Pyramidal shape would be of
(a) \[N{O_3}^ - \]
(b) \[{H_2}O\]
(c) \[{H_3}{O^ + }\]
(d) \[N{H_4}^ + \]
Answer
249.3k+ views
Hint: By utilizing the concept of hybridization we can determine the shape of the molecules. For example., an ammonia (\[N{H_3}\]) molecule has \[s{p^3}\]hybridization, and as expected its structure must be tetrahedral. But due to the presence of lone pairs of electrons, it has a pyramidal structure. Similarly, \[S{F_4}\] molecule has \[s{p^3}d\]hybridization but due to the presence of lone pairs of electrons it has a see-saw shape.
Complete step by step solution:By using the concept of hybridization, we will determine the correct shape of the given molecules.
For the determination of the hybridization of an atom in a molecule, we will count the total number of sigma bonds and lone pairs of electrons.
Now one by one we will check the hybridization for the given molecules.
(a) The \[N{O_3}^ - \]molecule contains 3 sigma bonds. The value of 3 sigma bonds will be equal to \[s{p^2}\] hybridization. Because all the five valence electrons of the nitrogen atom are engaged in bonding with three oxygen atoms, therefore, \[N{O_3}^ - \]does not have any lone pair of electrons i.e., it has trigonal planar geometry as well as shape.
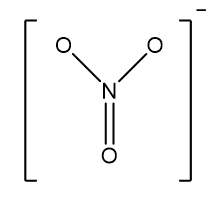
Image: Structure of nitrate.
(b) In the\[{H_2}O\]molecule, oxygen has six valence electrons, in which two are involved in the formation of sigma bonds with hydrogen atoms and the four-electrons remain as an unshared pair of electrons. Therefore, the total sum of the sigma bond and lone pair in\[{H_2}O\] will be 4. The value of 4 will be equal to \[s{p^3}\] hybridization and the geometry will be tetrahedral. But due to the presence of two lone pairs its shape will be V-shaped.
(c) In \[{H_3}{O^ + }\] molecule the oxygen also has six valence electrons, of which three are involved in the formation of sigma bonds with hydrogen atoms and the two-electron remain as unshared pair of electrons. Therefore, the total sum of the sigma bond and lone pair in\[{H_2}O\] will be 4. The value of 4 will be equal to \[s{p^3}\] hybridization and the geometry will be tetrahedral. But due to the presence of one lone pair its shape will be pyramidal.
(d) In \[N{H_4}^ + \] molecule the nitrogen has five valence electrons, which are involved in the formation of four sigma bonds with hydrogen atoms. Therefore, the total sum of sigma bonds in\[N{H_4}^ + \]will be 4. The value of 4 will be equal to \[s{p^3}\] hybridization and the geometry and shape will be tetrahedral.
Therefore from the above explanation we can say option (c) will be the correct option:
Note: The \[{H_3}{O^ + }\]is named a hydronium ion or protonated water molecule. Similarly, the\[N{H_4}^ + \]is called an ammonium ion and it is considered as the protonated form of an ammonia molecule.
Complete step by step solution:By using the concept of hybridization, we will determine the correct shape of the given molecules.
For the determination of the hybridization of an atom in a molecule, we will count the total number of sigma bonds and lone pairs of electrons.
Now one by one we will check the hybridization for the given molecules.
(a) The \[N{O_3}^ - \]molecule contains 3 sigma bonds. The value of 3 sigma bonds will be equal to \[s{p^2}\] hybridization. Because all the five valence electrons of the nitrogen atom are engaged in bonding with three oxygen atoms, therefore, \[N{O_3}^ - \]does not have any lone pair of electrons i.e., it has trigonal planar geometry as well as shape.

Image: Structure of nitrate.
(b) In the\[{H_2}O\]molecule, oxygen has six valence electrons, in which two are involved in the formation of sigma bonds with hydrogen atoms and the four-electrons remain as an unshared pair of electrons. Therefore, the total sum of the sigma bond and lone pair in\[{H_2}O\] will be 4. The value of 4 will be equal to \[s{p^3}\] hybridization and the geometry will be tetrahedral. But due to the presence of two lone pairs its shape will be V-shaped.
(c) In \[{H_3}{O^ + }\] molecule the oxygen also has six valence electrons, of which three are involved in the formation of sigma bonds with hydrogen atoms and the two-electron remain as unshared pair of electrons. Therefore, the total sum of the sigma bond and lone pair in\[{H_2}O\] will be 4. The value of 4 will be equal to \[s{p^3}\] hybridization and the geometry will be tetrahedral. But due to the presence of one lone pair its shape will be pyramidal.
(d) In \[N{H_4}^ + \] molecule the nitrogen has five valence electrons, which are involved in the formation of four sigma bonds with hydrogen atoms. Therefore, the total sum of sigma bonds in\[N{H_4}^ + \]will be 4. The value of 4 will be equal to \[s{p^3}\] hybridization and the geometry and shape will be tetrahedral.
Therefore from the above explanation we can say option (c) will be the correct option:
Note: The \[{H_3}{O^ + }\]is named a hydronium ion or protonated water molecule. Similarly, the\[N{H_4}^ + \]is called an ammonium ion and it is considered as the protonated form of an ammonia molecule.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




