In \[Ph-CH(OH)C{{H}_{3}}\xrightarrow{SOC{{l}_{2}}}Ph-CH(Cl)C{{H}_{3}}+S{{O}_{2}}+HCl\]
Which of the following acts as a leaving group?
A. \[O{{H}^{-}}\]
B. \[C{{l}^{-}}\]
C. \[S{{O}_{2}}\]
D. 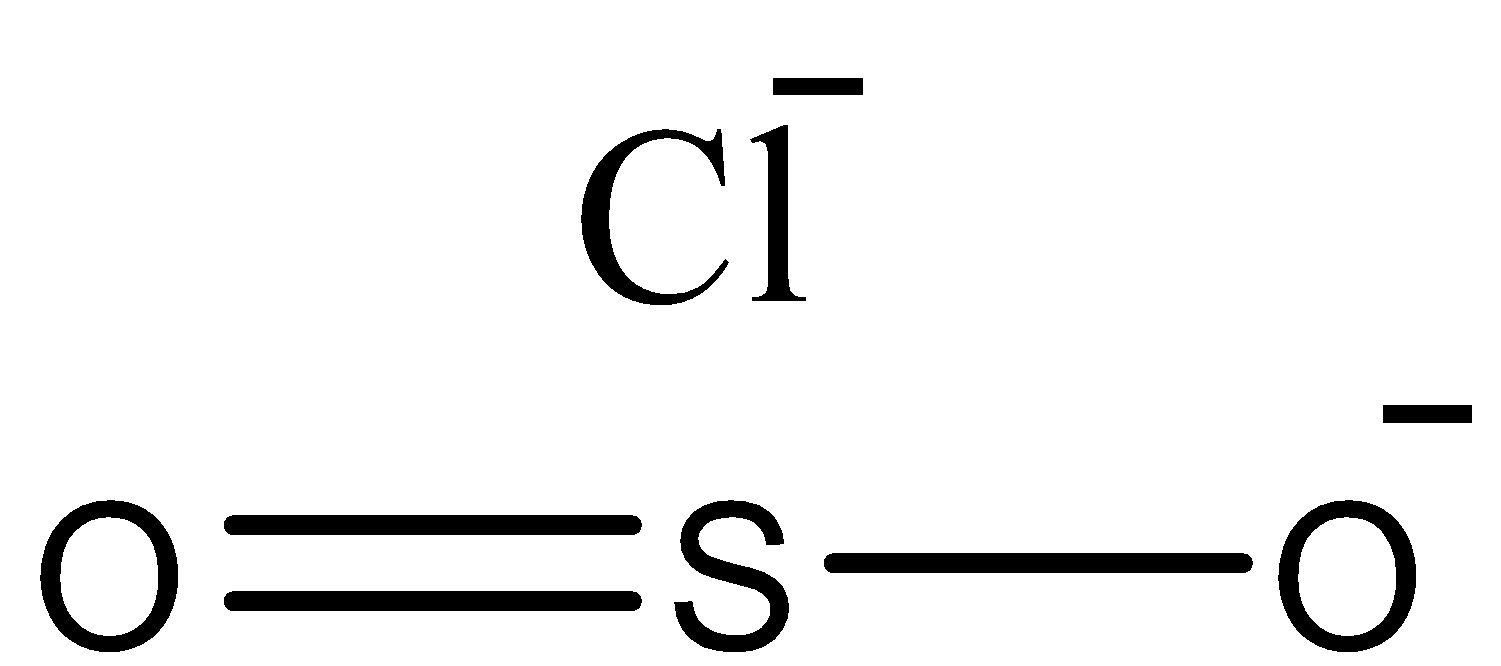
Answer
268.2k+ views
Hint: Alcohols react with thionyl chloride and form their chloro derivatives (alkyl or aryl halides) as the products. This reaction is called chlorination. Chlorination is a best example for substitution reactions.
Complete step by step answer:
* In the given reaction aryl halide is reacting with thionyl chloride and forming its halide derivative and Sulphur dioxide and hydrochloric acid as by products.
* In the question it mentioned that we have found the leaving group in the reaction.
* To know about the leaving group we should write the mechanism of the given reaction.
The mechanism of the given reaction is as follows.
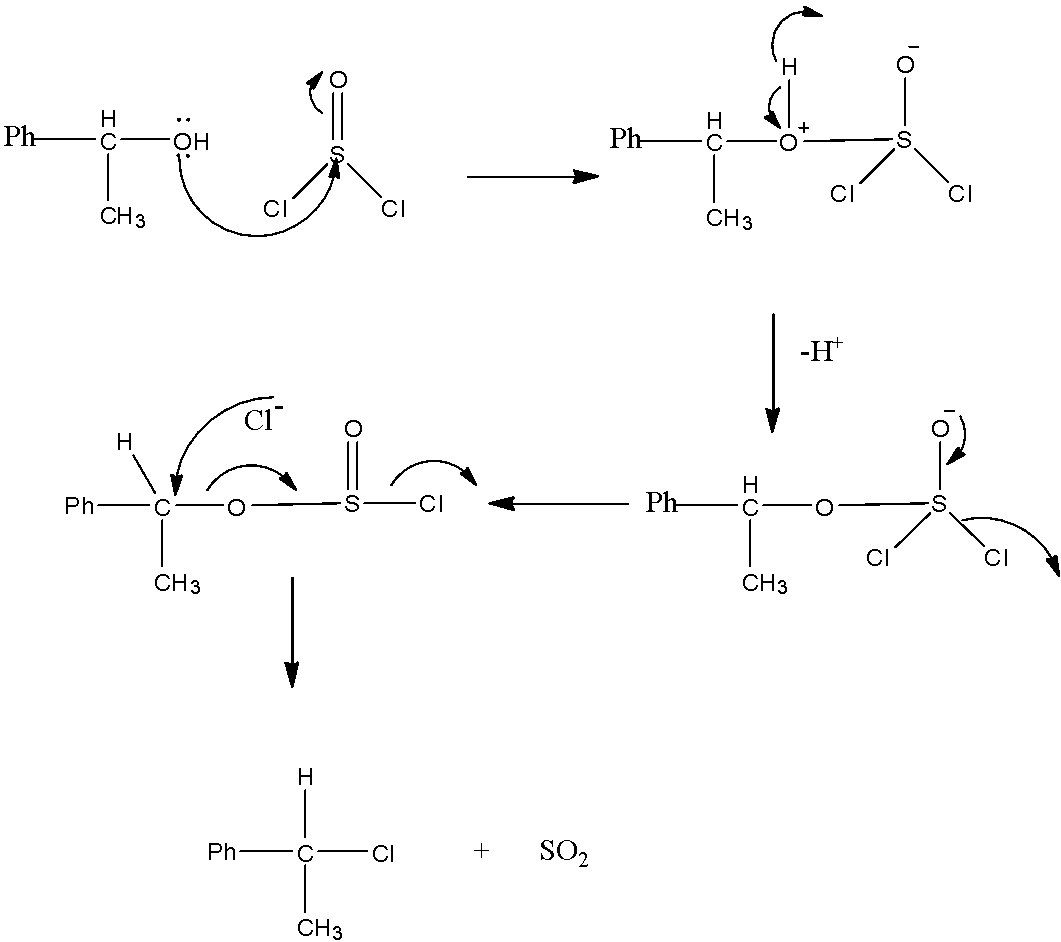
* In the first step, a lone pair of electrons on oxygen on alcohol attacks on the electron deficient Sulphur atom.
* Later to stabilize the hydrogen atom of alcohol comes out as a hydrogen ion, in continuation the negative charge on the oxygen attached to Sulphur is getting stabilized by donating electrons to Sulphur in the process chloride ion comes out.
* The liberated chloride ion attacks on the carbon which is attached to Sulphur through oxygen.
* At that time in the process of stabilization Sulphur dioxide was liberated as a by-product.
* In the given reaction the liberated product is Sulphur dioxide.
So, the correct option is C.
Note: All of us think that in the given reaction the hydroxyl group is converting into halogen in the product. So, the liberated product is the hydroxide group (\[O{{H}^{-}}\]). But through mechanism only we can say which group is going to substitute in the reaction.
Complete step by step answer:
* In the given reaction aryl halide is reacting with thionyl chloride and forming its halide derivative and Sulphur dioxide and hydrochloric acid as by products.
\[Ph-CH(OH)C{{H}_{3}}\xrightarrow{SOC{{l}_{2}}}Ph-CH(Cl)C{{H}_{3}}+S{{O}_{2}}+HCl\]
* In the question it mentioned that we have found the leaving group in the reaction.
* To know about the leaving group we should write the mechanism of the given reaction.
The mechanism of the given reaction is as follows.

* In the first step, a lone pair of electrons on oxygen on alcohol attacks on the electron deficient Sulphur atom.
* Later to stabilize the hydrogen atom of alcohol comes out as a hydrogen ion, in continuation the negative charge on the oxygen attached to Sulphur is getting stabilized by donating electrons to Sulphur in the process chloride ion comes out.
* The liberated chloride ion attacks on the carbon which is attached to Sulphur through oxygen.
* At that time in the process of stabilization Sulphur dioxide was liberated as a by-product.
* In the given reaction the liberated product is Sulphur dioxide.
So, the correct option is C.
Note: All of us think that in the given reaction the hydroxyl group is converting into halogen in the product. So, the liberated product is the hydroxide group (\[O{{H}^{-}}\]). But through mechanism only we can say which group is going to substitute in the reaction.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




