When acetamide is treated with $HN{{O}_{2}}$, gas evolved is
A. ${{H}_{2}}$
B. ${{O}_{2}}$
C. ${{N}_{2}}$
D. $C{{H}_{4}}$
Answer
262.2k+ views
Hint: Amides undergo hydrolysis under acidic or basic medium. An indirect process of hydrolysis is the reaction with nitrous acid $HN{{O}_{2}}$. Primary amides react with $HN{{O}_{2}}$ and are converted to carboxylic acid but secondary amides give N-nitroso compounds.
Complete step by step solution:
The products of amide hydrolysis depend upon whether the whole process occurs under an acidic or basic medium. Because under an acidic medium it forms carboxylic acid and in the basic condition carboxylic acid salts are formed. Acetamide is a kind of primary amide that reacts with nitrous acid under acidic conditions to give acetic acid, water, and nitrogen gas releases. The structure of acetamide is:
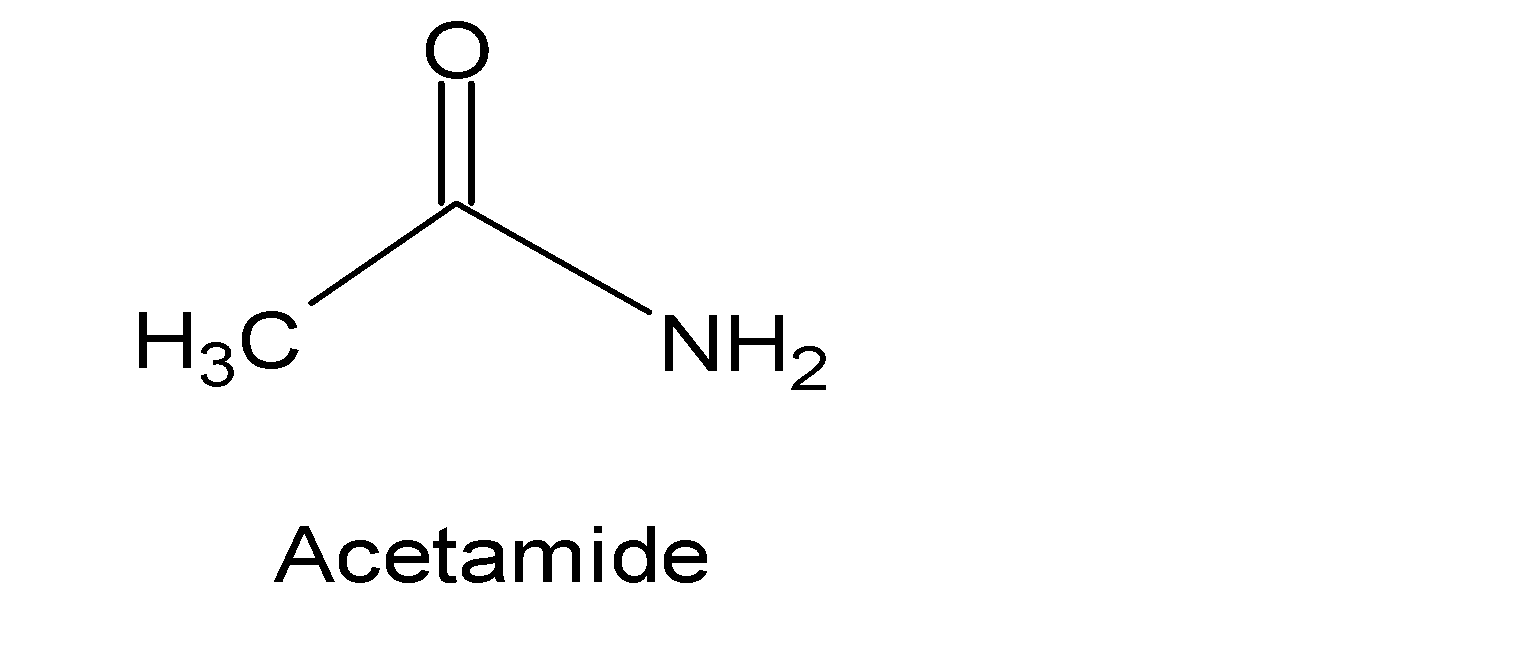
The main reactive group of this particular chemical reaction is the nitrosonium ion. The lone pair of nitrogen atoms attack the nitrosonium ion and form an intermediate N-nitrosamine. This intermediate undergoes proton shift by a sequence of keto-enol tautomerism to form diazoic acid followed by decompositions to diazonium ions in an acidic medium. Finally, diazonium salts decompose to trivalent gaseous nitrogen. An acyl cation$C{{H}_{3}}C{{O}^{+}}$is formed from the substrate acetamide. This carbocation further reacts with water to form acetic acid. The following mechanism gives us a brief idea of how the reaction proceeds.
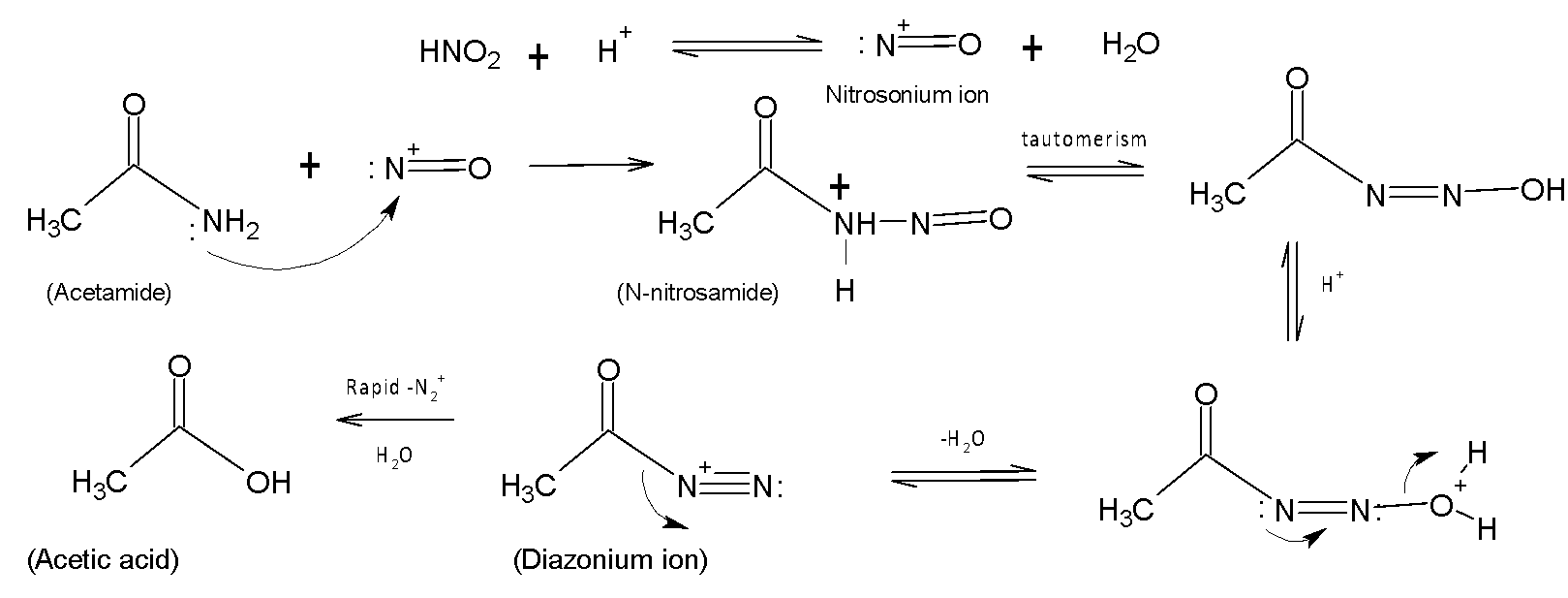
Therefore, acetamide reacts $HN{{O}_{2}}$to form acetic acid, and water and releases nitrogen, ${{N}_{2}}$and gas.
Thus, option (C) is correct.
Note: Amide hydrolysis has many applications in chemistry. Under biological conditions, peptide hydrolysis of protein occurs which is nothing but hydrolysis of an amide. They can also serve as a solvent and as a drug intermediate in the manufacture of (ampicillin, cephalexin, cephradine, etc) in many organic and inorganic synthesis.
Complete step by step solution:
The products of amide hydrolysis depend upon whether the whole process occurs under an acidic or basic medium. Because under an acidic medium it forms carboxylic acid and in the basic condition carboxylic acid salts are formed. Acetamide is a kind of primary amide that reacts with nitrous acid under acidic conditions to give acetic acid, water, and nitrogen gas releases. The structure of acetamide is:

The main reactive group of this particular chemical reaction is the nitrosonium ion. The lone pair of nitrogen atoms attack the nitrosonium ion and form an intermediate N-nitrosamine. This intermediate undergoes proton shift by a sequence of keto-enol tautomerism to form diazoic acid followed by decompositions to diazonium ions in an acidic medium. Finally, diazonium salts decompose to trivalent gaseous nitrogen. An acyl cation$C{{H}_{3}}C{{O}^{+}}$is formed from the substrate acetamide. This carbocation further reacts with water to form acetic acid. The following mechanism gives us a brief idea of how the reaction proceeds.

Therefore, acetamide reacts $HN{{O}_{2}}$to form acetic acid, and water and releases nitrogen, ${{N}_{2}}$and gas.
Thus, option (C) is correct.
Note: Amide hydrolysis has many applications in chemistry. Under biological conditions, peptide hydrolysis of protein occurs which is nothing but hydrolysis of an amide. They can also serve as a solvent and as a drug intermediate in the manufacture of (ampicillin, cephalexin, cephradine, etc) in many organic and inorganic synthesis.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




