When ethene reacts with bromine, it forms
A) Chloroethane
B) Ethylene dibromide
C) 1-bromopropane
D) 1,2-dichloroethene
Answer
256.2k+ views
Hint: We know that ethene is the simplest of alkenes having one double bond. It is present in the gaseous state having a sweet flavour. And we know that bromine is a halogen of red-brown colour present in the liquid state.
Complete Step by Step Answer:
Let’s discuss the reaction of bromine with ethene. Ethene, when a reaction with liquid bromine, or with a bromine solution in tetrachloromethane which is an organic solvent, breaks the double bond of ethene and attaching of one bromine atom at each carbon takes place. Therefore, the product formed in this reaction is dibromo-ethane or ethylene dibromide. The chemical reaction is,
\[{\rm{C}}{{\rm{H}}_{\rm{2}}} = {\rm{C}}{{\rm{H}}_{\rm{2}}} + {\rm{B}}{{\rm{r}}_{\rm{2}}} \to \mathop {{\rm{Br}} - {\rm{C}}{{\rm{H}}_{\rm{2}}} - {\rm{C}}{{\rm{H}}_{\rm{2}}} - {\rm{Br}}}\limits_{{\rm{Ethylene}}\,{\rm{bromide}}} \]
Let’s understand the mechanism of the reaction. In the first step, the attachment of one bromine atom to both carbon atoms. And the bromine atom is positively charged.
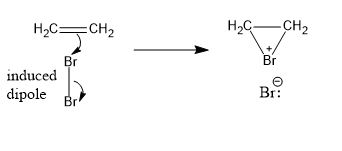
Image: Formation of bromonium ion
In the second step, the attack of the bromide ion at the bromonium ion from the back side happens. The resultant product is ethylene dibromide. The reaction is:
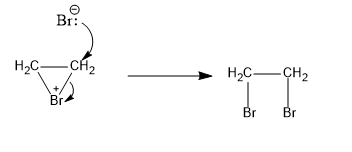
Image: Formation of ethylene dibromide
Therefore, the reaction of ethene and bromine gives ethylene dibromide.
Hence, option B is right.
Note: The reaction of ethene and bromine is an electrophilic addition. The electrophilic addition defines an addition reaction of a nucleophile and an electrophile that causes the addition of a double or a triple bond. An electrophile defines a species that has a tendency to react with other molecules having a donatable electron pair.
Complete Step by Step Answer:
Let’s discuss the reaction of bromine with ethene. Ethene, when a reaction with liquid bromine, or with a bromine solution in tetrachloromethane which is an organic solvent, breaks the double bond of ethene and attaching of one bromine atom at each carbon takes place. Therefore, the product formed in this reaction is dibromo-ethane or ethylene dibromide. The chemical reaction is,
\[{\rm{C}}{{\rm{H}}_{\rm{2}}} = {\rm{C}}{{\rm{H}}_{\rm{2}}} + {\rm{B}}{{\rm{r}}_{\rm{2}}} \to \mathop {{\rm{Br}} - {\rm{C}}{{\rm{H}}_{\rm{2}}} - {\rm{C}}{{\rm{H}}_{\rm{2}}} - {\rm{Br}}}\limits_{{\rm{Ethylene}}\,{\rm{bromide}}} \]
Let’s understand the mechanism of the reaction. In the first step, the attachment of one bromine atom to both carbon atoms. And the bromine atom is positively charged.

Image: Formation of bromonium ion
In the second step, the attack of the bromide ion at the bromonium ion from the back side happens. The resultant product is ethylene dibromide. The reaction is:

Image: Formation of ethylene dibromide
Therefore, the reaction of ethene and bromine gives ethylene dibromide.
Hence, option B is right.
Note: The reaction of ethene and bromine is an electrophilic addition. The electrophilic addition defines an addition reaction of a nucleophile and an electrophile that causes the addition of a double or a triple bond. An electrophile defines a species that has a tendency to react with other molecules having a donatable electron pair.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




