Can two isothermal curves cut each other?
A. Never
B. Yes
C. They will cut when temperature is ${0^ \circ }C$
D. Yes, when the pressure is critical pressure
Answer
256.5k+ views
Hint: In an Isothermal process in a thermodynamic system, the temperature is constant and at a constant temperature, both the parameters pressure and volume vary with the different conditions of the system and surroundings hence, we will plot a $P - V$ graph for an isothermal change for two different temperatures to state the answer of this problem.
Complete step by step solution:
An isothermal process in thermodynamics is defined as the process during which the temperature $T$ of a system remains constant that’s why it is also referred to as a constant-temperature process.
In an Isothermal process, $T = \text{constant}$ and $\text{Change in Temperature} = \Delta T = 0$. Now let us draw a $P - V$ graph for an isothermal expansion of a gas at two different temperatures to have two isothermal curves as shown below: -
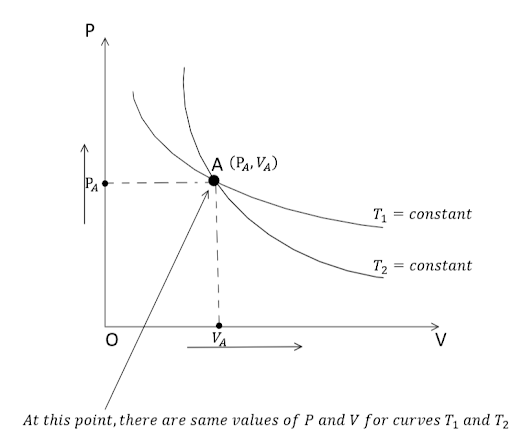
Clearly, from the above graph it can be observed that if the two isothermal curves intersect at point A, there will be the same values of pressure and volume i.e., ${P_A}$ and ${V_A}$ at that point. That’s why if the two curves intersect, the volume and pressure of the gas will be the same at two distinct isothermal temperatures, which is not possible. As a result, two isothermal curves can ‘Never’ intersect.
Hence, the correct option is A.
Note: In this problem, to determine whether the two isothermal curves intersect with each other or not, we need to find the conditions of other parameters of gas in a particular thermodynamic system such as pressure and volume hence, we will plot a $P - V$ graph and if the conditions of pressure and volume are justified for the given situation then the curves can intersect otherwise not.
Complete step by step solution:
An isothermal process in thermodynamics is defined as the process during which the temperature $T$ of a system remains constant that’s why it is also referred to as a constant-temperature process.
In an Isothermal process, $T = \text{constant}$ and $\text{Change in Temperature} = \Delta T = 0$. Now let us draw a $P - V$ graph for an isothermal expansion of a gas at two different temperatures to have two isothermal curves as shown below: -

Clearly, from the above graph it can be observed that if the two isothermal curves intersect at point A, there will be the same values of pressure and volume i.e., ${P_A}$ and ${V_A}$ at that point. That’s why if the two curves intersect, the volume and pressure of the gas will be the same at two distinct isothermal temperatures, which is not possible. As a result, two isothermal curves can ‘Never’ intersect.
Hence, the correct option is A.
Note: In this problem, to determine whether the two isothermal curves intersect with each other or not, we need to find the conditions of other parameters of gas in a particular thermodynamic system such as pressure and volume hence, we will plot a $P - V$ graph and if the conditions of pressure and volume are justified for the given situation then the curves can intersect otherwise not.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2025-26

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




