Aniline is not a major product in one of the following reactions. Identify that reaction.
A. ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{OH + N}}{{\text{H}}_{\text{3}}}\xrightarrow[{300^\circ {\text{C}}}]{{{\text{ZnC}}{{\text{l}}_{\text{2}}}}}$
B. ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{O}}_2}{\text{ + ZnPowder}}\xrightarrow{{{\text{alcoholicKOH}}}}$
C. ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{Cl + N}}{{\text{H}}_{\text{3}}}\xrightarrow[{{\text{C}}{{\text{u}}_{\text{2}}}{\text{O}}}]{{200^\circ {\text{C}}}}$
D. ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{O}}_2}{\text{ + Fe + }}{{\text{H}}_2}{\text{O}}\xrightarrow{{{\text{HCl}}}}$
Answer
263.1k+ views
Hint: The reduction of nitro compounds with active metals in alkaline medium gives hydrazobenzene.
The catalytic reduction of nitro compounds in acidic medium is a very convenient method of preparing aromatic primary amines as they cannot be prepared from the corresponding aryl halides on treatment with ammonia.
Complete step by step answer:
Let us study the given reactions one by one.
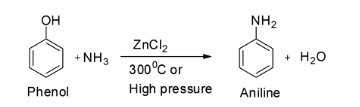
The first reaction is given to be ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{OH + N}}{{\text{H}}_{\text{3}}}\xrightarrow[{300^\circ {\text{C}}}]{{{\text{ZnC}}{{\text{l}}_{\text{2}}}}}$ .
This is a reaction between phenol and ammonia in presence of zinc chloride.
When a mixture of the vapours of phenol and ammonia are passed over zinc chloride, aniline is formed. Since phenols are less reactive towards nucleophilic substitution reactions, so, severe conditions like high pressure are usually employed. Thus, aniline can be prepared by the action of ammonia on phenol. So the major product of the given reaction is aniline and hence option A is not correct.
The next reaction is given to be ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{O}}_2}{\text{ + ZnPowder}}\xrightarrow{{{\text{alcoholicKOH}}}}$ .
This is a reaction between nitrobenzene and zinc powder in presence of alcoholic potassium hydroxide.
It is known that the reduction of nitrobenzene with zinc in presence of alcoholic potassium hydroxide gives hydrazobenzene.
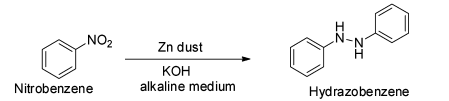
Thus, the reduction of nitrobenzene with zinc in alkaline medium produces hydrazobenzene. So, the major product of this reaction is not aniline and so the option B is correct.
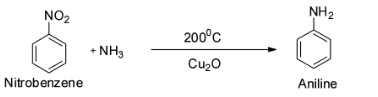
The next reaction is ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{Cl + N}}{{\text{H}}_{\text{3}}}\xrightarrow[{{\text{C}}{{\text{u}}_{\text{2}}}{\text{O}}}]{{200^\circ {\text{C}}}}$ .
This is a reaction between chlorobenzene with ammonia in presence of cuprous oxide. This reaction gives aniline as the major product and so C is wrong.
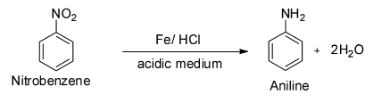
The last reaction is given to be ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{O}}_2}{\text{ + Fe + }}{{\text{H}}_2}{\text{O}}\xrightarrow{{{\text{HCl}}}}$ .
This is a reaction between nitrobenzene and iron in presence of hydrochloric acid.
It is known that the catalytic reduction of nitro compounds with an active metal like iron, tin, zinc etc. in acidic medium like concentrated hydrochloric acid gives aromatic primary amines.
Thus, the given reaction will give aniline as the major product and so option D is incorrect.
Note:
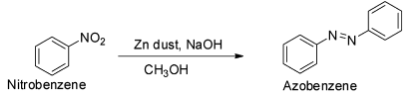
The reduction of nitrobenzene with zinc and methanolic sodium hydroxide gives azobenzene.
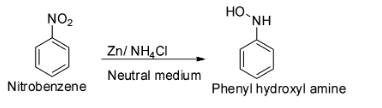
The reduction of nitrobenzene with zinc dust and aqueous ammonium chloride (neutral medium) gives phenyl hydroxylamine.
The catalytic reduction of nitro compounds in acidic medium is a very convenient method of preparing aromatic primary amines as they cannot be prepared from the corresponding aryl halides on treatment with ammonia.
Complete step by step answer:
Let us study the given reactions one by one.
The first reaction is given to be ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{OH + N}}{{\text{H}}_{\text{3}}}\xrightarrow[{300^\circ {\text{C}}}]{{{\text{ZnC}}{{\text{l}}_{\text{2}}}}}$ .
This is a reaction between phenol and ammonia in presence of zinc chloride.
When a mixture of the vapours of phenol and ammonia are passed over zinc chloride, aniline is formed. Since phenols are less reactive towards nucleophilic substitution reactions, so, severe conditions like high pressure are usually employed. Thus, aniline can be prepared by the action of ammonia on phenol. So the major product of the given reaction is aniline and hence option A is not correct.

The next reaction is given to be ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{O}}_2}{\text{ + ZnPowder}}\xrightarrow{{{\text{alcoholicKOH}}}}$ .
This is a reaction between nitrobenzene and zinc powder in presence of alcoholic potassium hydroxide.
It is known that the reduction of nitrobenzene with zinc in presence of alcoholic potassium hydroxide gives hydrazobenzene.

Thus, the reduction of nitrobenzene with zinc in alkaline medium produces hydrazobenzene. So, the major product of this reaction is not aniline and so the option B is correct.
The next reaction is ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{Cl + N}}{{\text{H}}_{\text{3}}}\xrightarrow[{{\text{C}}{{\text{u}}_{\text{2}}}{\text{O}}}]{{200^\circ {\text{C}}}}$ .
This is a reaction between chlorobenzene with ammonia in presence of cuprous oxide. This reaction gives aniline as the major product and so C is wrong.

The last reaction is given to be ${{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{O}}_2}{\text{ + Fe + }}{{\text{H}}_2}{\text{O}}\xrightarrow{{{\text{HCl}}}}$ .
This is a reaction between nitrobenzene and iron in presence of hydrochloric acid.
It is known that the catalytic reduction of nitro compounds with an active metal like iron, tin, zinc etc. in acidic medium like concentrated hydrochloric acid gives aromatic primary amines.
Thus, the given reaction will give aniline as the major product and so option D is incorrect.

Note:
The reduction of nitrobenzene with zinc and methanolic sodium hydroxide gives azobenzene.

The reduction of nitrobenzene with zinc dust and aqueous ammonium chloride (neutral medium) gives phenyl hydroxylamine.

Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




