Amylopectin is a polymer of:
(A) $\beta $ D glucose
(B) $\alpha $ D glucose
(C) $\beta $ D fructose
(D) $\alpha $ D fructose
Answer
260.7k+ views
Hint:
Amylopectin is a water soluble polysaccharide and highly branched polymer. It is one of the two components of starch, the other being amylose. Glucose units are linked in a linear way with $\alpha $ glycosidic bonds.
Complete step by step answer:
Starch is a colorless and odorless polysaccharide that is found in plants as stored carbohydrates. It is a polymer of glucose monomers that are linked with each other to form polysaccharide. It is composed of two types of polysaccharide molecules:
A. Amylose
B. Amylopectin
Now, we will discuss amylopectin.
Amylopectin is a polymer of several D-glucose molecules. $80\% $ of amylopectin is present in starch.
Further, the amylopectin molecules are linked by $\alpha 1,4$-glycosidic bonds and $\alpha 1,6 - $ glycosidic bonds. When iodine is added to starch, it gives reddish-brown appearance due to the presence of amylopectin. It readily dissolves in hot water.
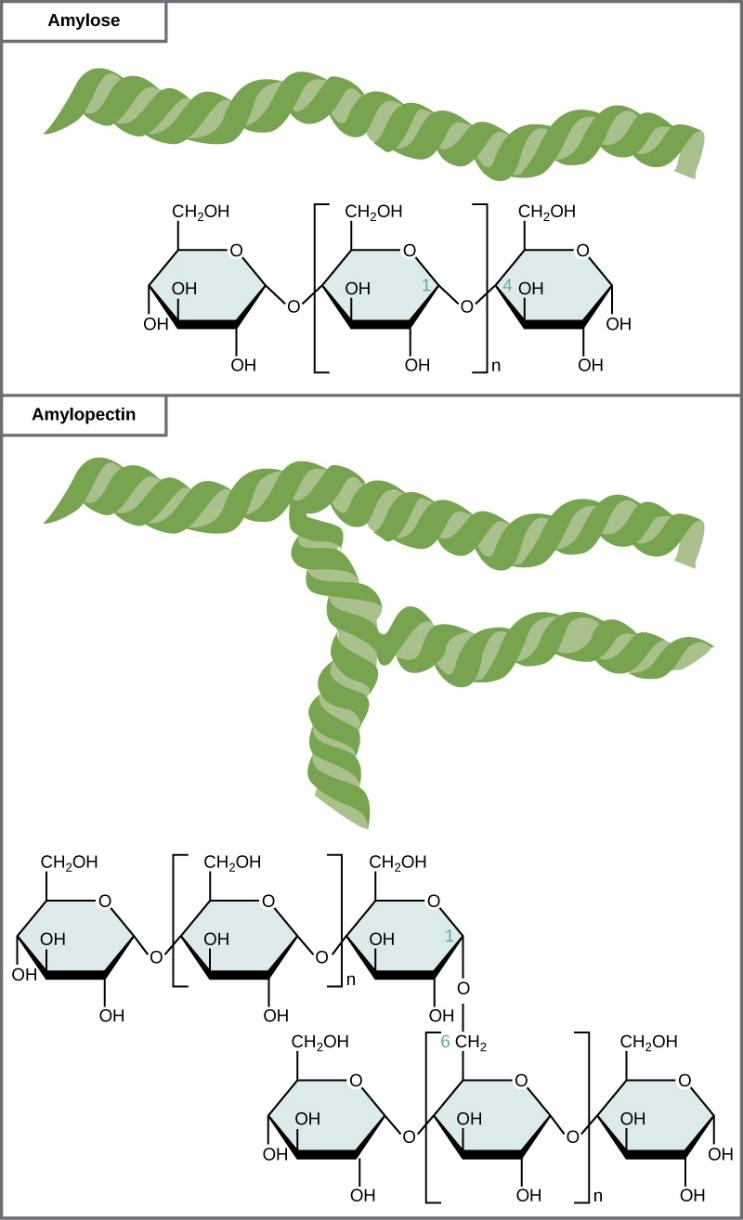
Hence, option B is correct.
Note: Polysaccharides are large molecules that feature high molecular weights and contain hundreds of glucose units joined by $\beta $ glycosidic bonds between C-$1$ and C-$4$ sites of adjacent sugars. The most important polysaccharides are cellulose, starch and glycogen.
Amylopectin is a water soluble polysaccharide and highly branched polymer. It is one of the two components of starch, the other being amylose. Glucose units are linked in a linear way with $\alpha $ glycosidic bonds.
Complete step by step answer:
Starch is a colorless and odorless polysaccharide that is found in plants as stored carbohydrates. It is a polymer of glucose monomers that are linked with each other to form polysaccharide. It is composed of two types of polysaccharide molecules:
A. Amylose
B. Amylopectin
Now, we will discuss amylopectin.
Amylopectin is a polymer of several D-glucose molecules. $80\% $ of amylopectin is present in starch.
Further, the amylopectin molecules are linked by $\alpha 1,4$-glycosidic bonds and $\alpha 1,6 - $ glycosidic bonds. When iodine is added to starch, it gives reddish-brown appearance due to the presence of amylopectin. It readily dissolves in hot water.

Hence, option B is correct.
Note: Polysaccharides are large molecules that feature high molecular weights and contain hundreds of glucose units joined by $\beta $ glycosidic bonds between C-$1$ and C-$4$ sites of adjacent sugars. The most important polysaccharides are cellulose, starch and glycogen.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




