What is the oxidation state of Chromium (Cr) in ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$.
Answer
626.1k+ views
Hint: Oxidation state is the number of electrons that an atom needs to lose or gain to result in a chemical bond. To know the oxidation state of ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$, first we have to draw its structure so that we identify the type of oxygen atom.
Complete step-by-step answer:
The structure of ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$is,
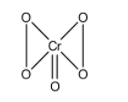
In the compounds, four oxygen atoms are part of peroxide linkage and we know that in peroxides the oxidation state of oxygen is -1. And in all other compounds, the oxidation state of oxygen atom is -2.
If we know the type of compound, we will easily know the oxidation number of compounds. For neutral compounds, such as ${{\rm{H}}_{\rm{2}}}{\rm{O}}$, ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$, etc. the addition of oxidation state of all atoms of the compound is equal to zero.
So, in the compound, ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$, four peroxide oxygen atoms has oxidation state of -1 each and the remaining oxygen has oxidation state of -2. Using these values, now we calculate the oxidation state of chromium.
We take x as the oxidation state of chromium in ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$.
$\begin{array}{c}x + \left( { - 1 \times 4} \right) + \left( { - 2} \right) = 0\\x - 4 - 2 = 0\\x = 6\end{array}$
Hence, the oxidation state of chromium in ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$ is 6.
Additional Information:
1) If a compound exists in elemental form (only one type of atoms present), the oxidation number of the element is always zero.
2) For ions, the charge indicates the oxidation number. For example, the oxidation number of chloride ions is -1.
Note: Students have to take care of taking the oxidation number of oxygen in the calculation of an oxidation number of an element in a compound. In peroxides, oxygen possesses oxidation state of -1. In ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$ four oxygen atoms has -1 oxidation state and the remaining one oxygen atom possesses oxidation state of -2.
Complete step-by-step answer:
The structure of ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$is,

In the compounds, four oxygen atoms are part of peroxide linkage and we know that in peroxides the oxidation state of oxygen is -1. And in all other compounds, the oxidation state of oxygen atom is -2.
If we know the type of compound, we will easily know the oxidation number of compounds. For neutral compounds, such as ${{\rm{H}}_{\rm{2}}}{\rm{O}}$, ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$, etc. the addition of oxidation state of all atoms of the compound is equal to zero.
So, in the compound, ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$, four peroxide oxygen atoms has oxidation state of -1 each and the remaining oxygen has oxidation state of -2. Using these values, now we calculate the oxidation state of chromium.
We take x as the oxidation state of chromium in ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$.
$\begin{array}{c}x + \left( { - 1 \times 4} \right) + \left( { - 2} \right) = 0\\x - 4 - 2 = 0\\x = 6\end{array}$
Hence, the oxidation state of chromium in ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$ is 6.
Additional Information:
1) If a compound exists in elemental form (only one type of atoms present), the oxidation number of the element is always zero.
2) For ions, the charge indicates the oxidation number. For example, the oxidation number of chloride ions is -1.
Note: Students have to take care of taking the oxidation number of oxygen in the calculation of an oxidation number of an element in a compound. In peroxides, oxygen possesses oxidation state of -1. In ${\rm{Cr}}{{\rm{O}}_{\rm{5}}}$ four oxygen atoms has -1 oxidation state and the remaining one oxygen atom possesses oxidation state of -2.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE




