Colour of CuO is
A. Black
B. Blue
C. Green
D. Red
Answer
262.5k+ views
Hint: In order to answer the question , we need to have a detailed idea about the structure of CuO. We should also be familiar with the physical and chemical properties of CuO.
Complete step-by-step answer:
Let us have a look into the structure of CuO.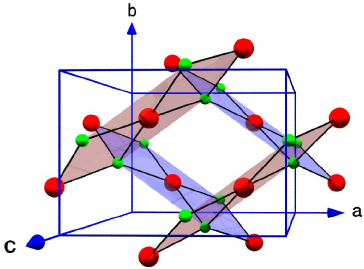
The red structures are the Copper atoms and the green structures are the oxygen atoms.
The atomic number of copper is 29. And that of oxygen is 8.
So the electronic configurations of both the elements are given below:
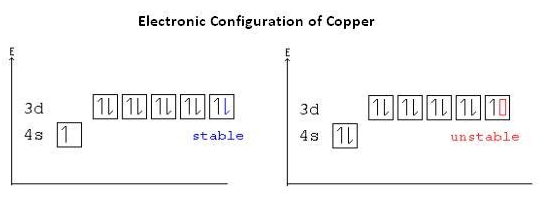
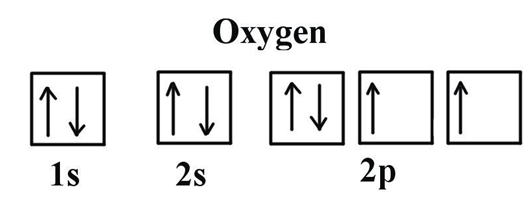
The main factor to occur the colour of the compounds of d block, is having the d orbitals with an incomplete electron filling in the middle atom.
There is an inequality of energy, among those d orbitals. Therefore d-d electrons exchange among those orbitals by absorbing energy. The amount of energy consumption causes the colour of the compound. Here, different amounts of energy need to exchange different numbers of electrons. Thereby the wavelengths absorbed in the process changes, and consequently different colours occur.
The colour of CuO is known to be Black. So the correct option is Option A.
Note: We should be knowing the few uses of CuO. So here are some uses of CuO listed below:
Used in the welding process along with various copper alloys.
Used as a precursor in various copper containing products like wood preservatives and ceramics.
Some properties of CuO are:
Super Thermal Conductivity.
CuO shows photovoltaic properties.
It has high stability and high antimicrobial activity.
Complete step-by-step answer:
Let us have a look into the structure of CuO.

The red structures are the Copper atoms and the green structures are the oxygen atoms.
The atomic number of copper is 29. And that of oxygen is 8.
So the electronic configurations of both the elements are given below:


The main factor to occur the colour of the compounds of d block, is having the d orbitals with an incomplete electron filling in the middle atom.
There is an inequality of energy, among those d orbitals. Therefore d-d electrons exchange among those orbitals by absorbing energy. The amount of energy consumption causes the colour of the compound. Here, different amounts of energy need to exchange different numbers of electrons. Thereby the wavelengths absorbed in the process changes, and consequently different colours occur.
The colour of CuO is known to be Black. So the correct option is Option A.
Note: We should be knowing the few uses of CuO. So here are some uses of CuO listed below:
Used in the welding process along with various copper alloys.
Used as a precursor in various copper containing products like wood preservatives and ceramics.
Some properties of CuO are:
Super Thermal Conductivity.
CuO shows photovoltaic properties.
It has high stability and high antimicrobial activity.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




