A reaction that occurs only when heat is added is best described as:
A. Exothermic
B. Endothermic
C. An equilibrium process
D. Spontaneous
E. Non-spontaneous
Answer
257.4k+ views
Hint: Chemical reaction is a process in which one or more substances are converted to more different substances. Substances may be either chemical elements or compounds. Some reactions may involve energy changes, like absorption of heat energy etc.
Complete step by step answer:
An endothermic reaction is a chemical reaction in which more energy is needed to break bonds in the reactants than the energy released when new bonds are formed in the form of products. These are basically the reactions in which the reactants absorb heat energy from the surroundings to form products. This reaction occurs only when the heat is added.
Moreover, these reactions lower the temperature of their surrounding area, thereby creating a cooling effect.
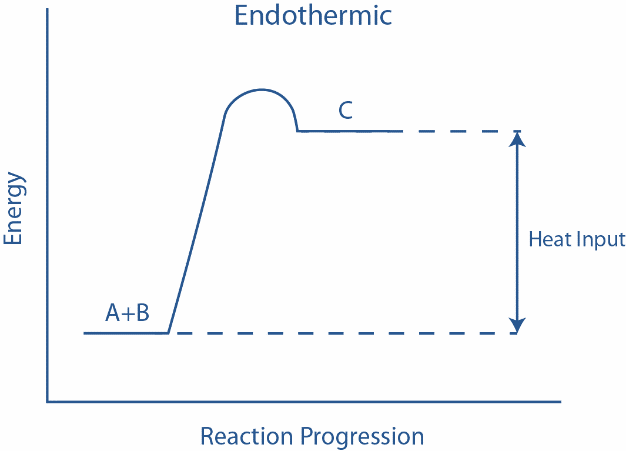
Now, in the given picture, the energy tied up in the molecular bonds is greater in the products than in the reactants. However, there is a need for the activation energy to commence the reaction but, once that hurdle is overcome, the reaction proceeds resulting in the formation of product C and the resulting product has more bond energy than the reactants. Some examples of endothermic reactions are melting of ice to form water, sublimation of solid $C{O_2}$ , baking of bread, evaporation of liquid water to form water vapor etc.
Hence, option B is correct.
Note: For exothermic reactions, the potential energy of the product is generally lower than that of the reactants. On the other hand, the potential energy of the product in an endothermic reaction is higher than that of the reactants.
Complete step by step answer:
An endothermic reaction is a chemical reaction in which more energy is needed to break bonds in the reactants than the energy released when new bonds are formed in the form of products. These are basically the reactions in which the reactants absorb heat energy from the surroundings to form products. This reaction occurs only when the heat is added.
Moreover, these reactions lower the temperature of their surrounding area, thereby creating a cooling effect.

Now, in the given picture, the energy tied up in the molecular bonds is greater in the products than in the reactants. However, there is a need for the activation energy to commence the reaction but, once that hurdle is overcome, the reaction proceeds resulting in the formation of product C and the resulting product has more bond energy than the reactants. Some examples of endothermic reactions are melting of ice to form water, sublimation of solid $C{O_2}$ , baking of bread, evaporation of liquid water to form water vapor etc.
Hence, option B is correct.
Note: For exothermic reactions, the potential energy of the product is generally lower than that of the reactants. On the other hand, the potential energy of the product in an endothermic reaction is higher than that of the reactants.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




