1, 3-butadiene reacts with ethylene to form
A. Benzene
B. Cyclohexane
C. Cyclohexene
D. 2, 3 dimethyl butane
Answer
267.6k+ views
Hint: Hydrocarbons containing two double bonds are called dienes. These are also called alkadienes. The reaction of 1,3-butadiene with ethylene involves the formation of a six-membered ring by the 1,4-addition of an alkene to a conjugated diene.
Complete Step by Step Answer:
Hydrocarbons containing two double bonds are called dienes. These are also called alkadienes.
These compounds are isomeric with alkynes.
1,3-butadiene is a conjugated diene or a single bond that intervenes with two double bonds. They have an arrangement of alternate single and double bonds.
The reaction of 1,3-butadiene with ethylene involves the formation of a six-membered ring by the 1,4-addition of an alkene to a conjugated diene. This reaction is known as the Diels-Alder reaction.
Alkene used is known as a dienophile and the product is known as Diels-Alder adduct. It is a cycloaddition reaction. Diels-Alder reaction is a 1,4 addition of ethene to 1,3-butadiene and also a 1,2 addition of butadiene to ethene. It is known as [4 + 2] cycloaddition leading to the formation of a six-membered ring. This six-membered ring is cyclohexene.
The reaction happens as follows:
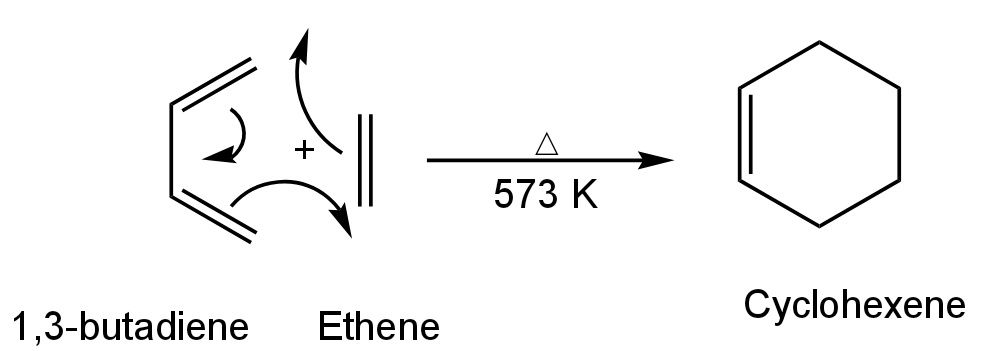
Image: Diels-Alder reaction
So, 1, 3-butadiene reacts with ethylene to form cyclohexane.
So, option C is correct.
Note: Ethene and other simple alkenes normally are weak dienophiles and react with 1,3-butadiene only under drastic conditions and in low yield. But, when the hydrogen atoms in the diene are replaced with various electron-attracting groups such as chlorine or bromine, electron-donating groups the reaction happens on its own.
Complete Step by Step Answer:
Hydrocarbons containing two double bonds are called dienes. These are also called alkadienes.
These compounds are isomeric with alkynes.
1,3-butadiene is a conjugated diene or a single bond that intervenes with two double bonds. They have an arrangement of alternate single and double bonds.
The reaction of 1,3-butadiene with ethylene involves the formation of a six-membered ring by the 1,4-addition of an alkene to a conjugated diene. This reaction is known as the Diels-Alder reaction.
Alkene used is known as a dienophile and the product is known as Diels-Alder adduct. It is a cycloaddition reaction. Diels-Alder reaction is a 1,4 addition of ethene to 1,3-butadiene and also a 1,2 addition of butadiene to ethene. It is known as [4 + 2] cycloaddition leading to the formation of a six-membered ring. This six-membered ring is cyclohexene.
The reaction happens as follows:

Image: Diels-Alder reaction
So, 1, 3-butadiene reacts with ethylene to form cyclohexane.
So, option C is correct.
Note: Ethene and other simple alkenes normally are weak dienophiles and react with 1,3-butadiene only under drastic conditions and in low yield. But, when the hydrogen atoms in the diene are replaced with various electron-attracting groups such as chlorine or bromine, electron-donating groups the reaction happens on its own.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




