On the velocity in a reversible reaction, the correct explanation of the effect of the catalyst is-
A. It provides a new reaction path of low activation energy
B. It increases the kinetic energy of reacting molecules
C. It displaces the equilibrium state on the right side
D. It decreases the velocity of the backward reaction
Answer
251.4k+ views
Hint: In a chemical reaction when reactants react to form the products at a certain concentration of the products, the products start to convert into the reactant simultaneously. Both reactions proceed simultaneously. Such types of reactions are called reversible reactions. A catalyst is used to change the rate of reaction.
Complete answer:A catalyst is a substance, compound, or element which is used in a reaction to change its rate of reaction or to form any specific product in the reaction. Catalyst themselves does not react in the reaction they only provide an alternative way to the reaction. The amount of catalyst used in the reaction remains constant. Catalysts are used to increase or decrease the rate of reaction, generally, it is used to increase the rate of reaction. A small amount of catalyst is required for increasing the rate of reaction for a large amount of reactants. When the reaction takes a longer duration to complete a reaction it shows that the reactant is having higher energy of activation to lower that energy of activation catalyst is used. The catalyst provides another pathway with a lower energy of activation of the reactant to the reaction.
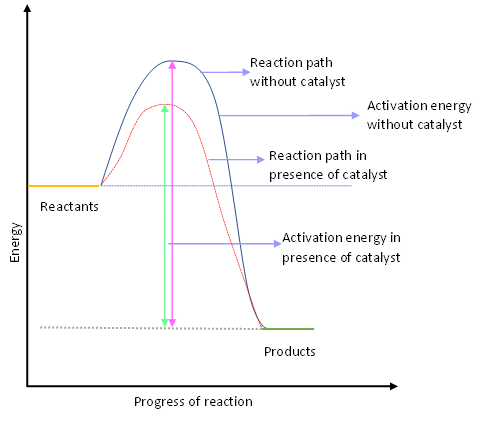
The above graph describes the activity of the catalyst in the reaction.
Thus, Option (A) is correct
Note: Not all reaction gets effect by the catalyst. Some reactions do not change their rate of reaction in presence of a catalyst. One catalyst cannot act as a catalyst in all reactions. Reactions have their suitable and specific catalysts.
Complete answer:A catalyst is a substance, compound, or element which is used in a reaction to change its rate of reaction or to form any specific product in the reaction. Catalyst themselves does not react in the reaction they only provide an alternative way to the reaction. The amount of catalyst used in the reaction remains constant. Catalysts are used to increase or decrease the rate of reaction, generally, it is used to increase the rate of reaction. A small amount of catalyst is required for increasing the rate of reaction for a large amount of reactants. When the reaction takes a longer duration to complete a reaction it shows that the reactant is having higher energy of activation to lower that energy of activation catalyst is used. The catalyst provides another pathway with a lower energy of activation of the reactant to the reaction.

The above graph describes the activity of the catalyst in the reaction.
Thus, Option (A) is correct
Note: Not all reaction gets effect by the catalyst. Some reactions do not change their rate of reaction in presence of a catalyst. One catalyst cannot act as a catalyst in all reactions. Reactions have their suitable and specific catalysts.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Angle of Deviation in a Prism

Understanding Electromagnetic Waves and Their Importance

Understanding Average and RMS Value in Electrical Circuits

How Does Fusion Reaction Happen Inside the Sun?

JEE Main 2025-26 Mock Tests: Practice, Analyze & Improve

Other Pages
NCERT Solutions For Class 12 Chemistry Chapter 8 Aldehydes Ketones And Carboxylic Acids - 2025-26

CBSE Notes Class 12 Chemistry Chapter 3 - Chemical Kinetics - 2025-26

Free Radical Substitution and Its Stepwise Mechanism

CBSE Notes Class 12 Chemistry Chapter 9 - Amines - 2025-26

CBSE Notes Class 12 Chemistry Chapter 7 - Alcohol Phenol and Ether - 2025-26

JEE Advanced 2026 Revision Notes for Practical Organic Chemistry




