
The compounds $[\text{Co(S}{{\text{O}}_{4}}){{(\text{N}{{\text{H}}_{3}})}_{5}}]\text{Br}$ and $[\text{Co(S}{{\text{O}}_{4}}){{(\text{N}{{\text{H}}_{3}})}_{5}}]\text{Cl}$ represent:
(A) linkage isomerism
(B) ionisation isomerism
(C) coordination isomerism
(D) no isomerism
Answer
599.7k+ views
Hint: Recall the types of isomerism given in the chapter Coordination compounds. Try to understand the types of isomerism given in the options with an example so as to check whether the compound given in question is that type of isomer or not. If the pair of complex compounds are not any type of isomers then there is no isomerism between the two compounds.
Complete step by step answer:
Chemical compounds that consist of an array of anions or neutral molecules that are bound to a central atom via coordinate covalent bonds are called coordination compounds. Coordination compounds are also referred to as coordination complexes.
The neutral molecules or ions that are bound to the central atom are referred to as ligands or complexing agents. They act as Lewis base and donate electron pairs to transition metal ions, thus a dative bond is formed between ligands and the transition metal ion. Hence these compounds are coordination complexes.
Linkage isomerism is the existence of coordination compounds that have the same composition but differ in their connectivity with the central metal atom. Example:
- Thiocyanate ($SC{{N}^{-}}$) and isothiocyanate ($NC{{S}^{-}}$),
- Selenocyanate ($SeC{{N}^{-}}$) and iso selenocyanate ($NCS{{e}^{-}}$).
Ionization isomers have the same composition and formula but give different ions upon dissociation. Example:
- \[[Co{{(N{{H}_{3}})}_{5}}Br]S{{O}_{4}}\]and \[[Co{{(N{{H}_{3}})}_{5}}S{{O}_{4}}]Br\]
Coordination isomerism is a form of structural isomerism in which the composition of complex ion is different in the pair of isomers. In a coordination isomer the total ratio of ligand to metal remains the same, but the ligands attached to a specific metal ion are different. Example:
- ${{[Zn{{(N{{H}_{3}})}_{4}}]}^{+2}}{{[CuC{{l}_{4}}]}^{-2}}$ and ${{[Cu{{(N{{H}_{3}})}_{4}}]}^{+2}}{{[ZnC{{l}_{4}}]}^{-2}}$.
From the above explanation we see that the given pair of complex compounds do not match with any of them examples mentioned. Hence there is no isomerism between the two complex compounds.
So, the correct answer is “Option D”.
Note: Like organic compounds, transition metal complexes show geometrical and optical isomers. For e.g. the below pair of complexes are in cis-trans form:
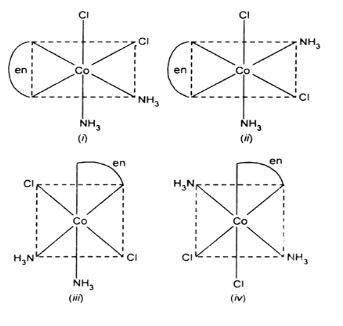
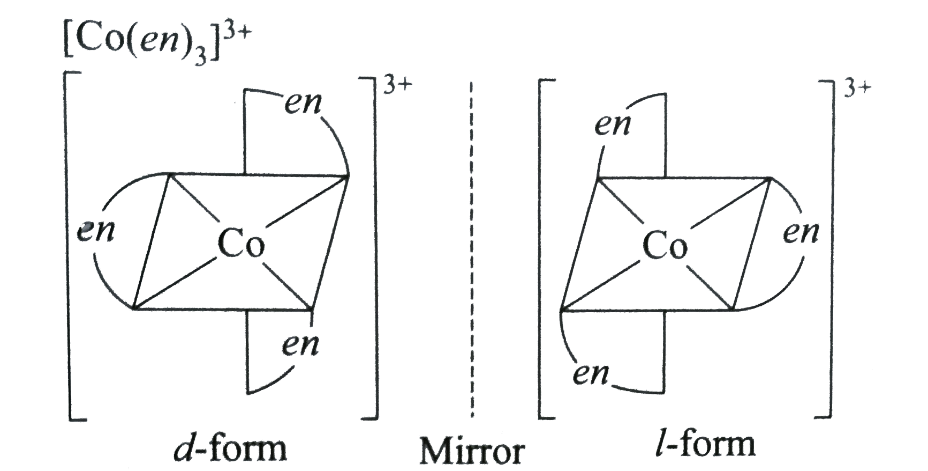
The complex ${{[Co{{(en)}_{3}}]}^{+3}}$ shows optical isomerism:
Complete step by step answer:
Chemical compounds that consist of an array of anions or neutral molecules that are bound to a central atom via coordinate covalent bonds are called coordination compounds. Coordination compounds are also referred to as coordination complexes.
The neutral molecules or ions that are bound to the central atom are referred to as ligands or complexing agents. They act as Lewis base and donate electron pairs to transition metal ions, thus a dative bond is formed between ligands and the transition metal ion. Hence these compounds are coordination complexes.
Linkage isomerism is the existence of coordination compounds that have the same composition but differ in their connectivity with the central metal atom. Example:
- Thiocyanate ($SC{{N}^{-}}$) and isothiocyanate ($NC{{S}^{-}}$),
- Selenocyanate ($SeC{{N}^{-}}$) and iso selenocyanate ($NCS{{e}^{-}}$).
Ionization isomers have the same composition and formula but give different ions upon dissociation. Example:
- \[[Co{{(N{{H}_{3}})}_{5}}Br]S{{O}_{4}}\]and \[[Co{{(N{{H}_{3}})}_{5}}S{{O}_{4}}]Br\]
Coordination isomerism is a form of structural isomerism in which the composition of complex ion is different in the pair of isomers. In a coordination isomer the total ratio of ligand to metal remains the same, but the ligands attached to a specific metal ion are different. Example:
- ${{[Zn{{(N{{H}_{3}})}_{4}}]}^{+2}}{{[CuC{{l}_{4}}]}^{-2}}$ and ${{[Cu{{(N{{H}_{3}})}_{4}}]}^{+2}}{{[ZnC{{l}_{4}}]}^{-2}}$.
From the above explanation we see that the given pair of complex compounds do not match with any of them examples mentioned. Hence there is no isomerism between the two complex compounds.
So, the correct answer is “Option D”.
Note: Like organic compounds, transition metal complexes show geometrical and optical isomers. For e.g. the below pair of complexes are in cis-trans form:

The complex ${{[Co{{(en)}_{3}}]}^{+3}}$ shows optical isomerism:

Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

What steps did the French revolutionaries take to create class 11 social science CBSE

Which among the following are examples of coming together class 11 social science CBSE




