Equilibrium - Free PDF Download
Free PDF download of NCERT Exemplar for Class 11 Chemistry Chapter 7 - Equilibrium solved by expert Chemistry teachers on Vedantu.com as per NCERT (CBSE) Book guidelines. All Chapter 7 - Equilibrium Exercise questions with solutions to help you to revise complete syllabus and score more marks in your examinations.
Question: How to download NCERT Exemplar for Class 11 Chemistry Chapter 7 Equilibrium?
Answer: Students can download NCERT Exemplar for Class 11 Chemistry Chapter 7 Equilibrium at Vedantu’s official website.
Equilibrium in a chemical reaction is defined as the state of rest in the reaction, from where there is no change in the concentration of product and the reactants. It is a crucial part of all chemical and biological processes.
For any given data of a chemical reaction, the Equilibrium constant can be defined as the ratio between the amount of product and the amount of reactant which is used to determine any chemical behaviour. At an Equilibrium state, Rate of the forward reaction = Rate of the backward reaction.
Class 11 students should study Equilibrium by solving many questions of balancing different types of chemical reactions. While balancing a chemical reaction many things should be kept in mind. Class 11 students can check out Vedantu’s official website to learn more about balancing chemical equations.
Access NCERT Exemplar Solutions for Class 11 Chemistry Chapter – 7 Equilibrium
Multiple Choice Questions (Type-I)
1.We know that the relationship between ${{\mathbf{K}}_{\mathbf{c}}}$ and $\mathbf{Kp}$ is $\mathbf{Kp}={{\mathbf{K}}_{\mathbf{c}}}{{\left( \mathbf{RT} \right)}^{\Delta \mathbf{n}}}$ What would be the value of ∆n for the reaction-
$\mathbf{N}{{\mathbf{H}}_{\mathbf{4}}}\mathbf{Cl}\left( \mathbf{s} \right)\to \mathbf{N}{{\mathbf{H}}_{\mathbf{3}}}\left(\mathbf{g}\right)+\mathbf{HCl}\text{ }\left( \mathbf{g} \right)$
(i) 1
(ii) 0.5
(iii) 1.5
(iv) 2
Ans: $\text{ }\!\!\Delta\!\!\text{ n}$= number of gaseous moles of products -number of gaseous moles of reactants
= 2-0= 2
Hence, option(iv) is the correct answer.
2.For the reaction ${{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)+{{\mathbf{I}}_{\mathbf{2}}}\left( \mathbf{g} \right)\to \mathbf{2HI}\left( \mathbf{g} \right)$ the standard free energy is $\mathbf{\Delta }{{\mathbf{G}}^{\odot }}$> 0. The equilibrium constant (K) would be __________.
(i) K = 0
(ii) K > 1
(iii) K = 1
(iv) K < 1
Ans: The given chemical reaction is:
${{\text{H}}_{\text{2}}}\left( \text{g} \right)\text{+}{{\text{I}}_{\text{2}}}\left( \text{g} \right)\to \text{2HI}\left( \text{g} \right)$
By, thermodynamical equation of equilibrium,
$\text{ }\!\!\Delta\!\!\text{ }{{\text{G}}^{\odot }}$=−RTlnK>0
⇒−RTlnK>0
Now, R>0&T>0
⇒−lnK>0
⇒K<1
Hence, option(iv) is the correct answer.
3. Which of the following is not a general characteristic of equilibria involving physical processes?
(i) Equilibrium is possible only in a closed system at a given temperature.
(ii) All measurable properties of the system remain constant.
(iii) All the physical processes stop at equilibrium.
(iv) The opposing processes occur at the same rate and there is dynamic but stable condition.
Ans: A physical equilibrium is having dynamic nature, both forward and reverse process occur at equal rates. Some kind of motion is always present. The individual molecules continuously move from one phase to another. Hence, option (iii) is the correct answer.
4.$\mathbf{PC}{{\mathbf{l}}_{\mathbf{5}}},\text{}\mathbf{PC}{{\mathbf{l}}_{\mathbf{3}}}\text{}\mathbf{and}\text{ }\mathbf{C}{{\mathbf{l}}_{\mathbf{2}}}$ are at equilibrium at 500K in a closed container and their concentrations are $\mathbf{0}.\mathbf{8}\text{}\times\text{}\mathbf{1}{{\mathbf{0}}^{\mathbf{3}\text{}}}\mathbf{mol}\text{}{{\mathbf{L}}^{\mathbf{1}}},\text{}\mathbf{1}.\mathbf{2}\text{ }\times \text{ }\mathbf{1}{{\mathbf{0}}^{\mathbf{3}}}\text{ }\mathbf{mol}\text{ }{{\mathbf{L}}^{\mathbf{1}}}$ $\text{ }\mathbf{and}\text{ }\mathbf{1}.\mathbf{2}\text{ }\times \text{ }\mathbf{1}{{\mathbf{0}}^{\mathbf{3}}}\text{ }\mathbf{mol}\text{ }{{\mathbf{L}}^{\mathbf{1}}}$ respectively. The value of ${{\mathbf{K}}_{\mathbf{c}}}$ for the reaction-
$\mathbf{PC}{{\mathbf{l}}_{\mathbf{5}\text{ }}}\left( \mathbf{g} \right)\to \mathbf{PC}{{\mathbf{l}}_{\mathbf{3}}}\text{ }\left( \mathbf{g} \right)\text{ }+\text{ }\mathbf{C}{{\mathbf{l}}_{\mathbf{2}}}\text{ }\left( \mathbf{g} \right)$will be
(i) 1.8 × $\mathbf{1}{{\mathbf{0}}^{\mathbf{3}\text{ }}}\mathbf{mol}\text{ }{{\mathbf{L}}^{\mathbf{1}}}$
(ii) 1.8 × $\mathbf{1}{{\mathbf{0}}^{-\mathbf{3}\text{ }}}$
(iii) 1.8 × $\mathbf{1}{{\mathbf{0}}^{\mathbf{3}}}\mathbf{L}\text{ }\mathbf{mo}{{\mathbf{l}}^{-1}}$
(iv) 0.55 × $\mathbf{1}{{\mathbf{0}}^{\mathbf{4}}}$
Ans: The chemical equation is given as-
${{K}_{c}}=\frac{\left[ PC{{l}_{5}} \right]\left[ PC{{l}_{3}} \right]}{\left[ C{{l}_{2}} \right]}$
=$\text{0}\text{.8 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{--3 }}}\text{mol }{{\text{L}}^{\text{--1}}}\times \text{ 1}\text{.2 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{--3}}}\text{ mol }{{\text{L}}^{\text{--1}}}$/$\text{1}\text{.2 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{--3}}}\text{ mol }{{\text{L}}^{\text{--1}}}$
=$\text{1}\text{.8}\!\!\times\!\!\text{1}{{\text{0}}^{\text{-3}}}\text{mol}{{\text{L}}^{\text{-1}}}$.
Hence, option(iii) is the correct answer.
5. Which of the following statements is incorrect?
(i) In equilibrium mixture of ice and water kept in perfectly insulated flask mass of ice and water does not change with time.
(ii) The intensity of red colour increases when oxalic acid is added to a solution containing iron (III) nitrate and potassium thiocyanate.
(iii) On addition of catalyst the equilibrium constant value is not affected. (iv) Equilibrium constant for a reaction with negative ∆H value decreases as the temperature increases.
Ans: Oxalic acid, reacts with ferric ions to form the stable complex ion$\left[ \text{Fe}{{\left( {{\text{C}}_{\text{2}}}{{\text{O}}_{\text{4}}}\text{ } \right)}_{\text{3}}} \right]{{\text{ }}^{\text{3 -}}}$, thus decreasing the concentration of free ferric ions. Because the concentration of ${{\left[ \text{Fe}\left( \text{SCN} \right) \right]}^{\text{2+}}}$decreases, as a result the intensity of red colour decreases. Hence, option (ii) is the correct answer.
6. When hydrochloric acid is added to cobalt nitrate solution at room temperature, the following reaction takes place, and the reaction mixture becomes blue. On cooling the mixture, it becomes pink. On the basis of this information mark the correct answer.
\[[Co (H_{2}O)_{6} ]_{3} +(aq) +4Cl^{-}(aq) \rightarrow [CoCl_{4}]^{2-}(aq) + 6H_{2}O (I) \]
(i) ∆H > 0 for the reaction
(ii) ∆H < 0 for the reaction
(iii) ∆H = 0 for the reaction
(iv) The sign of ∆H cannot be predicted on the basis of this information.
Ans: On cooling the mixture reaction moves towards backward direction it means it is an endothermic reaction i.e., $\Delta \text{H}$> 0. Hence, option (i) is the correct answer.
7. The pH of neutral water at 25°C is 7.0. As the temperature increases, ionisation of water increases, however, the concentration of ${{\mathbf{H}}^{+}}$ions and $\mathbf{O}{{\mathbf{H}}^{-}}$ions are equal. What will be the pH of pure water at 60°C?
(i) Equal to 7.0
(ii) Greater than 7.0
(iii) Less than 7.0
(iv) Equal to zero
Ans: As Kw increases Ans: As Kw increases $\left[ {{H}^{+}} \right]\left[ O{{H}^{-}} \right]>{{10}^{-14}}.$ As $\left[ {{H}^{+}} \right]=\left[ O{{H}^{-}} \right]$ or $\left[ {{H}^{+}} \right]=\left[ O{{H}^{-}} \right]$ $\left[ {{H}^{+}} \right]2={{110}^{-14}}$ or $\left[ {{H}^{+}} \right]>{{10}^{-7}}$
M pH < 7. Hence, option(iii) is the correct answer.
8. The ionisation constant of an acid, ${{\mathbf{K}}_{\mathbf{a}}}$, is the measure of strength of an acid. The ${{\mathbf{K}}_{\mathbf{a}}}$ values of acetic acid, hypochlorous acid and formic acid are respectively. Which of the following orders of pH of 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$solutions of these acids is correct?
(i) acetic acid > hypochlorous acid > formic acid
(ii) hypochlorous acid > acetic acid > formic acid
(iii) formic acid > hypochlorous acid > acetic acid
(iv) formic acid > acetic acid > hypochlorous acid
Ans: Higher the value of ${{\text{K}}_{\text{a}}}$ higher will be the acidic strength ${{\text{K}}_{\text{a}}}$ value for formic acid is highest and that will be of hypochlorous acid is lowest. Hence, option(iv) is the correct answer.
9. ${{\mathbf{K}}_{\mathbf{a}}}_{_{1\text{ }}},{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{2 }}}\mathbf{and}{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{3}}}$ are the respective ionisation constants for the following reactions.
${{\mathbf{H}}_{\mathbf{2}}}\mathbf{S }\to {{\mathbf{H}}^{\mathbf{+}}}\mathbf{ + H}{{\mathbf{S}}^{\mathbf{}}}$
$\mathbf{H}{{\mathbf{S}}^{\mathbf{}}}\mathbf{ }\to \mathbf{ }{{\mathbf{H}}^{\mathbf{+}}}\mathbf{ + }{{\mathbf{S}}^{\mathbf{2}}}\mathbf{ }$
${{\mathbf{H}}_{\mathbf{2}}}\mathbf{S }\to \mathbf{2}{{\mathbf{H}}^{\mathbf{+}}}\mathbf{ + }{{\mathbf{S}}^{\mathbf{2}}}$
The correct relationship between ${{\mathbf{K}}_{\mathbf{a}}}_{_{1\text{ }}}$ $,{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{2 }}}$and ${{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{3}}}$
$\left( \mathbf{i} \right)\text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{3}}}\text{ }=\text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{1\text{ }}}\text{ }\times \text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{2 }}}$
$\left( \mathbf{ii} \right)\text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{3}}}=\text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{1\text{ }}}\text{ }+{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{2 }}}$
$\left( \mathbf{iii} \right){{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{3}}}=\text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{1\text{ }}}{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{2 }}}$
$\left( \mathbf{iv} \right)\text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{3}}}\text{ }=\text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{1\text{ }}}/\text{ }{{\mathbf{K}}_{\mathbf{a}}}_{_{\mathbf{2 }}}$
Ans:
${{\text{H}}_{\text{2}}}\text{S }\to {{\text{H}}^{\text{+}}}\text{ + H}{{\text{S}}^{\text{--}}}$ ${{\text{K}}_{\text{a}}}_{_{\text{1 }}}$
$\text{H}{{\text{S}}^{\text{--}}}\to \text{ }{{\text{H}}^{\text{+}}}\text{ + }{{\text{S}}^{\text{2--}}}\text{ }$ ${{\text{K}}_{\text{a}}}_{_{\text{2 }}}$
${{\text{H}}_{\text{2}}}\text{S}\to \text{2}{{\text{H}}^{\text{+}}}\text{ + }{{\text{S}}^{\text{2--}}}$ ${{\text{K}}_{\text{a}}}_{_{\text{3}}}$
${{\text{K}}_{{{\text{a}}_{1}}}}=\frac{\left[ \text{H}{{\text{S}}^{-}} \right]\left[ {{\text{H}}^{+}} \right]}{\left[ {{\text{H}}_{2}}\text{S} \right]}$.
${{\text{K}}_{{{\text{a}}_{2}}}}=\frac{\left[ {{\text{H}}^{+}} \right]\left[ {{\text{S}}^{2-}} \right]}{\left[ \text{H}{{\text{S}}^{-}} \right]}$
${{\text{K}}_{\text{a}}}_{_{\text{3}}}$= ${{\text{K}}_{\text{a}}}_{_{\text{2 }}}$.${{\text{K}}_{\text{a}}}_{_{\text{1 }}}$
Hence, option(i) is the correct answer.
10. Acidity of $\mathbf{B}{{\mathbf{F}}_{\mathbf{3}}}$can be explained on the basis of which of the following concepts?
(i) Arrhenius concept
(ii) Bronsted Lowry concept
(iii) Lewis concept
(iv) Bronsted Lowry as well as Lewis concept.
Ans: $\text{B}{{\text{F}}_{\text{3}}}$ is an electron deficient species that is why it is a Lewis acid. (all Lewis acid can accept a pair of electrons). Hence, option (iii) is the correct answer.
11. Which of the following will produce a buffer solution when mixed in equal volumes?
(i) 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$ $\mathbf{N}{{\mathbf{H}}_{\mathbf{4}}}\mathbf{OH}$ and 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$HCl
(ii) 0.05 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$ $\mathbf{N}{{\mathbf{H}}_{\mathbf{4}}}\mathbf{OH}$ and 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$HCl
(iii) 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$ $\mathbf{N}{{\mathbf{H}}_{\mathbf{4}}}\mathbf{OH}$ and 0.05 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$HCl
(iv) 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$ $\mathbf{C}{{\mathbf{H}}_{\mathbf{4}}}\mathbf{COONa}$ and 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$NaOH
Ans: When 0.1 mol $\text{d}{{\text{m}}^{\text{-3}}}\text{N}{{\text{H}}_{\text{4}}}\text{OH}$ and 0.05 mol $\text{d}{{\text{m}}^{\text{-3}}}$ HCl react total amount of HCl reacts with $\text{N}{{\text{H}}_{\text{4}}}\text{OH}$ to form $\text{N}{{\text{H}}_{\text{4}}}\text{Cl}$ and some $\text{N}{{\text{H}}_{\text{4}}}\text{OH}$ will be left unreacted. Thus, the resultant solution contains $\text{N}{{\text{H}}_{\text{4}}}\text{Cl}$ and $\text{N}{{\text{H}}_{\text{4}}}\text{OH}$ which will produce a buffer solution. Hence, option(iii) is the correct answer.
12. In which of the following solvents is silver chloride most soluble?
(i) 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{-3}}}$ $\mathbf{AgN}{{\mathbf{O}}_{\mathbf{3}}}$solution
(ii) 0.1 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{-3}}}$ HCl solution
(iii) ${{\mathbf{H}}_{\mathbf{2}\text{ }}}\mathbf{O}$
(iv) Aqueous ammonia
Ans: Aqueous ammonia will absorb chloride ions and thus the equilibria will shift in forward direction and solubility of silver chloride will increase. Hence, option(iv) is the correct answer.
13. What will be the value of pH of 0.01 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{-3}}}$ $\mathbf{C}{{\mathbf{H}}_{\mathbf{3}}}\mathbf{COOH}$$({{\mathbf{K}}_{\mathbf{a}}}\text{ }=\text{ }\mathbf{1}.\mathbf{74}\text{ }\times \text{ }\mathbf{1}{{\mathbf{0}}^{\mathbf{5}}})$?
(i) 3.4
(ii) 3.6
(iii) 3.9
(iv) 3.0
Ans: pH for weak acid is given by the following equation:
\[pH = \frac{1}{2}[pK_{a}-log {C}] \]
\[= 21 [-log(1.74 \times 10^{-5}) - log {0.01}]\]
\[=3.4\].
Hence, option(i) is the correct answer.
14. ${{\mathbf{K}}_{\mathbf{a}}}$ for $\mathbf{C}{{\mathbf{H}}_{\mathbf{3}}}\mathbf{COOH}$is 1.8 × $\mathbf{1}{{\mathbf{0}}^{\mathbf{5}}}$and ${{\mathbf{K}}_{\mathbf{b}}}$ for $\mathbf{N}{{\mathbf{H}}_{\mathbf{4}}}\mathbf{OH}$ is 1.8 × $\mathbf{1}{{\mathbf{0}}^{\mathbf{5}}}$ . The pH of ammonium acetate will be-
(i) 7.005
(ii) 4.75
(iii) 7.0
(iv) Between 6 and 7
Ans: The equation for weak acid and weak base is given as-
pH=7+21(p${{\text{K}}_{\text{a}}}$−p${{\text{K}}_{\text{b}}}$)
${{\text{K}}_{\text{a}}}$=1.8×$\text{1}{{\text{0}}^{\text{--5}}}$
p${{\text{K}}_{\text{a}}}$=−log (1.8×$\text{1}{{\text{0}}^{\text{--5}}}$) =5−log1.8 =4.744
p${{\text{K}}_{\text{b}}}$=−log (1.8×$\text{1}{{\text{0}}^{\text{--5}}}$) =4.744
pH=7+21(p${{\text{K}}_{\text{a}}}$−p${{\text{K}}_{\text{b}}}$) =7.
Hence, option(iii) is the correct answer.
15. Which of the following options will be correct for the stage of half completion of the reaction $\mathbf{A}\rightleftharpoons \mathbf{B}$.
(i) $\mathbf{\Delta }{{\mathbf{G}}^{\odot }}$=0
(ii) $\mathbf{\Delta }{{\mathbf{G}}^{\odot }}$> 0
(iii) $\mathbf{\Delta }{{\mathbf{G}}^{\odot }}$ < 0
(iv) $\mathbf{\Delta }{{\mathbf{G}}^{\odot }}$= –RT ln2
Ans: At the stage of half completion of reaction [A] = [B]. Therefore, K = 1. Hence, $\text{ }\!\!\Delta\!\!\text{ }{{\text{G}}^{\odot }}$=0. Hence, option(i) is the correct answer.
16. On increasing the pressure, in which direction will the gas phase reaction proceed to re-establish equilibrium, is predicted by applying the Le Chatelier’s principle. Consider the reaction. ${{\mathbf{N}}_{\mathbf{2}}}\left( \mathbf{g} \right)\mathbf{+3}{{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)\to \mathbf{2N}{{\mathbf{H}}_{\mathbf{3}}}\mathbf{(g)}$ (g) Which of the following is correct, if the total pressure at which the equilibrium is established, is increased without changing the temperature? (i) K will remain same
(ii) K will decrease
(iii) K will increase
(iv) K will increase initially and decrease when pressure is very high.
Ans: Equilibrium constant K will remain same if temperature is not changed. Hence, option(i) is the correct answer.
17. What will be the correct order of vapour pressure of water, acetone, and ether at 30$^{\mathbf{{}^\circ }}\mathbf{C}$. Given that among these compounds, water has maximum boiling point and ether has minimum boiling point?
(i) Water < ether < acetone
(ii) Water < acetone < ether
(iii) Ether < acetone < water
(iv) Acetone < ether < water
Ans: Greater the boiling point, lower will be the vapour pressure. Hence, option(ii) is the correct answer.
18. At 500 K, equilibrium constant, ${{\mathbf{K}}_{\mathbf{c}}}$, for the following reaction is 5. $\frac{\mathbf{1}}{\mathbf{2}}\text{ }{{\mathbf{H}}_{\mathbf{2}}}\text{ }\left( \mathbf{g} \right)+\frac{\mathbf{1}}{\mathbf{2}}{{\mathbf{I}}_{\mathbf{2}}}\left( \mathbf{g} \right)\to \mathbf{HI}\text{ }(\mathbf{g})$ .What would be the equilibrium constant Kc for the reaction $2\mathbf{HI}\text{ }(\mathbf{g})\to \text{ }{{\mathbf{H}}_{\mathbf{2}}}\text{ }\left( \mathbf{g} \right)+{{\mathbf{I}}_{\mathbf{2}}}\left( \mathbf{g} \right)$.
(i) 0.04
(ii) 0.4
(iii) 25
(iv) 2.5
Ans: If the equation is multiplied by 2, the equilibrium constant for the new equation is the square of K it means we must do the square of 5 that is 25. Then and on reversing the reaction the value of the equilibrium constant is inversed means the value of K comes out to be 1/25= 0.04. Hence, option(i) is the correct answer.
19. In which of the following reactions, the equilibrium remains unaffected on addition of small amount of argon at constant volume?
$\left( \mathbf{i} \right)\text{ }{{\mathbf{H}}_{\mathbf{2}}}\text{ }\left( \mathbf{g} \right)+{{\mathbf{I}}_{\mathbf{2}}}\text{ }\left( \mathbf{g} \right)\rightleftharpoons \mathbf{2HI}\text{ }\left( \mathbf{g} \right)$
$\left( \mathbf{ii} \right)\text{ }\mathbf{PC}{{\mathbf{l}}_{\mathbf{5}}}\left( \mathbf{g} \right)\rightleftharpoons \mathbf{PC}{{\mathbf{l}}_{\mathbf{3}}}\left( \mathbf{g} \right)+\mathbf{C}{{\mathbf{l}}_{\mathbf{2}\text{ }}}\left( \mathbf{g} \right)$
$\left( \mathbf{iii} \right)\text{ }{{\mathbf{N}}_{\mathbf{2}}}\left( \mathbf{g} \right)+\mathbf{3}{{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)\rightleftharpoons \mathbf{2N}{{\mathbf{H}}_{\mathbf{3}}}\left( \mathbf{g} \right)$
(iv) The equilibrium will remain unaffected in all the three cases.
Ans: At constant volume equilibrium remains unaffected with the addition of inert gas. Hence, option(iv) is the correct answer.
Multiple Choice Questions (Type-II)
In the following questions two or more options may be correct.
20. For the reaction the value of K is 50 at 400 K and 1700 at 500 K. Which of the following options is correct?
(i) The reaction is endothermic.
(ii) The reaction is exothermic.
(iii) If $\mathbf{N}{{\mathbf{O}}_{\mathbf{2}}}\left( \mathbf{g} \right)\text{ }\mathbf{and}\text{ }{{\mathbf{N}}_{\mathbf{2}}}{{\mathbf{O}}_{\mathbf{4}}}\left( \mathbf{g} \right)$are mixed at 400 K at partial pressures 20 bar and 2 bar respectively, more ${{\mathbf{N}}_{\mathbf{2}}}{{\mathbf{O}}_{\mathbf{4}}}$ (g) will be formed.
(iv) The entropy of the system increases.
Ans: As, K increases with increase in temperature. K increases, it shows that the reaction must be endothermic. No. of molecules increases thus, there is increase in entropy. Hence, option(i), (iii) and (iv) are the correct answers.
21. At a particular temperature and atmospheric pressure, the solid and liquid phases of a pure substance can exist in equilibrium. Which of the following term defines this temperature?
(i) Normal melting point
(ii) Equilibrium temperature
(iii) Boiling point
(iv) Freezing point
Ans: Ice and water are in equilibrium only at particular temperature and pressure. For any pure substance at atmospheric pressure, the temperature at which the solid and liquid phases are at equilibrium is called the normal melting point or normal freezing point of the substance. For example, it is 4°C in case of water. The system here is in dynamic equilibrium. Hence, options (i) and (iv) are the correct answers.
Short Answer Type
22. The ionisation of hydrochloric acid in water is given below:
$\mathbf{HCl}\left( \mathbf{aq} \right)\mathbf{ + }{{\mathbf{H}}_{\mathbf{2}}}\mathbf{O }\left( \mathbf{l } \right)\mathbf{ }\rightleftharpoons \mathbf{ }{{\mathbf{H}}_{\mathbf{3}}}{{\mathbf{O}}^{\mathbf{+}}}\mathbf{ }\left( \mathbf{aq} \right)\mathbf{ + C}{{\mathbf{l}}^{\mathbf{}}}\mathbf{ }\left( \mathbf{aq} \right)$
Label two conjugate acid-base pairs in this ionisation.
Ans: Conjugate acid and conjugate base can be shown as-
$\text{HCl}\left( \text{aq} \right)\text{ + }{{\text{H}}_{\text{2}}}\text{O }\left( \text{l } \right)\text{ }\rightleftharpoons \text{ }{{\text{H}}_{\text{3}}}{{\text{O}}^{\text{+}}}\text{ }\left( \text{aq} \right)\text{ + C}{{\text{l}}^{\text{--}}}\text{ }\left( \text{aq} \right)$
acid base conjugate acid conjugate base
23. The aqueous solution of sugar does not conduct electricity. However, when sodium chloride is added to water, it conducts electricity. How will you explain this statement on the basis of ionisation and how is it affected by concentration of sodium chloride?
Ans: Conductance of a solution depends upon the number of ions present in the solution since sodium chloride is an ionic compound when mixed in water it ionizes completely forming an aqueous electrolytic solution. Hence, the solution conducts electricity while sugar being a non-electrolyte does not ionize in water and when concentration of NaCl is increased, more $\text{N}{{\text{a}}^{\text{+}}}$ and $\text{C}{{\text{l}}^{\text{-}}}$ ions will be produced. Hence, conductance increases.
24. $\mathbf{B}{{\mathbf{F}}_{\mathbf{3}}}$does not have proton but still acts as an acid and reacts with $\mathbf{N}{{\mathbf{H}}_{\mathbf{3}}}$. Why is it so? What type of bond is formed between the two?
Ans: $\text{B}{{\text{F}}_{\text{3}}}$ does not have a proton but still it acts as a Lewis acid as it is an electron pair acceptor and reacts with $\text{N}{{\text{H}}_{\text{3}}}$by accepting its lone pair of electrons. The reaction can be represented by Coordinate bond is formed between $\text{B}{{\text{F}}_{\text{3}}}\text{ and N}{{\text{H}}_{\text{3}}}$. It can be represented as-
$\text{ }\!\![\!\!\text{ B}{{\text{F}}_{\text{3}}}\leftarrow \mathbf{:}\text{N}{{\text{H}}_{\text{3}}}]$
Nitrogen acts as lone pair donator in this combination.
25. Ionisation constant of a weak base MOH, is given by the expression ${{\mathbf{k}}_{\mathbf{b}}}\mathbf{=}\frac{\mathbf{[}{{\mathbf{M}}^{\mathbf{+}}}\mathbf{][O}{{\mathbf{H}}^{\mathbf{-}}}\mathbf{]}}{\mathbf{[MOH]}}$
Values of ionisation constant of some weak bases at a particular temperature are given below:
Base | Dimethylamine | Urea | Pyridine | Ammonia |
${{\mathbf{K}}_{\mathbf{b}}}$ | $\mathbf{5}\mathbf{.4\times 1}{{\mathbf{0}}^{\mathbf{-4}}}$ | $\mathbf{1}\mathbf{.3\times 1}{{\mathbf{0}}^{\mathbf{-14}}}$ | $\mathbf{1}\mathbf{.77\times 1}{{\mathbf{0}}^{\mathbf{-9}}}$ | $\mathbf{1}\mathbf{.77\times 1}{{\mathbf{0}}^{\mathbf{-5}}}$ |
Arrange the bases in decreasing order of the extent of their ionisation at equilibrium. Which of the above base is the strongest?
Ans: Higher the value of ${{\text{K}}_{\text{b}}}$ stronger will be the base.
${{\text{K}}_{\text{b}}}$- $\text{5}\text{.4 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-4}}}$>$\text{1}\text{.77 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-5}}}$> $\text{1}\text{.77 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-9}}}$> $\text{1}\text{.3 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-14}}}$
Decreasing order of basic strength.
Dimethylamine > Ammonia > Pyridine > Urea
Hence among the given bases, the strongest base is Dimethylamine.
26. Conjugate acid of a weak base is always stronger. What will be the decreasing order of basic strength of the following conjugate bases? $\mathbf{O}{{\mathbf{H}}^{}}\text{ },\text{ }\mathbf{R}{{\mathbf{O}}^{}}\text{ },\text{ }\mathbf{C}{{\mathbf{H}}_{\mathbf{3}}}\mathbf{CO}{{\mathbf{O}}^{}}\text{ },\text{ }\mathbf{C}{{\mathbf{l}}^{}}$.
Ans: As the Conjugate acid of a weak base is always stronger and conjugate base of weak acid is always strong.
The weak acids are:
⇒ ${{\text{H}}_{\text{2}}}\text{O,ROH,C}{{\text{H}}_{\text{3}}}\text{COOH,HCl}$
⇒ $\text{HClC}{{\text{H}}_{\text{3}}}\text{COOHROH}{{\text{H}}_{\text{2}}}\text{O}$
⇒ Conjugate base order ⇒ $\text{O}{{\text{H}}^{\text{-}}}\text{R}{{\text{O}}^{\text{-}}}\text{C}{{\text{H}}_{\text{3}}}\text{CO}{{\text{O}}^{\text{-}}}\text{C}{{\text{l}}^{\text{-}}}$
27. Arrange the following in increasing order of pH.
$\mathbf{KN}{{\mathbf{O}}_{\mathbf{3}}}\text{ }\left( \mathbf{aq} \right),\text{ }\mathbf{C}{{\mathbf{H}}_{\mathbf{3}}}\mathbf{COONa}\text{ }\left( \mathbf{aq} \right),\text{ }\mathbf{N}{{\mathbf{H}}_{\mathbf{4}}}\mathbf{Cl}\text{ }\left( \mathbf{aq} \right),\text{ }{{\mathbf{C}}_{\mathbf{6}}}{{\mathbf{H}}_{\mathbf{5}}}\mathbf{COON}{{\mathbf{H}}_{\mathbf{4}\text{ }}}\left( \mathbf{aq} \right)$.
Ans: Correct order of pH is -
$\text{N}{{\text{H}}_{\text{4}}}\text{Cl}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}\text{COON}{{\text{H}}_{\text{4}}}\text{KN}{{\text{O}}_{\text{3}}}\text{C}{{\text{H}}_{\text{3}}}\text{COONa}$
Salts of strong acid and strong base do not hydrolyse and form neutral solution thus, pH will be nearly 7 of $\text{KN}{{\text{O}}_{\text{3}}}$.
In sodium acetate, acetic acid remains unionised this results in increase in $\text{O}{{\text{H}}^{\text{-}}}$ concentration and pH will be more than 7.
$\text{N}{{\text{H}}_{\text{4}}}\text{Cl}$formed from weak base, $\text{N}{{\text{H}}_{\text{4}}}\text{OH}$and strong acid, HCl, in water dissociates completely, aq. ammonium ions undergo hydrolysis with water to form $\text{N}{{\text{H}}_{\text{4}}}\text{OH}$and ${{\text{H}}^{\text{+}}}$ions resulting in less pH value.
28. The value of ${{\mathbf{K}}_{\mathbf{c}}}$ for the reaction $\mathbf{2HI }\left( \mathbf{g} \right)\to {{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)\mathbf{+ }{{\mathbf{I}}_{\mathbf{2}}}\mathbf{ }\left( \mathbf{g} \right)\mathbf{ is 1 \times 1}{{\mathbf{0}}^{\mathbf{4}}}$At a given time, the composition of reaction mixture is [HI] = 2 × $\mathbf{1 \times 1}{{\mathbf{0}}^{\mathbf{5}}}$mol, \[[H_{2}] = 1 \times 1 \times 10^{-5} \] mol and [${{\mathbf{I}}_{\mathbf{2}}}$] = 1 × $\mathbf{1 \times 1}{{\mathbf{0}}^{\mathbf{5}}}$ mol. In which direction will the reaction proceed?
Ans: We can calculate the reaction quotient Q for the reaction and that will be given by the expression:
\[ Q = \frac{[H_{2}][I_{2}]}{[HI]} = \frac{(2 \times 10 - 5)21 \times 10^{-5} }{(2 \times 10^{-5})^2 1} = \frac{1}{4}\]
=0.25=2.5×$\text{1}{{\text{0}}^{\text{-1}}}$
As the value of the reaction quotient is greater than the value of ${{\text{K}}_{\text{c}}}$ that is $\text{1 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{--4}}}$ the reaction will proceed in the reverse direction.
29. On the basis of the equation \[ pH = -log [H^{+}]\], the pH of $\mathbf{1}{{\mathbf{0}}^{\mathbf{8}\text{ }}}\mathbf{mol}\text{ }\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$solution of HCl should be 8. However, it is observed to be less than 7.0. Explain the reason.
Ans: Here concentration of water cannot be neglected since the solution is very dilute. pH will be less than 7.0. Hence, the total concentration is given as -
$\text{ }\!\![\!\!\text{ }{{\text{H}}_{\text{3}}}{{\text{O}}^{\text{+}}}\text{ }\!\!]\!\!\text{ =1}{{\text{0}}^{\text{-8}}}\text{+1}{{\text{0}}^{\text{-7}}}\text{M}$.
30. pH of a solution of a strong acid is 5.0. What will be the pH of the solution obtained after diluting the given solution 100 times?
Ans: Given with the pH = 5 as, $\text{pH=-- log }\!\![\!\!\text{ }{{\text{H}}^{\text{+}}}\text{ }\!\!]\!\!\text{ }$
$\text{ }\!\![\!\!\text{ }{{\text{H}}^{\text{+}}}\text{ }\!\!]\!\!\text{ =1}{{\text{0}}^{\text{-5}}}\text{M}$
On 100 times dilution-
$\text{ }\!\![\!\!\text{ }{{\text{H}}^{\text{+}}}\text{ }\!\!]\!\!\text{ =1}{{\text{0}}^{\text{-7}}}\text{M}$.
31. A sparingly soluble salt gets precipitated only when the product of concentration of its ions in the solution (${{\mathbf{Q}}_{\mathbf{sp}}}$) becomes greater than its solubility product. If the solubility of $\mathbf{BaS}{{\mathbf{O}}_{\mathbf{4}}}$in water is 8$\times \text{ }\mathbf{1}{{\mathbf{0}}^{\mathbf{4}\text{ }}}\mathbf{mol}\text{ }\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$. Calculate its solubility in 0.01 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$of ${{\mathbf{H}}_{\mathbf{2}}}\mathbf{S}{{\mathbf{O}}_{\mathbf{4}}}$.
Ans: The chemical equation is given below-
$\text{BaS}{{\text{O}}_{\text{4}}}\left( \text{s} \right)\to \text{B}{{\text{a}}^{\text{2+}}}\left( \text{aq} \right)\text{ + S}{{\text{O}}_{\text{4}}}^{\text{2-}}\left( \text{aq} \right)$
At t = 0 1 0 0
At equilibrium
in water 1-S S S
At equilibrium in
the presence 1-S S (S+0.01)
of sulphuric acid
${{\text{K}}_{\text{sp}}}\text{ for BaS}{{\text{O}}_{\text{4}}}\text{ in water = }\!\![\!\!\text{ B}{{\text{a}}^{\text{2+}}}\text{ }\!\!]\!\!\text{ }\!\![\!\!\text{ S}{{\text{O}}_{\text{2}}}^{\text{4-}}\text{ }\!\!]\!\!\text{ = }\left( \text{S} \right)\text{ }\left( \text{S} \right)\text{ = }{{\text{S}}^{\text{2}}}$
But S = 8 x $\text{1}{{\text{0}}^{\text{-4}}}\text{ mol d}{{\text{m}}^{\text{-3}}}$
${{\text{K}}_{\text{sp}}}\text{ = }{{\left( \text{8 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-4}}} \right)}^{\text{2}}}\text{ = 64 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-8}}}\text{ }$... (1)
The expression for ${{\text{K}}_{\text{sp}}}$ in the presence of sulphuric acid will be as follows:
${{\text{K}}_{\text{sp}}}$ = (S) (5 + 0.01) ... (2)
Since value of ${{\text{K}}_{\text{sp}}}$will not change in the presence of sulphuric acid, therefore from (1) and (2)
(S) (S + 0.01) = $\text{64 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-8}}}$
${{\text{S}}^{\text{2}}}$ + 0.01 S = $\text{64 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-8}}}$
${{\text{S}}^{\text{2}}}$ + 0.01 S - $\text{64 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-8}}}$= 0
$\text{S=}\frac{\text{-0}\text{.01 }\!\!\pm\!\!\text{ }\sqrt{{{\text{(0}\text{.01)}}^{\text{2}}}\text{+(}}\text{4 }\!\!\times\!\!\text{ 64 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-8}}}\text{)}}{\text{2}}$
When we solve the above equation, we will get the value of
S is= $\text{6 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-4}}}\text{ mo}{{\text{l}}^{\text{-1}}}\text{ d}{{\text{m}}^{\text{-3}}}$
32. pH of 0.08 mol $\mathbf{d}{{\mathbf{m}}^{\mathbf{3}}}$HOCl solution is 2.85. Calculate its ionisation constant.
Ans: Ionisation constant can be calculated as given below-
pH of HOCl=2.85
But \[pH = log[H^{+}]\]
\[ \therefore -2.85 = log [H^{+}]\]
\[\Rightarrow 3^{-} .15 = log [H^+] \]
\[\Rightarrow [H^+] antilog 3^{-} .15 \]
\[\Rightarrow [H^+] = 1.413 \times 10^{-5} \]
For weak mono basic acid \[[H^+] = K_{a} \times C \]
\[ K_{a} = C[H^+]^2 = 0.08(.413 \times 10^{-3})^{2} \]
\[ = 24.957 \times 10^{-6} = 2.4957 \times 10^{-5}\]
33. Calculate the pH of a solution formed by mixing equal volumes of two solutions A and B of a strong acid having pH = 6 and pH = 4 respectively.
Ans: we can find the pH of the solution as-
pH of solution A=6
Hence, concentration of [[{{\text{H}}^{\text{+}}}]] ion in solution A=$\text{1}{{\text{0}}^{\text{-6}}}$$\text{mol }{{\text{L}}^{\text{-1}}}$
pH of solution B=4
Therefore, concentration of [{{\text[{H}]}^{\text{+}}}] ion in solution B=$\text{1}{{\text{0}}^{\text{-4}}}$$\text{mol }{{\text{L}}^{\text{-1}}}$
On mixing one litre of each solution, total volume = 1L+1L=2L.
Amount of ${{\text{H}}^{\text{+}}}$ ions in 1L of solution
A= concentration× Volume(V)= $\text{1}{{\text{0}}^{\text{-6}}}$mol× IL
Amount of ${{\text{H}}^{\text{+}}}$ ions in 1L of solution B=$\text{1}{{\text{0}}^{\text{-4}}}$mol×1L
∴ Total amount of ${{\text{H}}^{\text{+}}}$ ions in the solution formed by mixing solutions A and B is ($\text{1}{{\text{0}}^{\text{-5}}}$mol+$\text{1}{{\text{0}}^{\text{-4}}}$mol)
This amount is present in 2L solution,
∴ Total \[{{\text{H}}^{\text{+}}}\]=2$\text{1}{{\text{0}}^{\text{-4}}}$ (1+0.01)=21.01×$\text{1}{{\text{0}}^{\text{-4}}}$$\text{mol }{{\text{L}}^{\text{-1}}}$
=21.01×$\text{1}{{\text{0}}^{\text{-4}}}$$\text{mol }{{\text{L}}^{\text{-1}}}$ =0.5×10−4mol L−1 =5×$\text{1}{{\text{0}}^{\text{-5}}}$$\text{mol }{{\text{L}}^{\text{-1}}}$.
pH=−log[{{\text[{H}]}^{\text{+}}}]=−log(5×$\text{1}{{\text{0}}^{\text{-5}}}$)=−[log5+(−5log10)]
=−log5+5=5−log5=5−0.6990=4.3010=4.3
Thus, the pH of the solution will be 4.3.
34. The solubility product of $\mathbf{Al}{{\left( \mathbf{OH} \right)}_{\mathbf{3}}}\text{ }\mathbf{is}\text{ }\mathbf{2}.\mathbf{7}\text{ }\times \text{ }\mathbf{1}{{\mathbf{0}}^{\mathbf{11}}}$. Calculate its solubility in $\mathbf{g}{{\mathbf{L}}^{\mathbf{1}}}$and also find out pH of this solution. (Atomic mass of Al = 27 u).
Ans: Let us assume S be the solubility of $\text{Al}{{\left( \text{OH} \right)}_{\text{3}}}$.
${{\text{K}}_{\text{s}}}_{\text{p}}\text{=}\left[ \text{A}{{\text{l}}^{\text{3+}}} \right]{{\left[ \text{O}{{\text{H}}^{\text{-}}} \right]}^{\text{3}}}\text{=}\left( \text{S} \right){{\left( \text{3S} \right)}^{\text{3}}}\text{=27}{{\text{S}}^{\text{4}}}$
${{\text{S}}^{\text{4}}}\text{=27}{{\text{K}}_{\text{sp}}}\text{=27 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{27}}}\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-11}}}\text{=1 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-12}}}$
S=1×${{10}^{-3}}$ $\text{mol }{{\text{L}}^{\text{-1}}}$.
(i) Solubility of $\text{Al}{{\left( \text{OH} \right)}_{\text{3}}}\text{ }$: Molar mass of $\text{Al}{{\left( \text{OH} \right)}_{\text{3}}}\text{ }$ is 78g. Hence, solubility of $\text{Al}{{\left( \text{OH} \right)}_{\text{3}}}\text{ }$in $\text{g}{{\text{L}}^{\text{-1}}}\text{=1 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{3}}}\text{ }\!\!\times\!\!\text{ 78g}{{\text{L}}^{\text{-1}}}\text{=78 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-3}}}\text{g}{{\text{L}}^{\text{-1}}}\text{=7}\text{.8 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-2}}}\text{g}{{\text{L}}^{\text{-1}}}$
(ii) pH of the solution: S=1× ${{10}^{-3}}$mol ${{\text{L}}^{\text{-1}}}$
[OH]=3S=3×1× ${{10}^{-3}}$=3× ${{10}^{-3}}$
p[OH]=3−log3
pH=14−pOH=11+log3=11.4771.
35. Calculate the volume of water required to dissolve 0.1 g lead (II) chloride to get a saturated solution. (${{\mathbf{K}}_{\mathbf{s}}}_{\mathbf{p}}$ of $\mathbf{PbC}{{\mathbf{l}}_{\mathbf{2}}}=\mathbf{3}.\mathbf{2}\text{ }\times \text{ }\mathbf{1}{{\mathbf{0}}^{\mathbf{8}}}$, atomic mass of Pb = 207 u).
Ans:
${{\text{K}}_{\text{sp}}}\text{=3}\text{.2 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-8}}}$
$\text{PbC}{{\text{l}}_{\text{2}}}\rightleftharpoons \text{P}{{\text{b}}^{\text{2+}}}\text{+2C}{{\text{l}}^{\text{-}}}$
${{\text{K}}_{\text{sp}}}\text{=}\left( \text{S} \right){{\left( \text{2S} \right)}^{\text{2}}}$
${{\text{K}}_{\text{sp}}}\text{=4}{{\text{S}}^{\text{3}}}$
${{\text{s}}^{\text{3}}}\text{=43}\text{.2 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-8}}}$
${{\text{s}}^{\text{3}}}\text{=8 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-9}}}$
$\text{s=2 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-3}}}$
$\text{Molar }\!\!~\!\!\text{ mass }\!\!~\!\!\text{ of }\!\!~\!\!\text{ PbC}{{\text{l}}_{\text{2}}}\text{=278g/mol }\!\!~\!\!\text{ }$
$\text{By using Molarity formula, }\!\!~\!\!\text{ }\!\!~\!\!\text{ }$
$\text{2 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-3}}}\text{=278 }\!\!\times\!\!\text{ x0}\text{.1}$
$\text{x=}\frac{\text{0}\text{.1}}{\text{278 }\!\!\times\!\!\text{ 2 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-3}}}}$
$\text{ }\!\!~\!\!\text{ =}\frac{\text{100}}{\text{278 }\!\!\times\!\!\text{ 2}}$
$\text{x=0}\text{.18L }\!\!~\!\!\text{ }$
36. A reaction between ammonia and boron trifluoride is given below: : $\mathbf{N}{{\mathbf{H}}_{\mathbf{3}}}+\mathbf{B}{{\mathbf{F}}_{\mathbf{3}}}\text{ }\to {{\mathbf{H}}_{\mathbf{3}}}\mathbf{N}\text{ }:\text{ }\mathbf{B}{{\mathbf{F}}_{\mathbf{3}}}$ Identify the acid and base in this reaction. Which theory explains it? What is the hybridisation of B and N in the reactants?
Ans: As, $\text{N}{{\text{H}}_{\text{3}}}$ is Lewis base while $\text{B}{{\text{F}}_{\text{3}}}\text{ }$ is Lewis acid. Lewis electronic theory of acids and bases explains it. Hybridisation state: N in $\text{N}{{\text{H}}_{\text{3}}}$ is $\text{s}{{\text{p}}^{\text{3}}}$hybridised and Boron in $\text{B}{{\text{F}}_{\text{3}}}$ is $\text{s}{{\text{p}}^{2}}$ hybridised.
37. Following data is given for the reaction: $\mathbf{CaC}{{\mathbf{O}}_{\mathbf{3}}}\left( \mathbf{s} \right)\to \mathbf{CaO}\left( \mathbf{s} \right)+\mathbf{C}{{\mathbf{O}}_{\mathbf{2}}}\mathbf{(g)}$
${{\mathbf{\Delta }}_{\mathbf{f}}}{{\mathbf{H}}^{\odot }}\left[ \mathbf{CaO}\left( \mathbf{s} \right) \right]\text{ }=\text{ }\text{ }\mathbf{635}.\mathbf{1}\text{ }\mathbf{kJmo}{{\mathbf{l}}^{\mathbf{1}}}$
${{\mathbf{\Delta }}_{\mathbf{f}}}{{\mathbf{H}}^{\odot }}\left[ \mathbf{C}{{\mathbf{O}}_{\mathbf{2}}}\left( \mathbf{g} \right) \right]\text{ }=\text{ }\text{ }\mathbf{393}.\mathbf{5}\text{ }\mathbf{kJmo}{{\mathbf{l}}^{\mathbf{1}}}$
${{\mathbf{\Delta }}_{\mathbf{f}}}{{\mathbf{H}}^{\odot }}\left[ \mathbf{CaC}{{\mathbf{O}}_{\mathbf{3}}}\left( \mathbf{s} \right) \right]\text{ }=\text{ }\text{ }\mathbf{1206}.\mathbf{9}\text{ }\mathbf{kJmo}{{\mathbf{l}}^{\mathbf{1}}}$
Predict the effect of temperature on the equilibrium constant of the above reaction.
Ans: As, enthalpy of formation is given by-
${{\text{ }\!\!\Delta\!\!\text{ }}_{\text{r}}}{{\text{H}}^{\odot }}\text{=}{{\text{ }\!\!\Delta\!\!\text{ }}_{\text{f}}}{{\text{H}}^{\odot }}\left[ \text{CaO}\left( \text{s} \right) \right]\text{+}{{\text{ }\!\!\Delta\!\!\text{ }}_{\text{f}}}{{\text{H}}^{\odot }}\left[ \text{C}{{\text{O}}_{\text{2}}}\left( \text{g} \right) \right]\text{-}{{\text{ }\!\!\Delta\!\!\text{ }}_{\text{f}}}{{\text{H}}^{\odot }}\left[ \text{CaC}{{\text{O}}_{\text{3}}}\left( \text{s} \right) \right]$
${{\text{ }\!\!\Delta\!\!\text{ }}_{\text{r}}}{{\text{H}}^{\odot }}\text{=178}\text{.3kJ }\!\!~\!\!\text{ mo}{{\text{l}}^{\text{-1}}}$.
The reaction is endothermic. Therefore, according to Le Chatelier’s principle, reaction will proceed in forward direction on increasing temperature.
Matching Type
38. Match the following equilibria with the corresponding condition.
(i) Liquid $\rightleftharpoons $ Vapour | (a) Saturated solution |
(ii) Solid $\rightleftharpoons $ Liquid | (b) Boiling point |
(iii) Solid $\rightleftharpoons $ Vapour | (c) Sublimation point |
(iv) Solute$\rightleftharpoons $ Solute(solution) | (d) Melting point |
(e) Unsaturated solution |
Ans: (i) $\to $ (b); (ii) $\to $ (d); (iii) $\to $ (c); (iv) $\to $ (a).
39. For the reaction: ${{\mathbf{N}}_{\mathbf{2}}}\left( \mathbf{g} \right)+\mathbf{3}{{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)\rightleftharpoons \mathbf{2N}{{\mathbf{H}}_{\mathbf{3}}}\left( \mathbf{g} \right)$
Equilibrium constant ${{\mathbf{K}}_{\mathbf{c}}}\mathbf{ = }\frac{{{\left[ \mathbf{N}{{\mathbf{H}}_{\mathbf{3 }}} \right]}^{\mathbf{2}}}}{{{\left[ \mathbf{N } \right]}^{\mathbf{2}}}{{\left[ \mathbf{H } \right]}^{\mathbf{3}}}}$. Some reactions are written below in Column I and their equilibrium constants in terms of Kc are written in Column II. Match the following reactions with the corresponding equilibrium constant-
Column I (Reaction) Column II (Equilibrium constant)
(i)$2{{\mathbf{N}}_{\mathbf{2}}}\left( \mathbf{g} \right)+6{{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)\rightleftharpoons 4\mathbf{N}{{\mathbf{H}}_{\mathbf{3}}}\left( \mathbf{g} \right)$ | (a) 2Kc |
(ii)$\mathbf{2N}{{\mathbf{H}}_{\mathbf{3}}}\left( \mathbf{g} \right)\rightleftharpoons {{\mathbf{N}}_{\mathbf{2}}}\left( \mathbf{g} \right)+\mathbf{3}{{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)$ | (b) 1 2 Kc |
(iii)$\frac{1}{2}{{\mathbf{N}}_{\mathbf{2}}}\left( \mathbf{g} \right)+\frac{3}{2}{{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)\rightleftharpoons \mathbf{N}{{\mathbf{H}}_{\mathbf{3}}}\left( \mathbf{g} \right)$ | (c) 1 Kc |
(d) Kc |
Ans: (i) $\to $ (d); (ii) $\to $ (c); (iii) $\to $ (b).
40. Match standard free energy of the reaction with the corresponding equilibrium constant
(i) $\mathbf{\Delta }{{\mathbf{G}}^{\odot }}$> 0 | (a) K > 1 |
(ii) $\mathbf{\Delta }{{\mathbf{G}}^{\odot }}$< 0 | (b) K = 1 |
(iii) $\mathbf{\Delta }{{\mathbf{G}}^{\odot }}$= 0 | (c) K = 0 |
(d) K < 1 |
Ans: (i) $\to $ (d); (ii) $\to $ (a); (iii) $\to $ (b).
41. Match the following species with the corresponding conjugate acid Species Conjugate -
$\left( \mathbf{i} \right)\mathbf{N}{{\mathbf{H}}_{\mathbf{3}}}$ | $\left( \mathbf{a} \right)\text{ }\mathbf{C}{{\mathbf{O}}_{\mathbf{3}}}^{\mathbf{2}}$ |
$\left( \mathbf{ii} \right)\text{ }\mathbf{HC}{{\mathbf{O}}_{\mathbf{3}}}^{}$ | $\left( \mathbf{b} \right)\text{ }\mathbf{N}{{\mathbf{H}}_{\mathbf{4}}}^{+}$ |
$\left( \mathbf{iii} \right){{\mathbf{H}}_{\mathbf{2}}}\mathbf{O}$ | $\left( \mathbf{c} \right)\text{ }{{\mathbf{H}}_{\mathbf{3}}}{{\mathbf{O}}^{+}}$ |
$\left( \mathbf{iv} \right)\text{ }\mathbf{HS}{{\mathbf{O}}_{\mathbf{4}}}^{}$ | $\left( \mathbf{d} \right)\text{ }{{\mathbf{H}}_{\mathbf{2}}}\mathbf{S}{{\mathbf{O}}_{\mathbf{4}}}$ |
$\left( \mathbf{e} \right)\text{ }{{\mathbf{H}}_{\mathbf{2}}}\mathbf{C}{{\mathbf{O}}_{\mathbf{3}}}$ |
Ans: (i) $\to $ (b); (ii) $\to $ (e); (iii) $\to $ (c); (iv) $\to $ (d)
42. Match the following graphical variation with their description-
A B
(i) 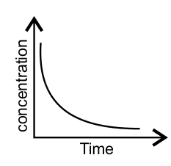
| (a) Variation in product concentration with time |
(ii) 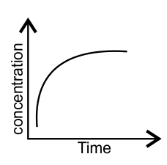
| (b) Reaction at equilibrium |
(iii) 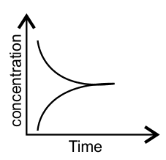
| (c) Variation in reactant concentration with time |
Ans: (i) $\to $ (c); (ii) $\to $ (a); (iii) $\to $ (b).
43. Match Column (I) with Column (II). Column I Column II-
(i) Equilibrium | (a) ∆G > 0, K < 1 |
(ii) Spontaneous reaction | (b) ∆G = 0 |
(iii) Nonspontaneous reaction | (c) $\Delta {{\mathbf{G}}^{\odot }}$= 0 |
(d) ∆G < 0, K > 1 |
Ans: (i) $\to $c; (ii) $\to $ (d); (iii) $\to $ (a).
Assertion and Reason Type
In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
44. Assertion (A): Increasing order of acidity of hydrogen halides is HF < HCl < HBr < HI
Reason (R): While comparing acids formed by the elements belonging to the same group of periodic table, H-A bond strength is a more important factor in determining acidity of an acid than the polar nature of the bond. (i) Both A and R are true and R is the correct explanation of A.
(ii) Both A and R are true but R is not the correct explanation of A.
(iii) A is true but R is false.
(iv) Both A and R are false.
Ans: As, H-A bond strength is a more important factor in determining acidity than its polar nature. As the size of A increases down the group, H-A bond strength decreases. Hence, option(i) is the correct answer.
45. Assertion (A): A solution containing a mixture of acetic acid and sodium acetate maintains a constant value of pH on addition of small amounts of acid or alkali.
Reason (R): A solution containing a mixture of acetic acid and sodium acetate acts as a buffer solution around pH 4.75.
(i) Both A and R are true and R is correct explanation of A.
(ii) Both A and R are true but R is not the correct explanation of A.
(iii) A is true but R is false.
(iv) Both A and R are false.
Ans: The solutions which resist change in pH on dilution or with the addition of small amounts of acid or alkali are called buffer solutions. Hence, option(ii) is the correct answer.
46. Assertion (A): The ionisation of hydrogen sulphide in water is low in the presence of hydrochloric acid.
Reason (R): Hydrogen sulphide is a weak acid.
(i) Both A and R are true and R is correct explanation of A.
(ii) Both A and R are true but R is not correct explanation of A.
(iii) A is true but R is false.
(iv) Both A and R are false.
Ans: In presence of ${{\text{H}}^{\text{+}}}$ ion, equilibrium shifts in backward direction. Hence, option(ii) is the correct answer.
47. Assertion (A): For any chemical reaction at a particular temperature, the equilibrium constant is fixed and is a characteristic property.
Reason (R): Equilibrium constant is independent of temperature.
(i) Both A and R are true and R is correct explanation of A.
(ii) Both A and R are true but R is not correct explanation of A.
(iii) A is true but R is false.
(iv) Both A and R are false.
Ans: The equilibrium constant for an exothermic reaction (negative H) decreases as the temperature increases. The equilibrium constant for an endothermic reaction (positive H) increases as the temperature increases. Hence, option(iii) is the correct answer.
48. Assertion (A): Aqueous solution of ammonium carbonate is basic. Reason (R): Acidic/basic nature of a salt solution of a salt of weak acid and weak base depends on ${{\mathbf{K}}_{\mathbf{a}}}$ and ${{\mathbf{K}}_{\mathbf{b}}}$ value of the acid and the base forming it. (i) Both A and R are true and R is correct explanation of A.
(ii) Both A and R are true but R is not correct explanation of A.
(iii) A is true but R is false.
(iv) Both A and R are false.
Ans: ${{\text{K}}_{\text{a}}}$ and ${{\text{K}}_{\text{b}}}$ values are responsible for acidic and basic characters of substances. Hence option (i) is the correct answer.
49. Assertion (A): An aqueous solution of ammonium acetate can act as a buffer. Reason (R): Acetic acid is a weak acid and $\mathbf{N}{{\mathbf{H}}_{\mathbf{4}}}\mathbf{OH}$ is a weak base. (i) Both A and R are true and R is correct explanation of A.
(ii) Both A and R are true but R is not correct explanation of A.
(iii) A is false but R is true.
(iv) Both A and R are false.
Ans: Salt of weak acid and weak base can form buffer solution. Hence option (iii) is the correct answer.
50. Assertion (A): In the dissociation of $\mathbf{PC}{{\mathbf{I}}_{\mathbf{5}}}$ at constant pressure and temperature addition of helium at equilibrium increases the dissociation of $\mathbf{PC}{{\mathbf{I}}_{\mathbf{5}}}$ .
Reason (R): Helium removes $\mathbf{C}{{\mathbf{I}}_{\mathbf{2}}}$ from the field of action.
(i) Both A and R are true and R is correct explanation of A.
(ii) Both A and R are true but R is not correct explanation of A.
(iii) A is true but R is false.
(iv) Both A and R are false.
Ans: If the volume is kept constant and an inert gas such as argon is added which does not take part in the reaction, the equilibrium remains undisturbed. Hence, option(iv) is the correct answer.
Long Answer Type
51. How can you predict the following stages of a reaction by comparing the value of ${{\mathbf{K}}_{\mathbf{c}}}\text{ }\mathbf{and}\text{ }{{\mathbf{Q}}_{\mathbf{c}}}$?
(i) Net reaction proceeds in the forward direction.
(ii) Net reaction proceeds in the backward direction.
(iii) No net reaction occurs.
Ans: The values of ${{\text{K}}_{\text{c}}}$ and ${{\text{Q}}_{\text{c}}}$ are itself sufficient to explain the direction of reaction and less than or greater than one another decides the direction in which reaction will proceed as follows-
(i) As ${{\text{Q}}_{\text{c}}}$ < ${{\text{K}}_{\text{c}}}$, the reaction proceeds in the forward direction.
(ii) If ${{\text{Q}}_{\text{c}}}$ > ${{\text{K}}_{\text{c}}}$, the reaction will proceed in the direction of reactants (reverse reaction).
(iii) If ${{\text{Q}}_{\text{c}}}$ = ${{\text{K}}_{\text{c}}}$ , no net reaction occurs.
52. On the basis of Le Chatelier principle explain how temperature and pressure can be adjusted to increase the yield of ammonia in the following reaction. ${{\mathbf{N}}_{\mathbf{2}}}\left( \mathbf{g} \right)\text{ }+\text{ }\mathbf{3}{{\mathbf{H}}_{\mathbf{2}}}\left( \mathbf{g} \right)\rightleftharpoons \mathbf{2N}{{\mathbf{H}}_{\mathbf{3}}}\left( \mathbf{g} \right)\text{ }\Delta \mathbf{H}\text{ }=\text{ }\text{ }\mathbf{92}.\mathbf{38}\text{ }\mathbf{kJ}\text{ }\mathbf{mo}{{\mathbf{l}}^{\mathbf{1}}}$. What will be the effect of addition of argon to the above reaction mixture at constant volume?
Ans: According to Le Chatelier’s principle, when we raise the temperature, it shifts the equilibrium to left and decreases the equilibrium concentration of ammonia since it is an exothermic reaction. In other words, low temperature and high pressure is favourable for high yield of ammonia. There will be no change in equilibria on addition of argon (Ar).
53. A sparingly soluble salt having general formula $\mathbf{A}{{\mathbf{ }}_{\mathbf{x}}}^{\mathbf{p+}}\mathbf{ }{{\mathbf{B}}_{\mathbf{y }}}^{\mathbf{q - }}$. and molar solubility S is in equilibrium with its saturated solution. Derive a relationship between the solubility and solubility product for such salt.
Ans: The equation can be written as-
\[ A_{x}^{p+} B_{y}^{q-} \rightleftarrows x A^{p+} (aq) + y B^{q-} (aq) \]
S moles of A and B dissolves to give x S moles of Ap+ and y Bq-.
\[ K_{sp} = [A^{p+}][Bq^-]^{y} = [x^{5}]^x [y^5]^y \]
\[= x^x y^y 5^{x+y} \]
54. Write a relation between ∆G and Q and define the meaning of each term and answer the following:
(a) Why a reaction proceeds forward when Q < K and no net reaction occurs when Q = K.
Ans: (a) As we know,
ΔG= $\text{ }\!\!\Delta\!\!\text{ }{{\text{G}}^{\odot }}$+RTlnQ
$\text{ }\!\!\Delta\!\!\text{ }{{\text{G}}^{\odot }}$= Change in free energy as the reaction proceeds,
ΔG = Standard free energy change,
Q = Reaction quotient,
R = Gas constant,
T = Absolute temperature.
Since, $\text{ }\!\!\Delta\!\!\text{ }{{\text{G}}^{\odot }}$ =−RTlnK
∴ΔG=−RTlnK+RTlnQ=RTlnKQ
If Q<K, ΔG will be negative. Reaction proceeds in the forward.
If Q=K, ΔG=0, reaction is in equilibrium and no net reaction is there.
(b) Explain the effect of increase in pressure in terms of reaction quotient Q. for the reaction: $\mathbf{CO}\text{ }\left( \mathbf{g} \right)\text{ }+\mathbf{3}{{\mathbf{H}}_{\mathbf{2}}}\text{ }\left( \mathbf{g} \right)\to \mathbf{C}{{\mathbf{H}}_{\mathbf{4}}}\text{ }\left( \mathbf{g} \right)+{{\mathbf{H}}_{\mathbf{2}}}\mathbf{O}\text{ }(\mathbf{g})$.
Ans: When we increase the pressure equilibrium will shift in forward direction it means Q<K.
Types of Chemical Reactions :
There are 4 main types of chemical reactions. These are-
1. Combination Reactions- Combination Reactions are chemical reactions with 2 or more substances combining to form one single compound.
2. Decomposition Reactions- Decomposition Reactions are chemical reactions with one compound decomposing to produce 2 or more different substances. The digestion process of food in our body is a Decomposition reaction.
3. Displacement Reactions- Displacement reactions are reactions that involve the displacement of 1 element or group by another.
4. Double Displacement- Displacement reactions are reactions with 2 compounds that react to form 2 new compounds. In the displacement reaction, no change in oxidation state occurs.
FAQs on Free PDF Download of NCERT Exemplar for Class 11 Chemistry Chapter 7 - Equilibrium
1. What are the characteristics of Chemical Equilibrium?
The Characteristics of Chemical Equilibrium are-
Chemical Equilibrium can be obtained from any side of the reaction.
Chemical Equilibrium is always dynamic in nature. That means at an Equilibrium state, the reaction does not stop.
At Chemical Equilibrium, there is no more change in the concentration of both reactants and products.
The Equilibrium state always remains unaffected by the presence of a catalyst. Catalyst helps to reach the Equilibrium state with more pace.
In Chemical Equilibrium observable physical properties of the process stay constant.
2. Why is Chemical Equilibrium also called Dynamic Equilibrium?
Chemical Equilibrium is the stage of a reaction in which the rate of the forward reaction is equal to the rate of backward reaction. This stage signifies that the number of reactant molecules turning into products and product molecules into reactants is the same. A similar Equilibrium can be carried out with the same reactants anywhere by achieving the same conditions with continuous interchanging of molecules. This is why Chemical Equilibrium or Equilibrium of chemical reactions is also called Dynamic Equilibrium.
3. What are the two types of Equilibrium?
Equilibrium is a state in which all the acting influences are cancelled by each other making the system stable. There are 2 types of Equilibrium
Physical Equilibrium
Physical Equilibrium in a physical process represents the existence of the same substance in 2 different physical states at a time. There is no change in chemical composition during Physical Equilibrium.
Chemical Equilibrium
Chemical Equilibrium is the state of a system in which the concentration of the products and the concentration of the reactants don't change with time.
4. Where can a Class 11 student study Chapter Equilibrium?
Equilibrium is an important Chapter of Class 11 Chemistry as its basics are used in many topics of Chemistry. A student can study Class 11 Equilibrium topics from Vedantu’s NCERT Exemplar for Equilibrium. They can also go through Vedantu’s Youtube Channel to understand the basics well. Class 11 students can then solve multiple previous year questions that will prepare them for real test situations and help them understand the pattern of questions. This is how a Class 11 student should study the Equilibrium Chapters and other Chapters of Chemistry for best results.
5. What is an Equilibrium state?
An Equilibrium state is a state of chemical reaction that it reaches under a certain set of conditions. If a reversible chemical reaction is carried out in a closed container, a constancy in the observable properties like pressure, colour intensity, density, is attained. This state of the chemical reaction is called the Equilibrium state. Students can study more about chemical reactions and the Equilibrium state of chemical reactions at Vedantu’s official site. They can also find other resources that they can use for free.

























