(A) In general phenolphthalein is used as an indicator for the titration of a weak acid (\[C{{H}_{3}}COOH\]) and strong base (\[NaOH\]).
(R) At the equivalence point solution is basic.
(A)- Both (R) and (A) are true and the reason is the correct explanation of assertion.
(B)- Both (R) and (A) are true but the reason is not the correct explanation of assertion.
(C) – Assertion (A) is true but reason (R) is false
(D)- Assertion (A) is false but reason (R) is true
Answer
260.7k+ views
Hint: At the equivalence point, the solution turns from colourless to pale pink colour.
The equivalence point is that point of the acid-base titration when the amount of the titrant added is sufficient to neutralize the analyte solution.
Phenolphthalein is a commonly used acid-base indicator and it has no colour in acidic conditions.
Complete step by step solution:
- In general, phenolphthalein is used as an indicator for the titration of a weak acid (\[C{{H}_{3}}COOH\]) and strong base (\[NaOH\]). This is because phenolphthalein changes its colour from colourless to pale pink at around a \[pH\]of 8 to 10 and the equivalence point for the titration of the weak acid and strong base lies around \[pH\]7 and upwards.
- Other indicators like methyl orange change its colour at around a \[pH\]of 4. So, there is a considerable difference between the \[pH\]of the colour change of the indicator and the equivalence point for the titration of the weak acid and strong base.
- So, phenolphthalein is the best indicator in this case.
The graph showing the equivalence point for the titration of the weak acid and strong base is as follows:
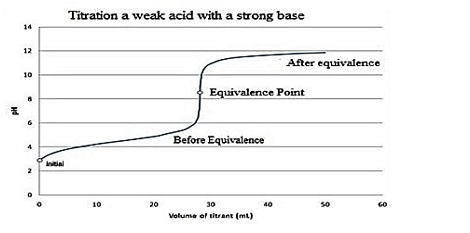
So, option B is the correct option.
Note: The reason behind the equivalence point for the titration of the weak acid and strong base lies around \[pH\]7 and upwards is due to the conjugate base of the weak acid. So, there will be a greater concentration of \[O{{H}^{-}}\]ions in the solution.
The equivalence point is that point of the acid-base titration when the amount of the titrant added is sufficient to neutralize the analyte solution.
Phenolphthalein is a commonly used acid-base indicator and it has no colour in acidic conditions.
Complete step by step solution:
- In general, phenolphthalein is used as an indicator for the titration of a weak acid (\[C{{H}_{3}}COOH\]) and strong base (\[NaOH\]). This is because phenolphthalein changes its colour from colourless to pale pink at around a \[pH\]of 8 to 10 and the equivalence point for the titration of the weak acid and strong base lies around \[pH\]7 and upwards.
- Other indicators like methyl orange change its colour at around a \[pH\]of 4. So, there is a considerable difference between the \[pH\]of the colour change of the indicator and the equivalence point for the titration of the weak acid and strong base.
- So, phenolphthalein is the best indicator in this case.
The graph showing the equivalence point for the titration of the weak acid and strong base is as follows:

So, option B is the correct option.
Note: The reason behind the equivalence point for the titration of the weak acid and strong base lies around \[pH\]7 and upwards is due to the conjugate base of the weak acid. So, there will be a greater concentration of \[O{{H}^{-}}\]ions in the solution.
Recently Updated Pages
JEE Main 2024 (January 25 Shift 1) Chemistry Question Paper with Solutions [PDF]

Differential Calculus: Concepts, Rules & Applications Explained

Torque and Rotational Motion Explained: Physics Made Simple

Second Law of Thermodynamics Explained: Simple Guide for Students

JEE Main 2024 (January 24 Shift 2) Maths Question Paper with Solutions [PDF]

Matrices Explained: Definition, Types & Examples

Trending doubts
Understanding the Different Types of Solutions in Chemistry

JEE Main Marks vs Percentile 2026: Predict Your Score Easily

JEE Main Cutoff 2026: Category-wise Qualifying Percentile

JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main 2026 Session 2 Result Out Live Scorecard Link Active Cutoff Released

JEE Main Marks vs Rank 2026: Expected Rank for 300 to 0 Marks

Other Pages
Valentine Week 2026 Complete List, Dates, Quotes, Wishes & Celebration Guide

Ambedkar Jayanti 2026 – Date, History, Significance, Wishes, Quotes & Celebration

Navratri 2026: Day-to-Day Navratri Colours Guide with Correct Dates, Devi Names, and Significance

Labour Day 2026: Theme, History, Significance and Quotes

UP Board 10th Result 2026 OUT: Check Result, Pass Percentage, Toppers and Marksheet Download Steps

Odisha 10th Result 2026 (LIVE Soon): BSE HSC Result Link, Date & Updates




