Which of the following is the correct order of strength of H-bonding in the given compound?
A. ${\rm{HF}} < {\rm{N}}{{\rm{H}}_3}$
B. ${{\rm{H}}_2}{\rm{O}} > {{\rm{H}}_2}{{\rm{O}}_{\rm{2}}}$
C. ${{\rm{H}}_2}{{\rm{O}}_{\rm{2}}} > {{\rm{H}}_2}{\rm{O}}$
D. ${\rm{N}}{{\rm{H}}_3} > {{\rm{H}}_{\rm{2}}}{\rm{O}}$
Answer
259.8k+ views
Hint: Hydrogen bond is a chemical bond in which formation of a covalent link of hydrogen atoms with other electronegative atoms, such as, fluorine, nitrogen and oxygen atoms takes place in the same or another molecule.
Complete step by step answer:
We know that the extent of hydrogen bonding depends on electronegativity and the number of hydrogen atoms available for bonding. Among Fluorine, nitrogen and oxygen atoms, increasing order of electronegativities is ${\rm{N}} < {\rm{O}} < {\rm{F}}$.
> Between ammonia $\left( {{\rm{N}}{{\rm{H}}_3}} \right)$ and hydrogen fluoride $\left( {{\rm{HF}}} \right)$, HF has more H-bonding than ${\rm{N}}{{\rm{H}}_3}$ because in ammonia, only one lone pair present which cannot satisfy all hydrogen also fluorine is more electronegative than nitrogen. So, option A is incorrect.
> Between water $\left( {{{\rm{H}}_2}{\rm{O}}} \right)$and hydrogen peroxide $\left( {{{\rm{H}}_2}{{\rm{O}}_{\rm{2}}}} \right)$, hydrogen peroxide has more H-bonding because of presence of one more oxygen that water. So, option B is incorrect and option C is correct.
> Between ammonia $\left( {{\rm{N}}{{\rm{H}}_3}} \right)$ and water $\left( {{{\rm{H}}_2}{\rm{O}}} \right)$, water has more H-bonding than ammonia $\left( {{\rm{N}}{{\rm{H}}_3}} \right)$ because of more number of hydrogen bond and also electronegativity of Oxygen is more than Nitrogen .
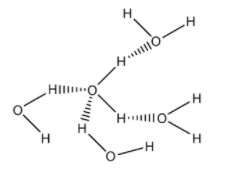
In case of water, it can form four hydrogen bonds with four other water molecules. But in case of ammonia, only one lone pair is present, which can form only one hydrogen bond with one ammonia molecule.
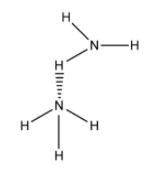
So, water molecules have more extent of hydrogen bonding. So, option D is incorrect.
Note: The extent of hydrogen bonding is in the order ${\rm{H}} - {\rm{F}} > {\rm{H}} - {\rm{O}} > {\rm{H}} - {\rm{N}}$, that means, extent of hydrogen bonding is more in case of HF than water $\left( {{{\rm{H}}_2}{\rm{O}}} \right)$, that is, ${\rm{HF}} > {{\rm{H}}_{\rm{2}}}{\rm{O}}$ . But we also have to consider the effect of the number of hydrogen bonding in the molecule. Water can form four H-bonds with four other hydrogen atoms but HF can form only one H-bond. So, HF has less effect of H-bonding than ${{\rm{H}}_2}{\rm{O}}$.
Complete step by step answer:
We know that the extent of hydrogen bonding depends on electronegativity and the number of hydrogen atoms available for bonding. Among Fluorine, nitrogen and oxygen atoms, increasing order of electronegativities is ${\rm{N}} < {\rm{O}} < {\rm{F}}$.
> Between ammonia $\left( {{\rm{N}}{{\rm{H}}_3}} \right)$ and hydrogen fluoride $\left( {{\rm{HF}}} \right)$, HF has more H-bonding than ${\rm{N}}{{\rm{H}}_3}$ because in ammonia, only one lone pair present which cannot satisfy all hydrogen also fluorine is more electronegative than nitrogen. So, option A is incorrect.
> Between water $\left( {{{\rm{H}}_2}{\rm{O}}} \right)$and hydrogen peroxide $\left( {{{\rm{H}}_2}{{\rm{O}}_{\rm{2}}}} \right)$, hydrogen peroxide has more H-bonding because of presence of one more oxygen that water. So, option B is incorrect and option C is correct.
> Between ammonia $\left( {{\rm{N}}{{\rm{H}}_3}} \right)$ and water $\left( {{{\rm{H}}_2}{\rm{O}}} \right)$, water has more H-bonding than ammonia $\left( {{\rm{N}}{{\rm{H}}_3}} \right)$ because of more number of hydrogen bond and also electronegativity of Oxygen is more than Nitrogen .

In case of water, it can form four hydrogen bonds with four other water molecules. But in case of ammonia, only one lone pair is present, which can form only one hydrogen bond with one ammonia molecule.

So, water molecules have more extent of hydrogen bonding. So, option D is incorrect.
Note: The extent of hydrogen bonding is in the order ${\rm{H}} - {\rm{F}} > {\rm{H}} - {\rm{O}} > {\rm{H}} - {\rm{N}}$, that means, extent of hydrogen bonding is more in case of HF than water $\left( {{{\rm{H}}_2}{\rm{O}}} \right)$, that is, ${\rm{HF}} > {{\rm{H}}_{\rm{2}}}{\rm{O}}$ . But we also have to consider the effect of the number of hydrogen bonding in the molecule. Water can form four H-bonds with four other hydrogen atoms but HF can form only one H-bond. So, HF has less effect of H-bonding than ${{\rm{H}}_2}{\rm{O}}$.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




