Alkyl cyanides when react with Grignard reagent, the product on hydrolysis found is
A. Aldehyde
B. Ketone
C. Alcohol
D. Acid
Answer
266.7k+ views
Hint: Toluene is a transparent, colourless liquid that, when heated to room temperature, turns into a vapour. Toluene is a naturally occurring component of crude oil and is used in the production of paints, lacquers, explosives (TNT), and glues, in addition to oil refining. Toluene exposure can lead to a variety of symptoms, including irritation of the eyes and nose, exhaustion, headache, dilated pupils, tears, anxiety, muscle fatigue, sleeplessness, and nerve damage.
Complete Step by Step Answer:
Grignard reagent has two parts. In the Grignard reagent the alkyl part acts as a nucleophile that is electron efficient species and wants to donate its electrons. In this reaction in the first step the alkyl part of the Grignard reagent attacks the carbon atom of the alkyl cyanide and the metal part of the Grignard reagent that is magnesium part connects with the nitrogen atom of cyanide.
In the next step water attacks the carbon atom and makes ketone along with ammonia as a side product. The reaction can be given as follows:
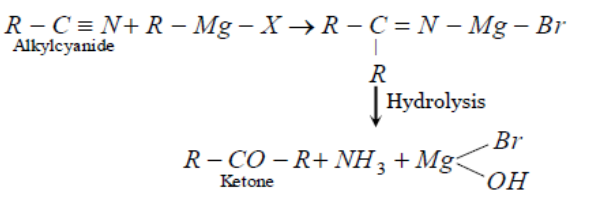
Thus we can write that Alkyl cyanides when react with Grignard reagent, the product on hydrolysis found is ketone.
Thus the correct option is B.
Note: Grignard reagent is alkyl or aryl magnesium halide type of compound. In Grignard reagent the alkyl or aryl part acts as a nucleophile and the metal part gets positive charge. Grignard reagents can convert aldehyde and ketone to alcohol. It can also convert acid to alcohol. In these Grignard reagents the metal that is magnesium acts as a hard species.
Complete Step by Step Answer:
Grignard reagent has two parts. In the Grignard reagent the alkyl part acts as a nucleophile that is electron efficient species and wants to donate its electrons. In this reaction in the first step the alkyl part of the Grignard reagent attacks the carbon atom of the alkyl cyanide and the metal part of the Grignard reagent that is magnesium part connects with the nitrogen atom of cyanide.
In the next step water attacks the carbon atom and makes ketone along with ammonia as a side product. The reaction can be given as follows:
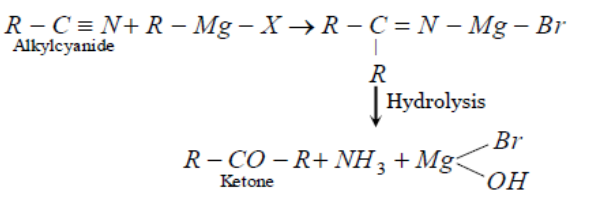
Thus we can write that Alkyl cyanides when react with Grignard reagent, the product on hydrolysis found is ketone.
Thus the correct option is B.
Note: Grignard reagent is alkyl or aryl magnesium halide type of compound. In Grignard reagent the alkyl or aryl part acts as a nucleophile and the metal part gets positive charge. Grignard reagents can convert aldehyde and ketone to alcohol. It can also convert acid to alcohol. In these Grignard reagents the metal that is magnesium acts as a hard species.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




