Write IUPAC name and chemical formula of acetone.
Answer
632.4k+ views
Hint: The prefix suggests the presence of two carbon atoms. The suffix –one is used for showing the presence of ketone functional groups in the organic compound. Ketones have two alkyl groups bonded to the carbonyl carbon.
Complete step by step solution:
Let’s see first which functional group is present in acetone and how its structure would look like.
- There is only one suffix present in the name which indicates the presence of a related functional group. The suffix –one suggests the presence of ketone functional groups.
- We know that ketone functional group can be shown as
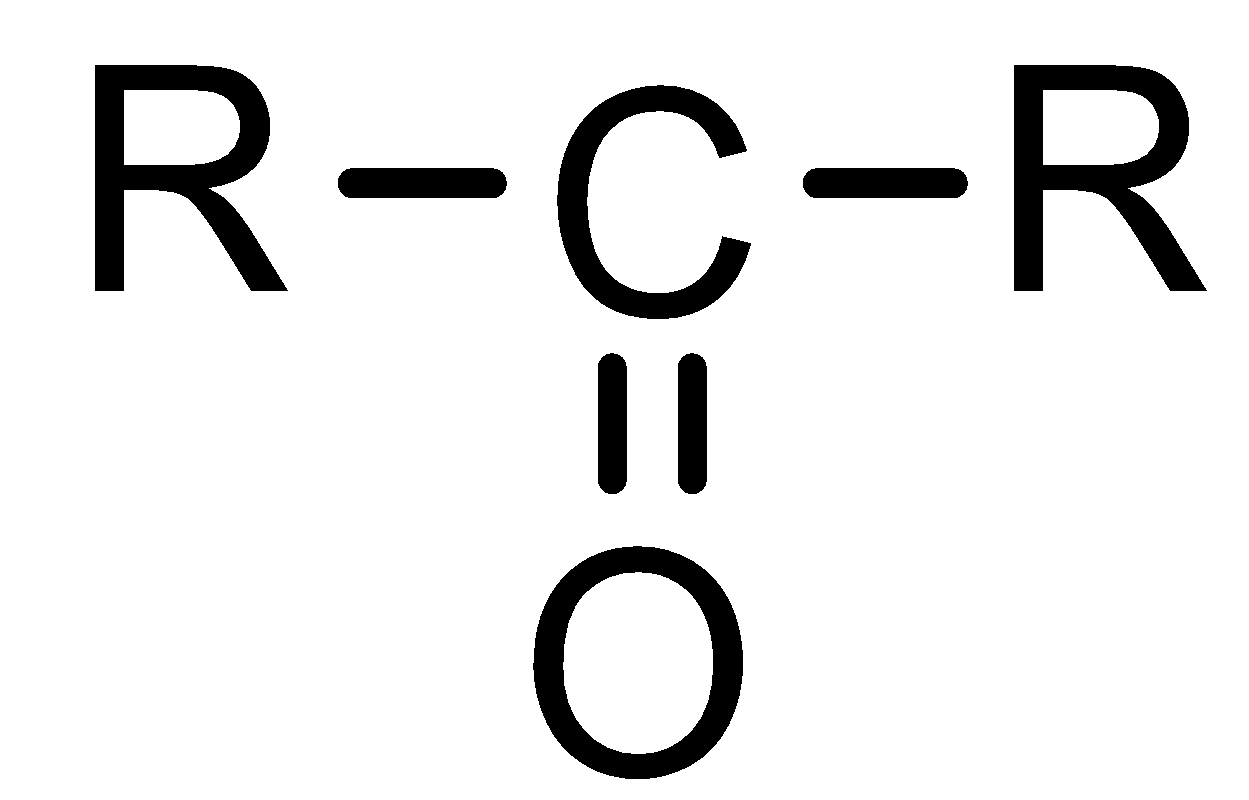
- Now ‘aceto’ prefix suggests the presence of two carbon atoms. That means one of the alkyl groups should be a methyl group because one carbonyl carbon is already there. So, which alkyl group will be there on the other side of the carbonyl carbon?
- As it is necessary that two alkyl groups need to be bonded with carbonyl carbon in ketones, the other alkyl group will be the smallest possible alkyl group and that is methyl group. So, both alkyl groups will be methyl groups.
Chemical Formula: \[{H_3}C - CO - C{H_3}\]
Now, we will assign IUPAC names to that compound.
- First we will need to identify the functional group present in the compound and that is only the ketone group because of \[ - CO - \].
- When ketones are given IUPAC names, we need to identify the longest carbon chain first. We can see that all three carbons are in line and will form a chain of three carbon atoms.
- As three carbon atoms are present in the chain, we will call it Propane and we will need to use the suffix –one as ketone is present. So, we can write its name as Propanone.
- Still it does not show the position of the ketone carbon. If we give numbering, ketone carbon will be carbon number-2. So, we can write the IUPAC name of Acetone as 2-Propanone or Propan-2-one.
IUPAC name: 2-Propanone or Propan-2-one.
Note: As ‘aceto’ prefix suggests the presence of two carbon atoms only, do not consider that acetone has only two carbon atoms in its structure, actually acetone is the smallest member of the ketone family. Do not forget to put the number at which the carbonyl group is present while naming the ketone compound.
Complete step by step solution:
Let’s see first which functional group is present in acetone and how its structure would look like.
- There is only one suffix present in the name which indicates the presence of a related functional group. The suffix –one suggests the presence of ketone functional groups.
- We know that ketone functional group can be shown as

- Now ‘aceto’ prefix suggests the presence of two carbon atoms. That means one of the alkyl groups should be a methyl group because one carbonyl carbon is already there. So, which alkyl group will be there on the other side of the carbonyl carbon?
- As it is necessary that two alkyl groups need to be bonded with carbonyl carbon in ketones, the other alkyl group will be the smallest possible alkyl group and that is methyl group. So, both alkyl groups will be methyl groups.
Chemical Formula: \[{H_3}C - CO - C{H_3}\]
Now, we will assign IUPAC names to that compound.
- First we will need to identify the functional group present in the compound and that is only the ketone group because of \[ - CO - \].
- When ketones are given IUPAC names, we need to identify the longest carbon chain first. We can see that all three carbons are in line and will form a chain of three carbon atoms.
- As three carbon atoms are present in the chain, we will call it Propane and we will need to use the suffix –one as ketone is present. So, we can write its name as Propanone.
- Still it does not show the position of the ketone carbon. If we give numbering, ketone carbon will be carbon number-2. So, we can write the IUPAC name of Acetone as 2-Propanone or Propan-2-one.
IUPAC name: 2-Propanone or Propan-2-one.
Note: As ‘aceto’ prefix suggests the presence of two carbon atoms only, do not consider that acetone has only two carbon atoms in its structure, actually acetone is the smallest member of the ketone family. Do not forget to put the number at which the carbonyl group is present while naming the ketone compound.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




