Isoprene is:
(A) 3-methyl-1,2-butadiene
(B) 2-methyl-1,3-butadiene
(C) 3-chloro-1, -butadiene
(D) 2-chloro-1,3-butadiene
Answer
269.4k+ views
Hint: Isoprene is mainly used in manufacturing of rubber. It is an unsaturated hydrocarbon with a methyl group.
Complete step by step answer:
The structure of isoprene is –
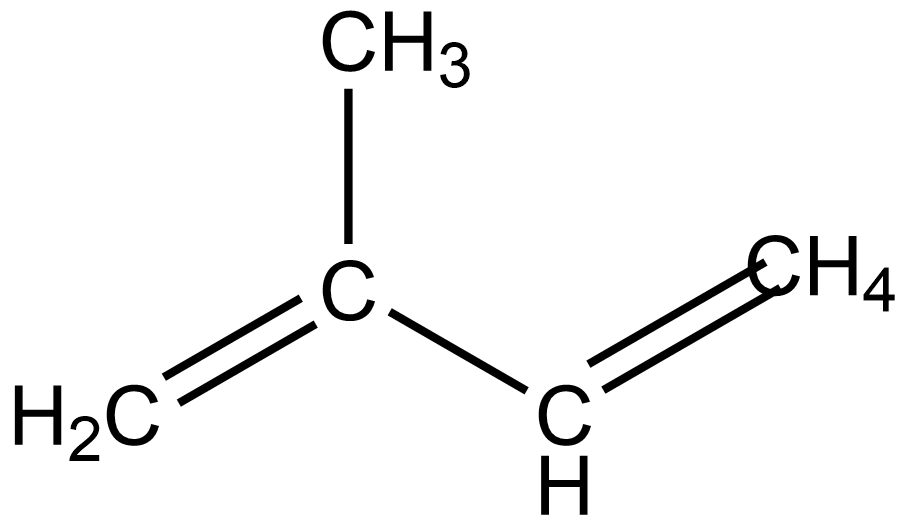
So, the name of this structure will be 2-methyl-1,3-butadiene.So, option B is correct.
Additional information:
It is an unsaturated hydrocarbon produced by many plants and animals. It is a colourless, volatile liquid in its pure form. Polymer of isoprene is a main component in production of natural rubber.
Natural Rubber and Synthetic Rubber are two types of rubber —
Natural rubbers are made up of solid particles suspended in a milky white liquid. They are found dripping from the bark of tropical and subtropical trees. We also call it Latex. It is made by the polymerization of isoprene i.e. 2-methyl-1,3-butadiene. They are made by loosely joining the monomers of isoprene in the form of a long chain.
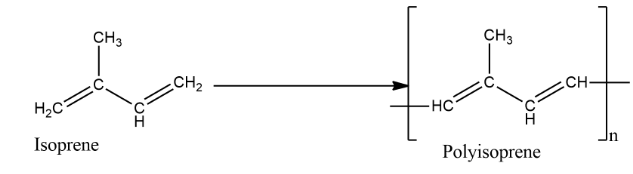
The polymeric chains in rubber contain between 320 and 35,000 isoprene units bound together with double bonds. This makes the chains stretchy. Large quantities of isoprene are used industrially today, nearly all of it to make synthetic rubber, and it is produced by the petroleum industry through cracking oil. Due to its outstanding mechanical properties and low cost, isoprene rubber is the preferred material for many engineering applications.
Note: Polymers of isoprene are mainly used in making of tires, anti-vibration mounts, drive couplings, bearings, and adhesives.
Complete step by step answer:
The structure of isoprene is –

So, the name of this structure will be 2-methyl-1,3-butadiene.So, option B is correct.
Additional information:
It is an unsaturated hydrocarbon produced by many plants and animals. It is a colourless, volatile liquid in its pure form. Polymer of isoprene is a main component in production of natural rubber.
Natural Rubber and Synthetic Rubber are two types of rubber —
Natural rubbers are made up of solid particles suspended in a milky white liquid. They are found dripping from the bark of tropical and subtropical trees. We also call it Latex. It is made by the polymerization of isoprene i.e. 2-methyl-1,3-butadiene. They are made by loosely joining the monomers of isoprene in the form of a long chain.

The polymeric chains in rubber contain between 320 and 35,000 isoprene units bound together with double bonds. This makes the chains stretchy. Large quantities of isoprene are used industrially today, nearly all of it to make synthetic rubber, and it is produced by the petroleum industry through cracking oil. Due to its outstanding mechanical properties and low cost, isoprene rubber is the preferred material for many engineering applications.
Note: Polymers of isoprene are mainly used in making of tires, anti-vibration mounts, drive couplings, bearings, and adhesives.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




