Grignard reagent is a chemical compound with a general formula of \[RMgX\]. Here, R is the alkyl group and X is the halide group. It is an organomagnesium compound. The grignard reagent is formed by the reaction of aryl or alkyl halide with magnesium.
When Grignard reagent reacts with ethyl formate an intermediate product is formed which on hydrolysis leads to isopropyl alcohol.
First, the negative charge of the methyl group of Grignard reagent attacks the central carbon of ethyl formate and the ethyl group is removed, again the negative charge methyl group attack the carbon and an intermediate compound magnesium iodide propan-2-olate. The intermediate compound then undergoes hydrolysis which means the addition of a water molecule which results in the formation of the main product i.e, Isopropyl alcohol.
The reaction of ethyl formate with an excess of \[C{H_3}MgI\] followed by hydrolysis gives Isopropyl alcohol. Therefore, option B is correct.
The reaction mechanism is shown below.

Image: Formation of Isopropyl alcohol
Note: It should be noted that the ester compound needs two equivalent of a Grignard reagent to produce tertiary alcohol. On the other hand aldehyde and ketone requires one equivalent of Grignard reagent to form secondary alcohol and tertiary alcohol respectively.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




