Explain the formation of the following molecules using valence bond theory
(a) ${N_2}$ molecules
(b) ${O_2}$ molecules
Answer
258.9k+ views
Hint: Valence bond theory explains formation of molecule. If in a system, force of attraction between electrons of an atom and nucleus of another atom increases, energy of the system decreases and then the possibility of chemical bonding exists.
Complete step by step answer:
We can explain the formation of ${O_2}$ and ${N_2}$ molecules on the basis of valence bond theory. A covalent bond is formed by partial overlay of two half-filled atomic orbitals containing electrons with opposite spin.
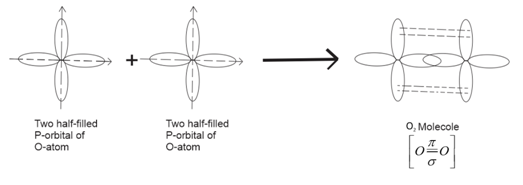
Formation of ${O_2}$molecule:
Electronic configuration of oxygen is, $1{s^2}2{s^2}2p_x^22p_y^12p_2^1.$
When an atom of oxygen approaches another atom unpaired electrons present in $2{p_y}$ and $2{p_z}$ orbital overlap and form covalent bonds.
Two atoms are held by one $\sigma $-bond and one $\pi $-bond.
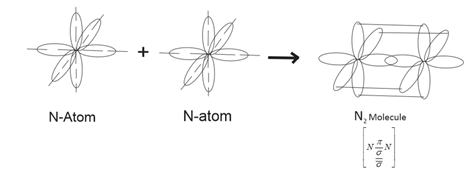
Formation of ${N_2}$ molecule:
Electronic configuration of nitrogen is, $1{s^2}2{s^2}2p_x^12p_y^12p_2^1.$
When one atom of Nitrogen approaches another atom of $N$,then$2{p_x},2{p_y}$ and $2{p_z}$ atomic orbitals overlap and form a covalent bond. As orbitals approach each other the energy of the system gets lower and forms stable molecules both nitrogen atoms held together by one $\sigma $ and two $\pi $ bonds.
Sigma bonds form by overlap of atomic orbitals of two atoms along the internuclear axis.
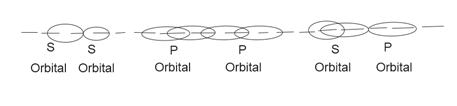
P1 $(\pi )$ bond formed by lateral overlapping of p-orbitals.

Note: Oxygen molecule has double bond between two O-atoms one bond is sigma bond and other bond is $\pi $ bond. Nitrogen molecules have a Triple bond between two N-atoms of which one bond is a sigma bond and two bonds or $\pi $-bonds.
Triple bond is stronger than double bond.
Complete step by step answer:
We can explain the formation of ${O_2}$ and ${N_2}$ molecules on the basis of valence bond theory. A covalent bond is formed by partial overlay of two half-filled atomic orbitals containing electrons with opposite spin.
Formation of ${O_2}$molecule:
Electronic configuration of oxygen is, $1{s^2}2{s^2}2p_x^22p_y^12p_2^1.$
When an atom of oxygen approaches another atom unpaired electrons present in $2{p_y}$ and $2{p_z}$ orbital overlap and form covalent bonds.
Two atoms are held by one $\sigma $-bond and one $\pi $-bond.

Formation of ${N_2}$ molecule:
Electronic configuration of nitrogen is, $1{s^2}2{s^2}2p_x^12p_y^12p_2^1.$
When one atom of Nitrogen approaches another atom of $N$,then$2{p_x},2{p_y}$ and $2{p_z}$ atomic orbitals overlap and form a covalent bond. As orbitals approach each other the energy of the system gets lower and forms stable molecules both nitrogen atoms held together by one $\sigma $ and two $\pi $ bonds.

Sigma bonds form by overlap of atomic orbitals of two atoms along the internuclear axis.

P1 $(\pi )$ bond formed by lateral overlapping of p-orbitals.

Note: Oxygen molecule has double bond between two O-atoms one bond is sigma bond and other bond is $\pi $ bond. Nitrogen molecules have a Triple bond between two N-atoms of which one bond is a sigma bond and two bonds or $\pi $-bonds.
Triple bond is stronger than double bond.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




