



Properties of Solids and Liquids: Complete Class 11 Physics Guide for NEET
Properties of Solids and Liquids is an important chapter in Class 11 Physics and a high-weightage topic for NEET. This chapter explains how solids behave when forces are applied, how liquids respond to temperature changes, and how heat is transferred from one body to another. It includes important concepts such as elasticity, stress, strain, Hooke’s law, modulus of elasticity, heat, calorimetry, latent heat, thermal expansion, anomalous expansion of water, thermometry, conduction, convection, radiation, and Newton’s law of cooling.
What are Solids and Liquids?
A solid has a definite shape and definite volume because its particles are closely packed and strongly bound by intermolecular forces.
A liquid has a definite volume but no fixed shape, so it takes the shape of its container.
Solids are mainly studied for their elastic and mechanical properties, while liquids are especially important in topics like heat, expansion, buoyancy, and thermal behaviour.
Nature of Solids and Liquids
In solids, particles remain fixed at their mean positions and only vibrate about them.
Strong intermolecular forces in solids make them resist changes in shape and volume.
In liquids, particles are less tightly bound and can move or slide past each other.
This ability allows liquids to flow while still maintaining a definite volume.
The behaviour of solids and liquids depends on intermolecular forces, temperature, and external forces.
Hence, the study of solids and liquids includes both mechanical properties and thermal properties of matter.
.
Intermolecular Force and Potential Energy
The molecules of matter exert forces on one another. When two molecules are far apart, the force between them is almost zero. As they come closer, attractive forces act between them. If they come too close, repulsive forces become dominant.
The relation between force and potential energy is given by
$F=-\dfrac{dU}{dr}$,
where,
$U$ is the intermolecular potential energy
$r$ is the separation between the particles
At the equilibrium separation $r=r_0$, the force becomes zero, so $F=0$. At this point, the potential energy is at a minimum, which means the system is most stable. If the separation becomes less than $r_0$, repulsion increases sharply. If the separation becomes greater than $r_0$, attraction dominates. This explains why solids are rigid and why liquids can flow more easily.
Elasticity
Elasticity is the property of a material by virtue of which it regains its original shape and size after the deforming force is removed, provided the elastic limit is not crossed.
When an external force changes the shape, size, or volume of a body, the body tries to oppose this change. This happens because of intermolecular restoring forces present inside the material.
Important Terms Related to Elasticity
A deforming force is an external force that changes the shape or size of a body.
A restoring force is the internal force produced inside the body that tries to bring it back to its original state.
The cause of elasticity is the intermolecular force between the particles of the material. When the particles are displaced from their equilibrium positions, these forces act to restore the original arrangement.
Stress
Stress is the internal restoring force acting per unit area of cross-section of a body.
Its formula is $Stress=\dfrac{F}{A}$.
Here, $F$ is the restoring force and $A$ is the area over which the force acts.
The SI unit of stress is pascal, where $1 , Pa = 1 , N/m^2$.
The dimensional formula of stress is $[ML^{-1}T^{-2}]$.

Types of Stress
1. Longitudinal Stress
When the deforming force acts perpendicular to the area of cross-section and changes the length of a body, the stress produced is called longitudinal stress.
Its formula is $Longitudinal , Stress=$\dfrac{F}{A}$.
Longitudinal stress is of two types:
Tensile stress is produced when the body is stretched.
Compressive stress is produced when the body is compressed.
2. Shear or Tangential Stress
When the deforming force acts tangentially or parallel to the surface of a body, the resulting stress is called shear stress.
Its formula is $Shear , Stress=$\dfrac{F}{A}$.
This type of stress changes the shape of the body without necessarily changing its volume.
3. Hydraulic or Volume Stress
When pressure acts equally in all directions on a body, as in liquids or submerged objects, the stress produced is called hydraulic stress.
Its formula is $Hydraulic , Stress = Pressure = \dfrac{F}{A}$.
Strain
Strain is the ratio of change in configuration to the original configuration of a body.
In general, $Strain=\dfrac{Change , in , dimension}{Original , dimension}$.
Strain has no unit because it is a ratio. Hence, it is dimensionless.
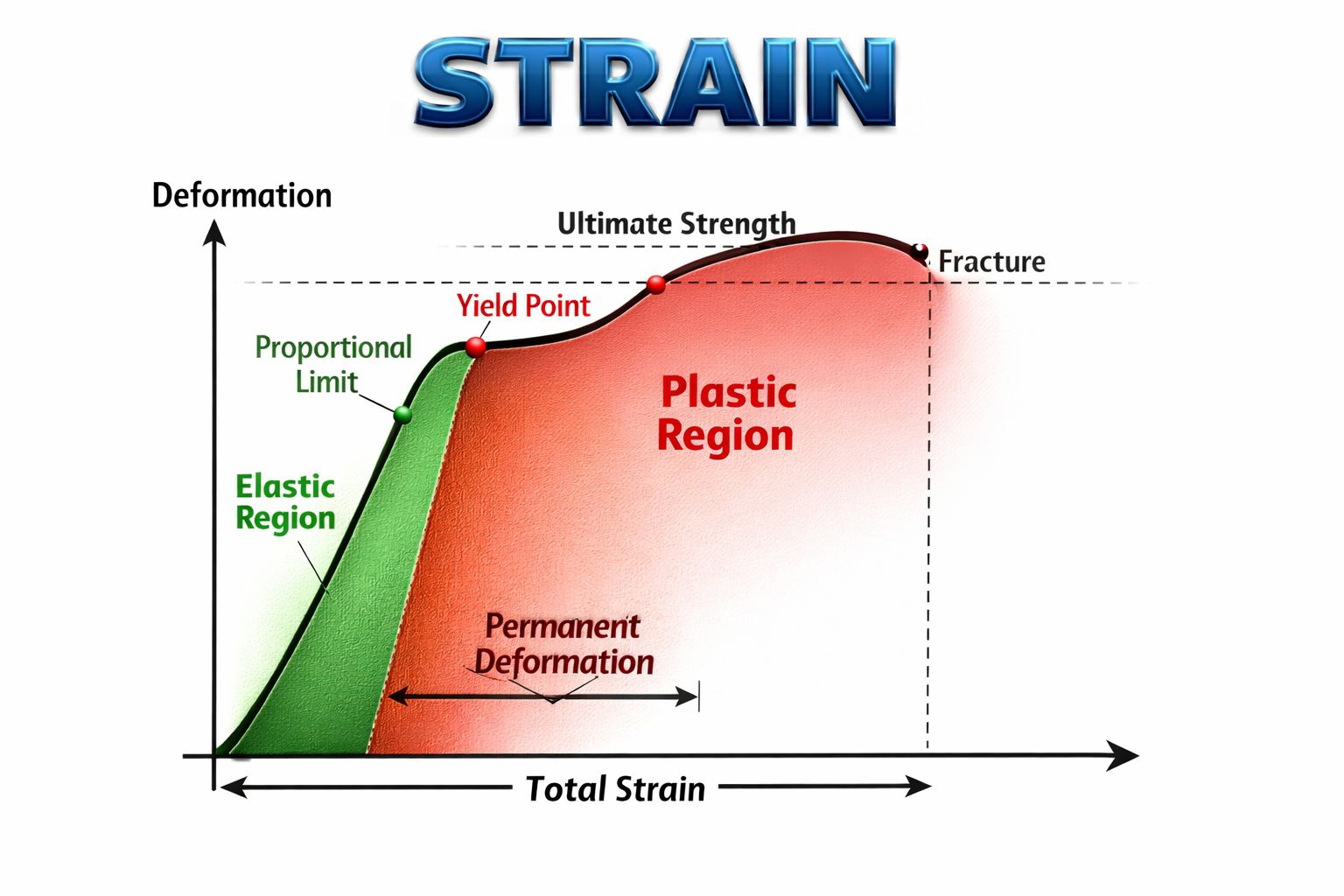
Types of Strain
1. Longitudinal Strain
If a rod of original length $L$ undergoes a change in length $\Delta L$, then longitudinal strain is given by Longitudinal, $Strain=\dfrac{\Delta L}{L}$.
It is produced when the body is stretched or compressed along its length.
2. Volume or Hydraulic Strain
If a body of original volume $V$ undergoes a change in volume $\Delta V$, then volume strain is given by Volume, $Strain=\dfrac{\Delta V}{V}$.
It is produced when the volume of the body changes because of pressure.
3. Shear Strain
Shear strain is produced when the tangential force changes the shape of the body.
If the upper surface of a block is displaced by a small distance $x$ and the height of the block is $l$, then shear strain is Shear , Strain=$\dfrac{x}{l}$.
If the angular deformation is $\phi$, then for small deformations, Shear, Strain=$\tan \phi \approx \phi$.
Hooke’s Law
Hooke’s law states that within the elastic limit, stress is directly proportional to strain.
Mathematically, $Stress \propto Strain$.
So, $Stress = Constant \times Strain$.
This constant depends on the nature of the material and is called the modulus of elasticity.

Hooke’s law is valid only up to the elastic limit. Beyond this limit, the body may not regain its original dimensions fully.
Modulus of Elasticity
The ratio of stress to corresponding strain is called the modulus of elasticity.
In general, $Modulus , of , Elasticity=\dfrac{Stress}{Strain}$.
Its SI unit is pascal or $N/m^2$.
A greater value of modulus means the material is stiffer and harder to deform.
1. Young’s Modulus
Young’s modulus is defined as the ratio of longitudinal stress to longitudinal strain.
So, $Y=\dfrac{Longitudinal , Stress}{Longitudinal , Strain}$.
Substituting the expressions, $Y=\dfrac{F/A}{\Delta L/L}$.
Therefore, $Y=\dfrac{FL}{A\Delta L}$.
Young’s modulus is used when the length of a wire, rod, or bar changes under tension or compression.
2. Bulk Modulus
Bulk modulus is defined as the ratio of hydraulic stress to volume strain.
So, $B=\dfrac{Hydraulic , Stress}{Volume , Strain}$.
Since volume decreases when pressure increases, bulk modulus is written as $B=-\dfrac{\Delta P}{\Delta V/V}$.
It can also be written as $B=-V\dfrac{\Delta P}{\Delta V}$.
The negative sign shows that pressure increase causes a decrease in volume.
The reciprocal of bulk modulus is called compressibility, so $Compressibility=\dfrac{1}{B}$.
3. Modulus of Rigidity or Shear Modulus
Modulus of rigidity is the ratio of shear stress to shear strain.
So, $\eta=\dfrac{Shear , Stress}{Shear , Strain}$.
Thus, $\eta=\dfrac{F/A}{x/l}$.
Hence, $\eta=\dfrac{Fl}{Ax}$.
If angular deformation is used, then $\eta=\dfrac{F/A}{\phi}$ for small values of $\phi$.
Stress-Strain Curve
The stress-strain curve helps us understand the mechanical behaviour of a material under load.
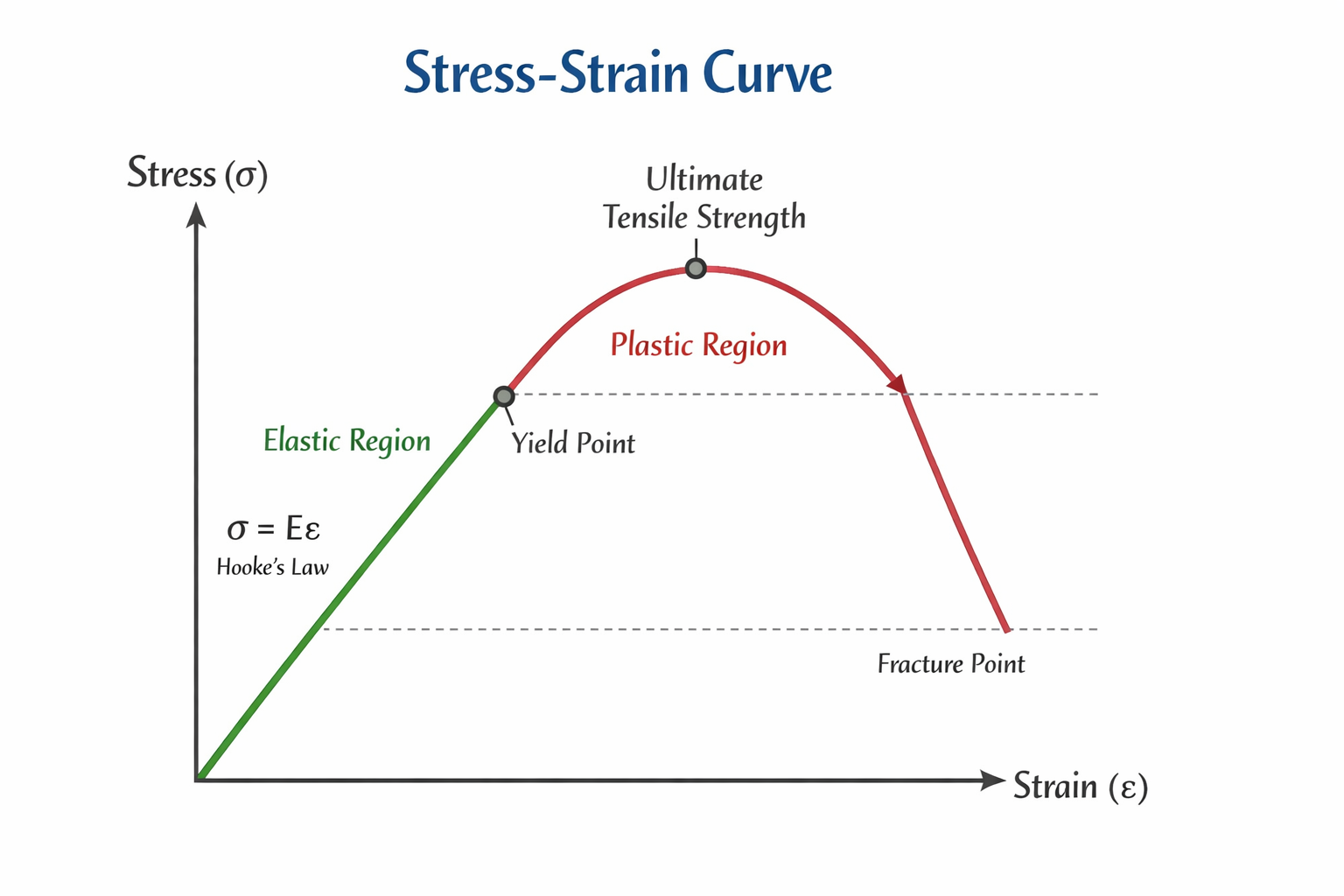
In the beginning, stress is directly proportional to strain. This part is a straight line and follows Hooke’s law.
Important Points on the Stress-Strain Curve
Limit of proportionality is the point up to which stress is directly proportional to strain.
Elastic limit is the maximum stress up to which the material returns to its original shape after removal of force.
Yield point is the point beyond which permanent deformation begins.
Plastic region is the region where strain increases without proportional increase in stress.
Ultimate strength is the maximum stress the material can bear.
Fracture point is the point where the material breaks.
Ductile and Brittle Materials
A ductile material can undergo large plastic deformation before breaking. Examples include copper and mild steel.
A brittle material breaks almost immediately after crossing the elastic limit. Examples include glass and cast iron.
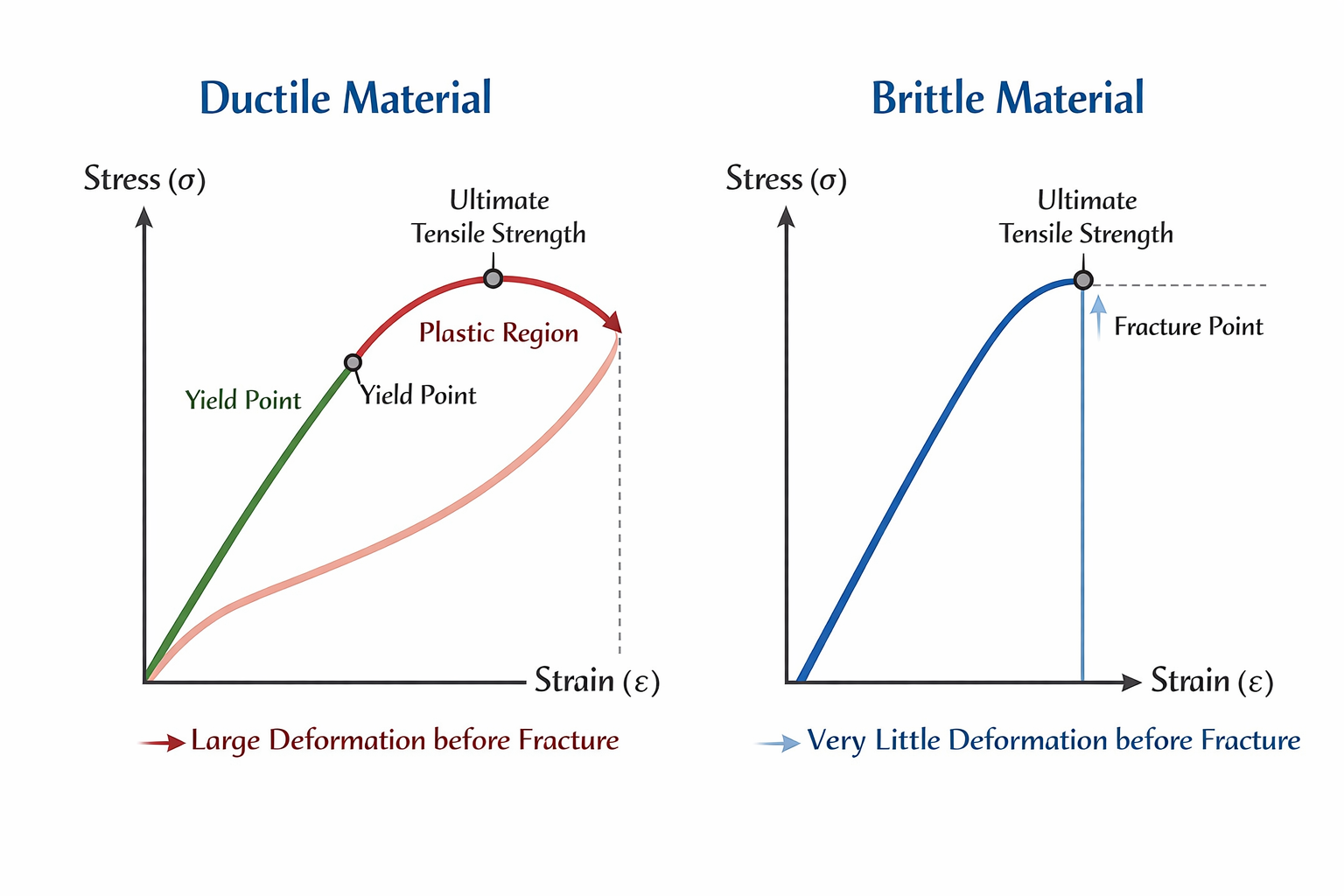
Elastic Fatigue
Elastic fatigue is the weakening of the elastic property of a material due to repeated stress or repeated loading and unloading.
A body that undergoes repeated deformation may lose its ability to recover fully. This is important in bridges, machine parts, springs, and metal wires.
Extension of a Wire Due to Applied Force
Suppose a wire of length $L$ and cross-sectional area $A$ is stretched by a force $F$. If the extension produced is $\Delta L$, then from Young’s modulus, $Y=\dfrac{FL}{A\Delta L}$.
Hence, $\Delta L=\dfrac{FL}{AY}$.
This formula shows that extension increases with force and original length, but decreases with area and Young’s modulus.
Increment in Length Due to Own Weight
If a vertical wire has density $\rho$, length $L$, and Young’s modulus $Y$, then the extension due to its own weight is given by $\Delta L=\dfrac{\rho g L^2}{2Y}$.
This is because the tension is not the same throughout the wire. It increases from bottom to top.
Analogy Between a Rod and a Spring
A stretched rod behaves like a spring within the elastic limit. For a spring, Hooke’s law is written as $F=kx$, where $k$ is the spring constant.
For a rod, extension is given by $\Delta L=\dfrac{FL}{AY}$. Rearranging, $F=\dfrac{AY}{L}\Delta L$.
Comparing this with $F=kx$, the effective spring constant of a rod is $k=\dfrac{AY}{L}$.
This is why a rod can be treated as an equivalent spring in elastic problems.
Springs in Series and Parallel
Springs in Series
If two springs of spring constants $k_1$ and $k_2$ are connected in series, then the effective spring constant is given by $\dfrac{1}{k_{eq}}=\dfrac{1}{k_1}+\dfrac{1}{k_2}$.
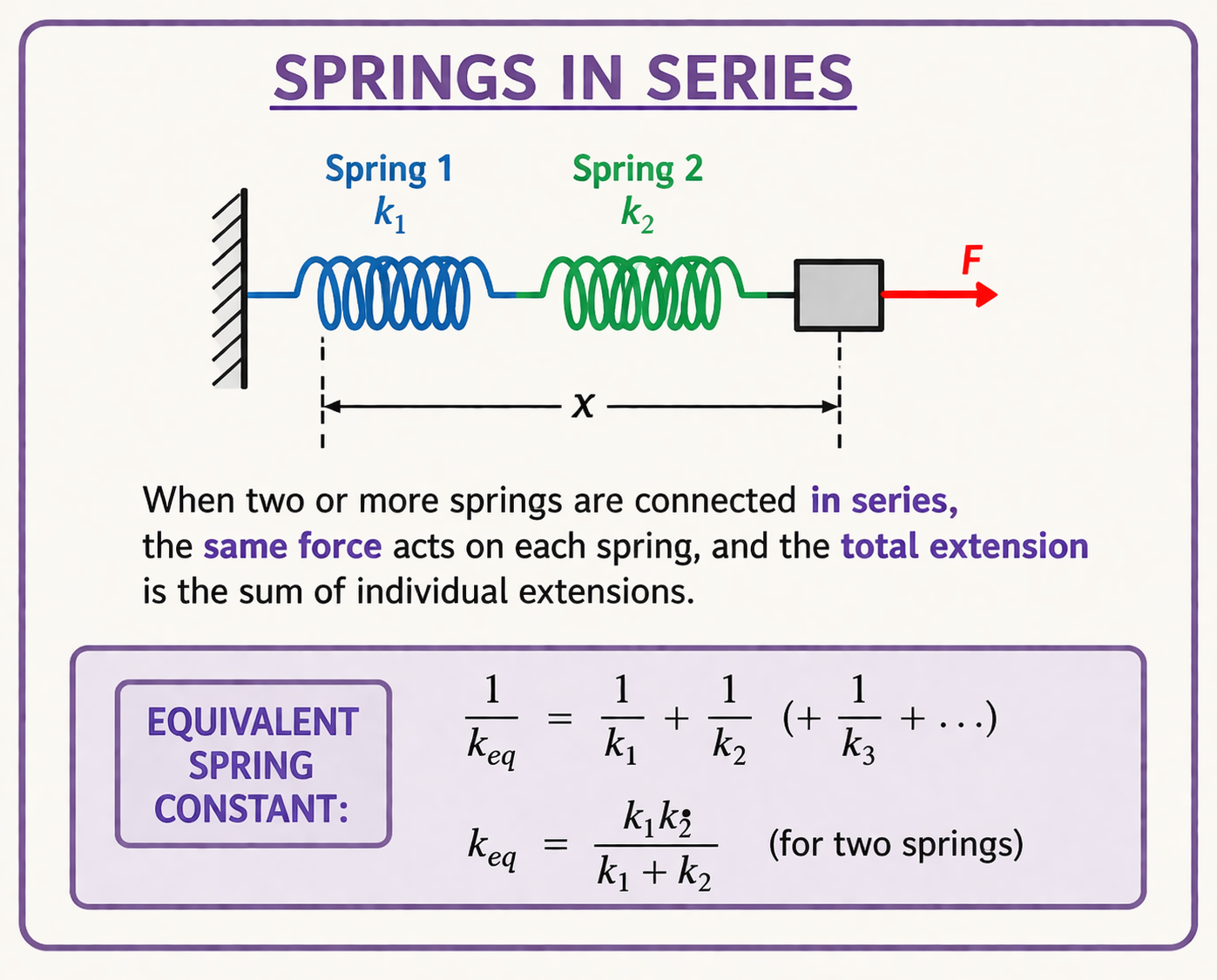
For three springs in series, $\dfrac{1}{k_{eq}}=\dfrac{1}{k_1}+\dfrac{1}{k_2}+\dfrac{1}{k_3}$.
Springs in Parallel
If two springs are connected in parallel, then their effective spring constant is $k_{eq}=k_1+k_2$.

For more springs, the total becomes the sum of all spring constants.
Elastic Potential Energy Stored in a Wire or Rod
When a wire or rod is stretched, work is done against the restoring force. This work is stored as elastic potential energy.
For extension $x$, the force increases from $0$ to $F$, so average force is $\dfrac{F}{2}$.
Therefore, elastic potential energy is $U=\dfrac{1}{2}Fx$.
Using $F=\dfrac{AY}{L}x$, we get $U=\dfrac{1}{2}\dfrac{AY}{L}x^2$.
Since strain is $\dfrac{x}{L}$ and stress is $\dfrac{F}{A}$, strain energy per unit volume is $u=\dfrac{1}{2}\times Stress \times Strain$.
So, $u=\dfrac{1}{2}\sigma \epsilon$.
Heat
Heat is the form of energy that is transferred from one body to another because of temperature difference.
Heat always flows from a body at higher temperature to a body at lower temperature until thermal equilibrium is reached.
The SI unit of heat is joule.
Another common unit is calorie. One calorie is the amount of heat required to raise the temperature of $1 , g$ of water by $1^\circ C$.
The relation between calorie and joule is $1 , cal = 4.186 , J$.
Temperature
Temperature is the physical quantity that tells us how hot or cold a body is.
It determines the direction of heat flow. Heat flows from higher temperature to lower temperature.
Thermometry is the branch of physics that deals with temperature measurement.
Mechanical Equivalent of Heat
Mechanical equivalent of heat expresses the relationship between heat and work.
If $J$ is the mechanical equivalent of heat, then $W=JH$, where $W$ is mechanical work and $H$ is heat in calories.
Its value is approximately $J=4.186 , J/cal$.
This shows that heat is also a form of energy.
Heat Capacity and Specific Heat Capacity
Heat capacity is the amount of heat required to raise the temperature of a body by $1^\circ C$ or $1 , K$.
If $Q$ heat changes the temperature by $\Delta T$, then heat capacity is $C=\dfrac{Q}{\Delta T}$.
Its SI unit is $J/K$.
Specific heat capacity is the amount of heat required to raise the temperature of unit mass of a substance by $1^\circ C$ or $1 , K$.
Its formula is $s=\dfrac{Q}{m\Delta T}$.
Therefore, $Q=ms\Delta T$.
Its SI unit is $J , kg^{-1} K^{-1}$.
The relation between heat capacity and specific heat is $C=ms$.
Water Equivalent
The water equivalent of a body is the mass of water that has the same heat capacity as the body.
If $m$ is the mass of the body and $s$ is its specific heat, then water equivalent $W=ms$ in calorie units when specific heat of water is taken as $1$.
In general, if the specific heat of water is $s_w$, then $Water , Equivalent=\dfrac{ms}{s_w}$.
Water equivalent is useful in calorimetry.
Law of Mixture
When two substances at different temperatures are mixed in an insulated container, the heat lost by the hotter body equals the heat gained by the colder body.
So, $Heat , lost = Heat , gained$.
If two bodies of masses $m_1$ and $m_2$, specific heats $s_1$ and $s_2$, and temperatures $T_1$ and $T_2$ are mixed and final temperature becomes $T_m$, then $m_1 s_1 (T_1-T_m)=m_2 s_2 (T_m-T_2)$.
If a calorimeter is also involved, then the heat gained by the calorimeter must also be included.
Calorimeter
A calorimeter is an insulated vessel used to measure heat exchange.
It is designed so that heat does not escape to the surroundings significantly. It is used to find specific heat, latent heat, final temperature of mixtures, and heat capacity.
If a calorimeter has heat capacity $C$, then heat absorbed by calorimeter is $Q=C\Delta T$.
If its water equivalent is $W$, then heat absorbed by calorimeter is $Q=Ws_w\Delta T$.
Phase Change and Latent Heat
When a substance changes from one state to another, it absorbs or releases heat without change in temperature. This heat is called latent heat.
The general formula is $Q=mL$, where $m$ is the mass and $L$ is the latent heat.
Latent Heat of Fusion
Latent heat of fusion is the heat required to convert unit mass of a solid into liquid at its melting point without any temperature change.
So, $Q=mL_f$.
Latent Heat of Vaporisation
Latent heat of vaporisation is the heat required to convert unit mass of a liquid into vapour at its boiling point without any temperature change.
So, $Q=mL_v$.

For water, latent heat of vaporisation is very high. This is why boiling requires a large amount of heat.
Heating Curve
When heat is supplied to a substance at a constant rate, the graph between temperature and time is called the heating curve.
During heating of a solid, liquid, or gas, temperature rises with time.
During melting and boiling, temperature remains constant even though heat is being supplied. This is because the supplied heat is used in changing the state, not in increasing temperature.
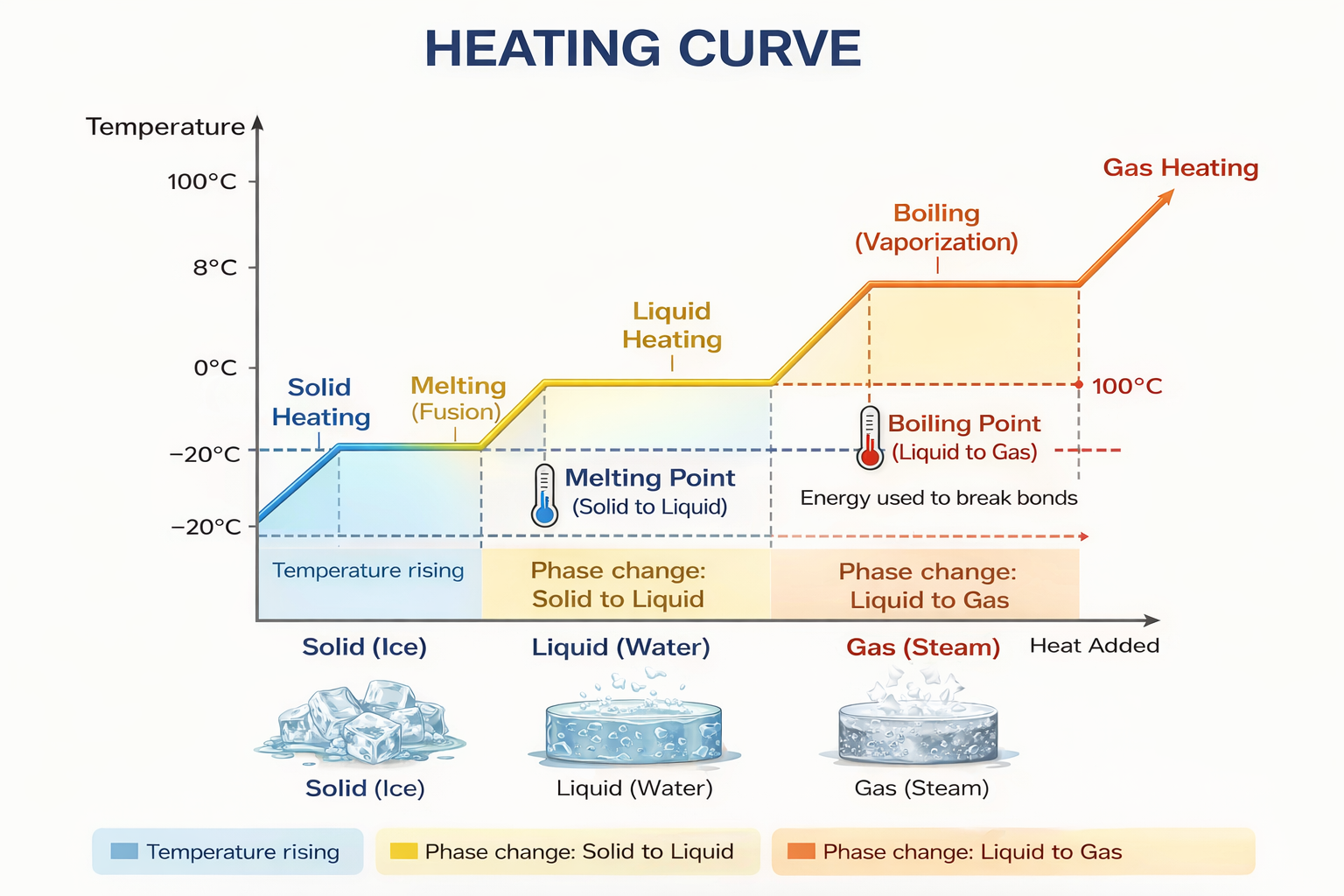
For solid heating, $Q=ms\Delta T$.
For melting, $Q=mL_f$.
For liquid heating, $Q=ms\Delta T$.
For boiling, $Q=mL_v$.
For gas heating, $Q=ms\Delta T$.
Thermal Expansion of Solids
When a body is heated, its particles vibrate with larger amplitude and mean separation between particles increases. As a result, the dimensions of the body increase. This is called thermal expansion.
1. Linear Expansion
If the original length of a rod is $L_0$ and the increase in temperature is $\Delta T$, then the final length becomes $L=L_0(1+\alpha \Delta T)$.
The increase in length is $\Delta L=\alpha L_0 \Delta T$.
Here, $\alpha$ is the coefficient of linear expansion.
Its unit is $K^{-1}$ or ${}^\circ C^{-1}$.
2. Superficial or Areal Expansion
If the original area is $A_0$, then after heating, the new area becomes $A=A_0(1+\beta \Delta T)$.
Hence, the change in area is $\Delta A=\beta A_0 \Delta T$.
Here, $\beta$ is the coefficient of area expansion.
3. Volume Expansion
If the original volume is $V_0$, then after heating, the new volume becomes $V=V_0(1+\gamma \Delta T)$.
So, the change in volume is $\Delta V=\gamma V_0 \Delta T$.
Here, $\gamma$ is the coefficient of volume expansion.
Relation Between Expansion Coefficients
For isotropic solids, the relation is $\beta=2\alpha$ and $\gamma=3\alpha$.
Therefore, $\alpha:\beta:\gamma = 1:2:3$.
Thermal Stress
If a rod is heated or cooled but is not allowed to expand or contract freely, then internal stress develops in it. This is called thermal stress.
The thermal strain is $Strain=\alpha \Delta T$.
Therefore, thermal stress is $Stress=Y\alpha \Delta T$.
This concept is important in rails, bridges, and metal structures.
Measurement of Length by Metallic Scale
When a metallic scale expands due to temperature rise, the spacing between its markings increases. Therefore, the apparent measurement changes.
If the scale has coefficient of linear expansion $\alpha$ and temperature change $\Delta T$, then the actual length corresponding to a reading $l$ becomes approximately $L=l(1+\alpha \Delta T)$.
This is important in precision measurements.
Bimetallic Strip
A bimetallic strip consists of two different metals joined together. Since the two metals have different coefficients of linear expansion, one expands more than the other on heating. Hence, the strip bends.
This principle is used in thermostats, automatic switches, electric irons, and temperature-control devices.
Thermal Expansion in Liquids
Liquids do not have fixed shape, so only volume expansion is important in liquids.
If a liquid is heated in a vessel, both the liquid and the vessel expand. Therefore, two types of expansion are studied.
Real Expansion
Real expansion is the actual increase in volume of the liquid.
Apparent Expansion
Apparent expansion is the increase in volume observed in the vessel.
If $\gamma_l$ is the coefficient of real expansion of the liquid and $\gamma_v$ is the coefficient of cubical expansion of the vessel, then coefficient of apparent expansion is $\gamma_a=\gamma_l-\gamma_v$.
Thus, real expansion = apparent expansion + expansion of vessel.
Anomalous Expansion of Water
Most liquids expand on heating and contract on cooling. Water behaves differently between $0^\circ C$ and $4^\circ C$.
When water is heated from $0^\circ C$ to $4^\circ C$, its volume decreases and density increases. At $4^\circ C$, the density of water becomes maximum.
From $4^\circ C$ onward, water expands normally with increase in temperature.
This unusual behaviour is called anomalous expansion of water.
Importance of Anomalous Expansion of Water
In winter, the surface water of lakes cools first. As it cools from higher temperatures to $4^\circ C$, it becomes denser and sinks. This continues until the lower part of the lake reaches about $4^\circ C$.
When the surface water cools below $4^\circ C$, it becomes less dense, so it remains at the top and eventually freezes at $0^\circ C$.
The ice formed on the top acts as an insulating layer because ice is a poor conductor of heat. Therefore, the water below remains liquid and aquatic life survives.
Variation of Density with Temperature
Density is given by $\rho=\dfrac{m}{V}$.
If volume changes with temperature, then density also changes.
For most substances, as temperature increases, volume increases and density decreases.
For water between $0^\circ C$ and $4^\circ C$, density increases with temperature because volume decreases in that range.
Variation of Force of Buoyancy with Temperature
Force of buoyancy is $F_b=\rho V g$.
Since density depends on temperature, buoyant force also changes with temperature.
If the density of the liquid decreases with increase in temperature, then buoyant force decreases.
This is important in fluid mechanics and thermal effects in liquids.
Measurement of Temperature and Temperature Scales
Thermometers use a measurable property that changes with temperature. Such a property is called a thermometric property.
Common thermometric properties include volume of a liquid, pressure of a gas, and electrical resistance.

Celsius Scale
In Celsius scale, the lower fixed point is $0^\circ C$ and the upper fixed point is $100^\circ C$.
Fahrenheit Scale
In Fahrenheit scale, the lower fixed point is $32^\circ F$ and the upper fixed point is $212^\circ F$.
Kelvin Scale
Kelvin is the SI scale of temperature. It starts from absolute zero.
The relation between Celsius and Kelvin is $K = {}^\circ C + 273.15$.
The relation between Celsius and Fahrenheit is $\dfrac{C}{5}=\dfrac{F-32}{9}$.
The relation between Fahrenheit and Kelvin can be written as $\dfrac{K-273.15}{5}=\dfrac{F-32}{9}$.
Modes of Heat Transfer
Heat is transferred in three ways: conduction, convection, and radiation.
Conduction
Conduction is the transfer of heat from one part of a body to another without actual movement of the particles of the medium.
It mainly occurs in solids. In metals, conduction takes place due to free electrons as well as lattice vibrations.
The rate of heat flow through a slab is given by $Q=\dfrac{kA(T_1-T_2)t}{L}$.
Therefore, heat current is $\dfrac{Q}{t}=\dfrac{kA(T_1-T_2)}{L}$.
Here, $k$ is the thermal conductivity, $A$ is the area, $L$ is the length or thickness, and $(T_1-T_2)$ is the temperature difference.
The SI unit of thermal conductivity is $W , m^{-1} K^{-1}$.
The dimensional formula is $[MLT^{-3}K^{-1}]$.
Temperature Gradient
Temperature gradient is the rate of change of temperature with distance.
It is written as $\dfrac{dT}{dx}$.
Using differential form, heat current is $\dfrac{dQ}{dt}=-kA\dfrac{dT}{dx}$.
The negative sign shows that heat flows from higher temperature to lower temperature.
Steady State
A body is said to be in steady state when temperature at every point remains constant with time, though heat continues to flow through it.
In steady state, the amount of heat entering a section in a given time is equal to the amount of heat leaving it in the same time.
Thermal Resistance
Thermal resistance in conduction is defined as $R=\dfrac{L}{kA}$.
Thus, heat current can be written as $\dfrac{Q}{t}=\dfrac{\Delta T}{R}$.
This form is similar to electric current in Ohm’s law.
Slabs in Series
If slabs are arranged in series, then total thermal resistance is $R_{eq}=R_1+R_2+R_3+\dots$.
Hence, $R_{eq}=\dfrac{L_1}{k_1A}+\dfrac{L_2}{k_2A}+\dots$.
The equivalent thermal conductivity can then be found from the combined expression.
Slabs in Parallel
If slabs are arranged in parallel, then the temperature difference across each slab is the same, and heat currents add.
The equivalent thermal conductivity is obtained by adding heat currents through all slabs.
Convection
Convection is the transfer of heat by actual movement of particles of a fluid.
It occurs in liquids and gases.
When a fluid is heated, it expands, its density decreases, and it rises. The cooler and denser fluid sinks. This produces convection currents.
Convection explains sea breeze, land breeze, room ventilation, and heating of liquids.
Radiation
Radiation is the transfer of heat in the form of electromagnetic waves. It does not require any material medium.
All bodies continuously emit and absorb radiation.
The amount of radiation incident on a body may be divided into three parts: reflected, absorbed, and transmitted.
If $r$, $a$, and $t$ are the coefficients of reflection, absorption, and transmission, then $r+a+t=1$.
For a perfect reflector, $r=1$, $a=0$, and $t=0$.
For a perfect absorber or black body, $a=1$, $r=0$, and $t=0$.
For a perfect transmitter, $t=1$, $a=0$, and $r=0$.
Emissive Power and Emissivity
Emissive power is the heat radiated per unit area per unit time by a body.
Emissivity is the ratio of emissive power of a body to the emissive power of a perfectly black body at the same temperature.
So, $e=\dfrac{E}{E_b}$.
For a perfect black body, $e=1$.
Prevost’s Theory of Heat Exchange
According to Prevost’s theory, all bodies continuously emit and absorb radiation at all temperatures.
If a body emits more energy than it absorbs, it cools.
If it absorbs more energy than it emits, it gets heated.
If the rates of absorption and emission are equal, the temperature remains constant.
Kirchhoff’s Law
Kirchhoff’s law states that at a given temperature, the ratio of emissive power to absorptive power is the same for all bodies and is equal to the emissive power of a perfectly black body.
In practical form, good absorbers are good emitters and bad absorbers are bad emitters.
For a body in thermal equilibrium, $e=a$.
Stefan-Boltzmann Law
According to Stefan-Boltzmann law, the radiant energy emitted per unit area per unit time by a perfectly black body is directly proportional to the fourth power of its absolute temperature.
So, $E=\sigma T^4$.
For a real body, $E=e\sigma T^4$.
Here, $\sigma$ is the Stefan-Boltzmann constant.
If a body of area $A$ is at temperature $T$ and surroundings are at temperature $T_0$, then net rate of radiation is $P=e\sigma A(T^4-T_0^4)$.

Newton’s Law of Cooling
Newton’s law of cooling states that for small temperature differences between a body and its surroundings, the rate of loss of heat is directly proportional to the temperature difference.
So, $\dfrac{dQ}{dt}\propto (T-T_s)$.
It can be written as $\dfrac{dT}{dt}=-k(T-T_s)$.
Here, $T$ is the temperature of the body and $T_s$ is the temperature of the surroundings.
This law is valid when the temperature difference is small and cooling takes place mainly by radiation.

Solar Constant
The solar constant is the amount of solar energy received per unit area per unit time on a surface held perpendicular to the Sun’s rays at the mean distance of Earth from the Sun, outside the atmosphere.
Its value is approximately $1340 , W/m^2$.
Wien’s Displacement Law
Wien’s displacement law states that the wavelength corresponding to maximum emission is inversely proportional to the absolute temperature.
So, $\lambda_m T = constant$.
This constant is Wien’s displacement constant.
This means as temperature increases, the wavelength of maximum emission shifts towards lower values.
Important Practical Applications
The concepts of this chapter are used in many real-life situations. Elasticity is used in the design of bridges, cranes, springs, and machine parts. Thermal expansion explains expansion joints in railway tracks and bridges. Calorimetry is used in determining heat capacities and mixing temperatures. Anomalous expansion of water explains survival of aquatic life in winter. Radiation laws are used in thermometers, furnaces, and astronomy.
Conclusion
The chapter Properties of Solids and Liquids connects the mechanical behaviour and thermal behaviour of matter in a very meaningful way. It explains how solids get deformed, how materials resist force, how heat changes temperature and state, why substances expand, and how energy moves by conduction, convection, and radiation. A strong understanding of this chapter builds the base for mechanics, thermodynamics, and modern applications in engineering and everyday life.
FAQs on Properties of Solids and Liquids for NEET (Class 11 Physics)
1. What is the main difference between solids and liquids?
Solids have a fixed shape and volume due to strong intermolecular forces, whereas liquids have a fixed volume but no definite shape and can flow because their particles can move past each other.
2. What is stress and what is its SI unit?
Stress is the internal restoring force per unit area of a material. Its SI unit is pascal (Pa), where $1 , Pa = 1 , N/m^2$.
3. What is strain and why is it dimensionless?
Strain is the ratio of change in dimension to the original dimension. It is dimensionless because it is a ratio of two similar physical quantities.
4. State Hooke’s Law.
Hooke’s Law states that within the elastic limit, stress is directly proportional to strain.
5. What is Young’s Modulus?
Young’s Modulus is the ratio of longitudinal stress to longitudinal strain and measures the stiffness of a material.
6. What is the difference between ductile and brittle materials?
Ductile materials can undergo large deformation before breaking (e.g., copper), while brittle materials break suddenly without significant deformation (e.g., glass).
7. What is elastic fatigue?
Elastic fatigue is the weakening of a material’s elasticity due to repeated loading and unloading cycles.
8. What is latent heat?
Latent heat is the heat absorbed or released during a change of state without any change in temperature.
9. What is the difference between latent heat of fusion and vaporisation?
Latent heat of fusion is the heat required to convert solid into liquid, while latent heat of vaporisation is the heat required to convert liquid into vapour.
10. What is the law of mixture?
The law of mixture states that heat lost by the hot body is equal to the heat gained by the cold body when mixed in an insulated system.
11. What is anomalous expansion of water?
Water contracts when heated from $0^\circ C$ to $4^\circ C$ and expands beyond this temperature. It has maximum density at $4^\circ C$.
12. What are the three modes of heat transfer?
The three modes are conduction, convection, and radiation.
13. What is thermal expansion?
Thermal expansion is the increase in dimensions of a substance when temperature increases due to increased molecular vibrations.
14. State Stefan-Boltzmann Law.
It states that the energy radiated per unit area per unit time by a black body is proportional to the fourth power of its absolute temperature.
15. What is Kirchhoff’s Law of radiation?
Kirchhoff’s Law states that for a body in thermal equilibrium, emissivity is equal to absorptivity.
16. What is Newton’s Law of Cooling?
It states that the rate of loss of heat of a body is directly proportional to the difference in temperature between the body and its surroundings (for small temperature differences).
17. What is the SI unit of thermal conductivity?
The SI unit of thermal conductivity is $W , m^{-1} K^{-1}$.
18. Why does ice float on water?
Ice floats because it has lower density than water due to the expansion of water during freezing.
19. What is the heating curve?
A heating curve is a graph showing the variation of temperature with time as heat is supplied, indicating phase changes.
20. Why is latent heat of vaporisation high for water?
Water has strong intermolecular hydrogen bonding, so a large amount of energy is required to break these bonds during vaporisation.























