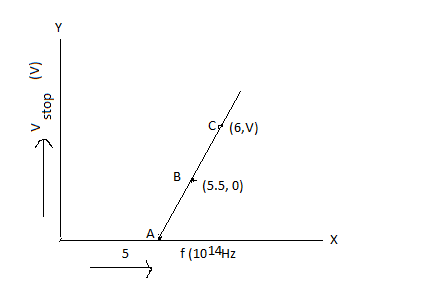
Given figure shows few data points in a photo-electric experiment for a certain metal. The minimum energy for ejection of electron from its surface is: (Planck’s constant \[h = 6.62 \times {10^{ - 34}}\,{\rm{J}}{\rm{.s}}\] )
A. 2.10 eV
B. 2.27 eV
C. 2.59 eV
D. 1.93 eV
Answer
249.6k+ views
Hint: The electrons get emitted from the metallic surface on exposing it to a suitable frequency light. This phenomenon is termed photoelectric effect. Some light energies such as UV rays, X rays, Infrared rays etc. cause photoelectric effect.
Formula used:
Formula of threshold energy \[\varphi \] is,
\[\varphi = h{f_0}\]
Where, h is Planck’s constant and \[{f_0}\] is frequency.
Complete Step by Step Solution:
Let’s understand threshold energy first. The threshold energy means the minimum requirement of energy for a reaction to undergo.
In the given question, a graph of frequency and stopping potential is shown for the photoelectric effect of the metal. Now, we have to calculate the threshold energy using the values given in the graph.
We should consider the frequency of point B, to calculate the threshold energy of the metal. Because at this point, stopping potential is zero.
So, \[f = 5.5 \times {10^{14}}\,{\rm{Hz}}\] and \[h = 6.62 \times {10^{ - 34}}\,{\rm{J}}{\rm{.s}}\].
Now, Putting the value of f and h in the threshold energy formula, that is,\[\varphi = h{f_0}\]
\[\varphi = 6.62 \times {10^{ - 34}}\,{\rm{J}}{\rm{.s}} \times 5.5 \times {10^{14}}\,{\rm{Hz}}\]
\[\varphi \,{\rm{ = }}\;{\rm{36}}{\rm{.41}} \times {\rm{1}}{{\rm{0}}^{ - 20}}\,{\rm{Hz}}\]
Now, we have to convert energy from Hertz to electron volt.
1 electron volt=\[1.6021 \times {10^{ - 19}}\,{\rm{J}}\]
So, \[\varphi \,{\rm{ = }}\;\dfrac{{{\rm{36}}{\rm{.41}} \times {\rm{1}}{{\rm{0}}^{ - 20}}\,}}{{1.6021 \times {{10}^{ - 19}}}} = 22.73 \times {10^{ - 1}} = 2.27\,\,{\rm{eV}}\]
Therefore, the correct threshold energy is 2.27 eV. Hence, the correct option is B.
Note: It is to be noted that photoelectric effect is also sometimes shown by non-metals. To a limited extent, gases and liquids also show this effect. The emitted electrons due to the photoelectric effect is known as photoelectrons.
Formula used:
Formula of threshold energy \[\varphi \] is,
\[\varphi = h{f_0}\]
Where, h is Planck’s constant and \[{f_0}\] is frequency.
Complete Step by Step Solution:
Let’s understand threshold energy first. The threshold energy means the minimum requirement of energy for a reaction to undergo.
In the given question, a graph of frequency and stopping potential is shown for the photoelectric effect of the metal. Now, we have to calculate the threshold energy using the values given in the graph.
We should consider the frequency of point B, to calculate the threshold energy of the metal. Because at this point, stopping potential is zero.
So, \[f = 5.5 \times {10^{14}}\,{\rm{Hz}}\] and \[h = 6.62 \times {10^{ - 34}}\,{\rm{J}}{\rm{.s}}\].
Now, Putting the value of f and h in the threshold energy formula, that is,\[\varphi = h{f_0}\]
\[\varphi = 6.62 \times {10^{ - 34}}\,{\rm{J}}{\rm{.s}} \times 5.5 \times {10^{14}}\,{\rm{Hz}}\]
\[\varphi \,{\rm{ = }}\;{\rm{36}}{\rm{.41}} \times {\rm{1}}{{\rm{0}}^{ - 20}}\,{\rm{Hz}}\]
Now, we have to convert energy from Hertz to electron volt.
1 electron volt=\[1.6021 \times {10^{ - 19}}\,{\rm{J}}\]
So, \[\varphi \,{\rm{ = }}\;\dfrac{{{\rm{36}}{\rm{.41}} \times {\rm{1}}{{\rm{0}}^{ - 20}}\,}}{{1.6021 \times {{10}^{ - 19}}}} = 22.73 \times {10^{ - 1}} = 2.27\,\,{\rm{eV}}\]
Therefore, the correct threshold energy is 2.27 eV. Hence, the correct option is B.
Note: It is to be noted that photoelectric effect is also sometimes shown by non-metals. To a limited extent, gases and liquids also show this effect. The emitted electrons due to the photoelectric effect is known as photoelectrons.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




