What is not true about ice?
A. It has open cage like structure
B. It has less density than water
C. Each \[O\]atom is surrounded by \[4\,H\]atoms
D. Each \[O\]atom has four \[H\]-bonds around it
Answer
268.5k+ views
Hint: Ice is a material that solidifies when liquid water or water vapour is frozen. When it is below zero degrees Celsius, water vapour condenses into snowflakes in the clouds and frost on the ground. When a system is in a static equilibrium, a reaction is totally stopped and there is no movement between the reactants and products that correspond to the chemical reaction.
Complete Step-by-Step Explanation:
In order to know when ice or solid water is described as a normally liquid fluid that freezes to a solid-state at a temperature of \[0^\circ C\]or below and expands to the gaseous form at a temperature of \[100^\circ C\] or higher. Water is an amazing substance that exhibits nearly no normal physical or chemical characteristics. typically. It is the most complex single-chemical compound known. A water molecule's three-dimensional structure can be represented as a tetrahedron, with an oxygen nucleus at the centre and four legs that may each have a high electron density.
At the normal air temperatures and pressures which is close to \[0^\circ C\]and the ice crystal that is often to take the form of the planes or some sheets of the oxygen atoms that are joined in an open hexagonal ring series. Additionally, the optical axis of the crystal structure is the \[c - \]axis which is also known as the parallel to the hexagonal rings axis.
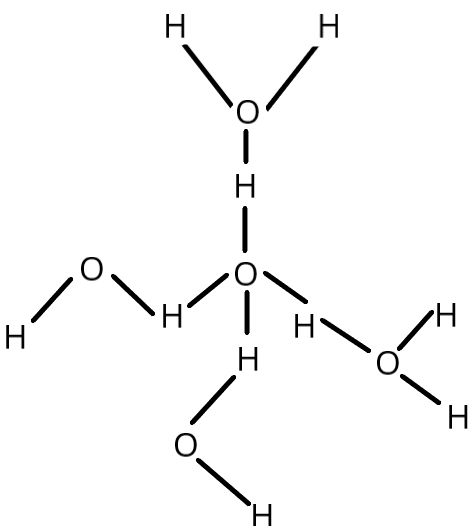
As seen in the image, there are two \[H - \]bonds around each \[O\] atom. Covalent bonds are used to join two \[H\].
Thus, the correct option is:(D) Each \[O\]atom has four \[H\]-bonds around it
Note: It should be noted that the steam which is water in its gaseous condition begins to change into the liquid form when the energy is removed because the temperature of the water molecules is dropping. And condensation is the term for this occurrence. Our daily lives allow us to observe this phenomenon. All of these phenomena are significant components of our natural water cycle.
Complete Step-by-Step Explanation:
In order to know when ice or solid water is described as a normally liquid fluid that freezes to a solid-state at a temperature of \[0^\circ C\]or below and expands to the gaseous form at a temperature of \[100^\circ C\] or higher. Water is an amazing substance that exhibits nearly no normal physical or chemical characteristics. typically. It is the most complex single-chemical compound known. A water molecule's three-dimensional structure can be represented as a tetrahedron, with an oxygen nucleus at the centre and four legs that may each have a high electron density.
At the normal air temperatures and pressures which is close to \[0^\circ C\]and the ice crystal that is often to take the form of the planes or some sheets of the oxygen atoms that are joined in an open hexagonal ring series. Additionally, the optical axis of the crystal structure is the \[c - \]axis which is also known as the parallel to the hexagonal rings axis.

As seen in the image, there are two \[H - \]bonds around each \[O\] atom. Covalent bonds are used to join two \[H\].
Thus, the correct option is:(D) Each \[O\]atom has four \[H\]-bonds around it
Note: It should be noted that the steam which is water in its gaseous condition begins to change into the liquid form when the energy is removed because the temperature of the water molecules is dropping. And condensation is the term for this occurrence. Our daily lives allow us to observe this phenomenon. All of these phenomena are significant components of our natural water cycle.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




