Assertion: Millon’s test is a test for the identification of proteins.
Reason: Millon’s reagent is a solution of mercurous nitrate and mercuric nitrate in nitric acid containing little nitrous acid.
(A) Both Assertion and Reason are correct, and the reason is the correct explanation for the assertion.
(B) Both assertion and reason are correct, but the reason is not the correct explanation for the assertion.
(C) Assertion is correct, but the reason is incorrect.
(D) Both assertion and reason are incorrect.
Answer
266.4k+ views
Hint: Millon’s test is used to detect amino acids. Amino acids are structural units of proteins. When Millon’s reagent is added to an aqueous solution of protein white precipitate is formed which on heating turns red. Millon’s test is used to detect amino acids containing phenol. All proteins contain tyrosine residue.
Complete step by step solution:
-Presence of proteins is detected by adding Millon’s reagent.
-When a few drops of this reagent are added in the aqueous test solution to be tested, then it is heated gently. If reddish-brown colouration or precipitate appears, it indicates the presence of tyrosine.
-Tyrosine is present in nearly all proteins.
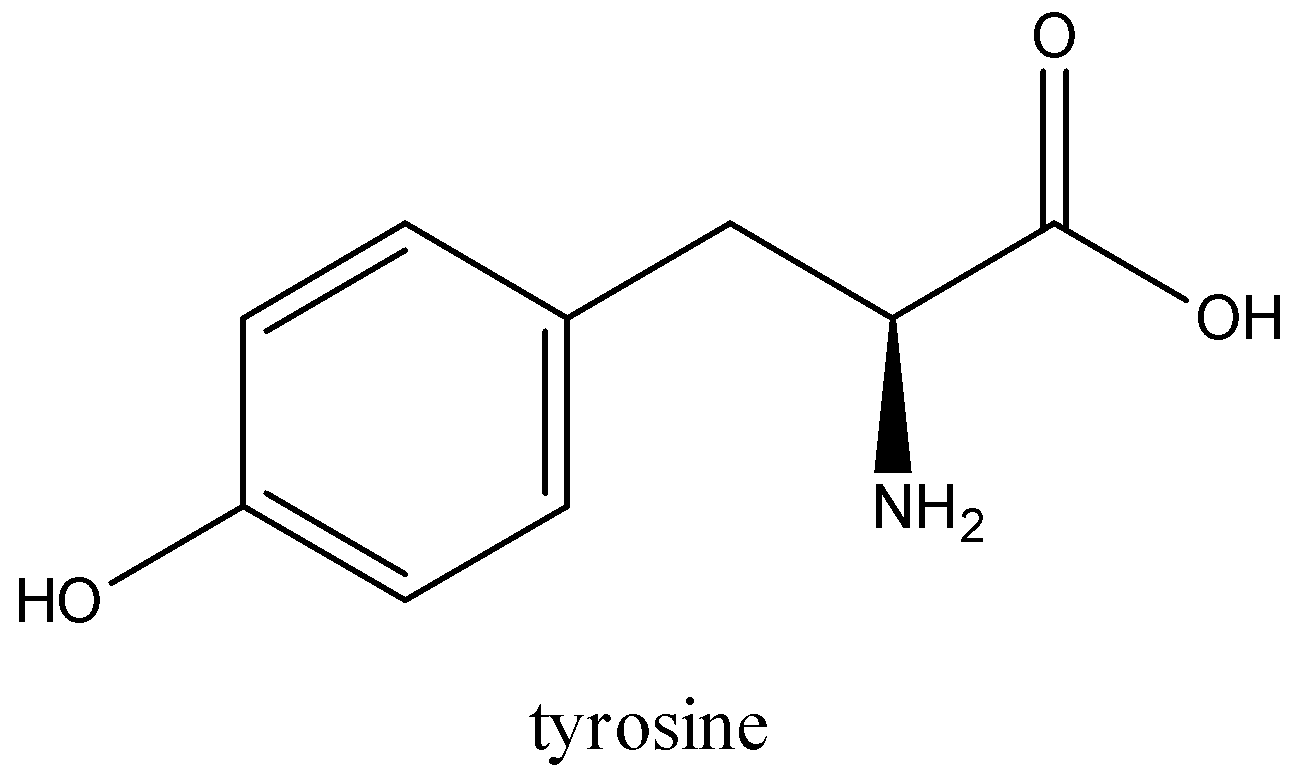
-Compounds having hydroxybenzene radical form red complexes with Millon’s reagent. Tyrosine is only amino acid which has a hydroxybenzene ring.
-Tyrosine with acidified mercuric sulphate solution produces mercury amino acid complex which is yellow precipitate. When sodium nitrate solution is added in this and heated, the mercury amino acid complex gets converted to red colour mercury phenolate.
So, (B) Both assertion and reason are correct, but the reason is not the correct explanation for the assertion.
Additional information:
The correct explanation is When Millon’s reagent is added to an aqueous solution of protein white precipitate is formed which on heating turns red. Millon’s test is used to detect amino acids containing phenol. All proteins contain tyrosine residue.
Note: When a few drops of reagent are added in the aqueous test solution to be tested, then it is heated gently. If reddish-brown colouration or precipitate appears, it indicates the presence of tyrosine. Tyrosine is present in nearly all proteins. Tyrosine with acidified mercuric sulphate solution produces mercury amino acid complex which is yellow precipitate. When sodium nitrate solution is added in this and heated, the mercury amino acid complex gets converted to red colour mercury phenolate.
Complete step by step solution:
-Presence of proteins is detected by adding Millon’s reagent.
-When a few drops of this reagent are added in the aqueous test solution to be tested, then it is heated gently. If reddish-brown colouration or precipitate appears, it indicates the presence of tyrosine.
-Tyrosine is present in nearly all proteins.

-Compounds having hydroxybenzene radical form red complexes with Millon’s reagent. Tyrosine is only amino acid which has a hydroxybenzene ring.
-Tyrosine with acidified mercuric sulphate solution produces mercury amino acid complex which is yellow precipitate. When sodium nitrate solution is added in this and heated, the mercury amino acid complex gets converted to red colour mercury phenolate.
So, (B) Both assertion and reason are correct, but the reason is not the correct explanation for the assertion.
Additional information:
The correct explanation is When Millon’s reagent is added to an aqueous solution of protein white precipitate is formed which on heating turns red. Millon’s test is used to detect amino acids containing phenol. All proteins contain tyrosine residue.
Note: When a few drops of reagent are added in the aqueous test solution to be tested, then it is heated gently. If reddish-brown colouration or precipitate appears, it indicates the presence of tyrosine. Tyrosine is present in nearly all proteins. Tyrosine with acidified mercuric sulphate solution produces mercury amino acid complex which is yellow precipitate. When sodium nitrate solution is added in this and heated, the mercury amino acid complex gets converted to red colour mercury phenolate.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




