Aniline in a set of reactions yielded a product D?
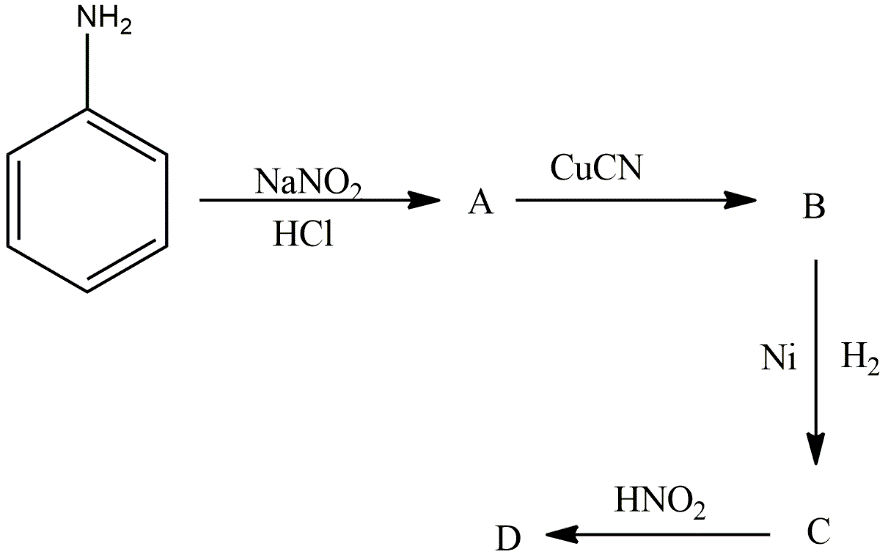
The product D is:
A.\[{C_6}{H_5}NHOH\]
B.\[{C_6}{H_5}NHC{H_2}C{H_3}\]
C.\[{C_6}{H_5}C{H_2}N{H_2}\]
D.\[{C_6}{H_5}C{H_2}OH\]
Answer
254.1k+ views
Hint: Aniline is an aromatic amine having a primary aromatic amine functional group. The reaction here is an example of a coupling reaction where carbon couples with carbon to form a C-C single bond.
Complete Step by Step Solution:
In the first step, \[\left( {NaN{O_2} + HCl} \right)\]reacts with each other to form nitrous acid\[\left( {HONO} \right)\], which reacts with aniline to give a diazotization reaction and form benzene diazonium chloride salt product (A). This product (A) further reacts with copper cyanide \[\left( {CuCN} \right)\]to give a Sandmayer’s reaction and forms cyano benzene or benzonitrile product (B). Then product (B) reacts with hydrogen gas in presence of nickel catalyst to give benzylamine (C) by reduction, which further reacts with nitrous acid \[\left( {HONO} \right)\]which is an oxidising agent and forms benzyl alcohol, the final product (D). The reaction mechanism is shown below:
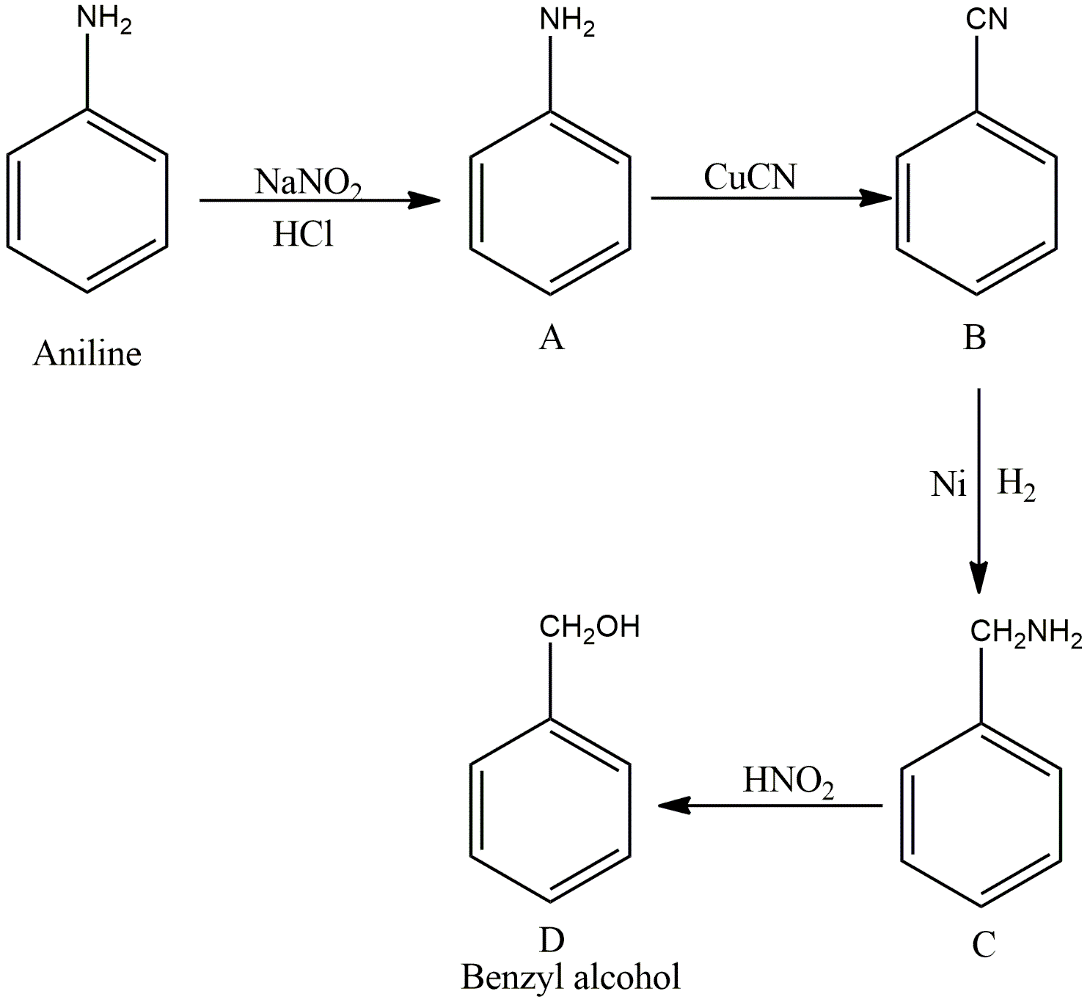
Image:Reaction with aniline to give benzyl alcohol
So, option D is correct.
Additional Information:Aromatic diazonium chloride salts formed by the reaction of aniline and nitrous acid are electrophiles and undergo electrophilic substitution reaction i.e., electron-loving compounds attack them. In this way, it works in coupling reactions.
In the reaction of copper cyanide with aromatic diazonium chloride salt, partial positive charges on Cu forms a bond with negative charges on chloride ion of salts and form \[CuCl\]and \[{N_2}\]releases as nitrogen gas and partial negative charges on cyanide attack the aromatic ring to form cyanobenzene.
Note: Anilines are also known as phenyl benzene or aminobenzene. Nitrous acid is a weak acid used in the preparation of diazonium salts. The reaction asked in this question is an azo coupling reaction used to form C-C bonds.
Complete Step by Step Solution:
In the first step, \[\left( {NaN{O_2} + HCl} \right)\]reacts with each other to form nitrous acid\[\left( {HONO} \right)\], which reacts with aniline to give a diazotization reaction and form benzene diazonium chloride salt product (A). This product (A) further reacts with copper cyanide \[\left( {CuCN} \right)\]to give a Sandmayer’s reaction and forms cyano benzene or benzonitrile product (B). Then product (B) reacts with hydrogen gas in presence of nickel catalyst to give benzylamine (C) by reduction, which further reacts with nitrous acid \[\left( {HONO} \right)\]which is an oxidising agent and forms benzyl alcohol, the final product (D). The reaction mechanism is shown below:

Image:Reaction with aniline to give benzyl alcohol
So, option D is correct.
Additional Information:Aromatic diazonium chloride salts formed by the reaction of aniline and nitrous acid are electrophiles and undergo electrophilic substitution reaction i.e., electron-loving compounds attack them. In this way, it works in coupling reactions.
In the reaction of copper cyanide with aromatic diazonium chloride salt, partial positive charges on Cu forms a bond with negative charges on chloride ion of salts and form \[CuCl\]and \[{N_2}\]releases as nitrogen gas and partial negative charges on cyanide attack the aromatic ring to form cyanobenzene.
Note: Anilines are also known as phenyl benzene or aminobenzene. Nitrous acid is a weak acid used in the preparation of diazonium salts. The reaction asked in this question is an azo coupling reaction used to form C-C bonds.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




