Name the source from which litmus solution is obtained. What is the use of this solution ?
Answer
616.2k+ views
Hint: We know that litmus is obtained from a mixture of dyes. Try to figure out the source of these dyes. We know that litmus is used in many chemical reactions. Chemical reactions involving acids and bases use this litmus for identifying equivalence points.
Complete step by step answer:
We know that litmus is an organic compound prepared from a mixture of dyes which are extracted from lichens, cellulose compounds, and some other organic compounds. Impurities are removed from the litmus solution to prepare litmus paper. Litmus preparation requires several varieties of lichens. Cellulose treated with some solvents is used in the preparation of litmus paper. Litmus paper preparation is almost similar to the normal preparation but this litmus paper is infused with lichens.
We use litmus paper to test whether a substance is acidic or basic or neutral. For testing solutions whether it is acid or base, we need two colors of litmus papers. Blue litmus turns to red in the presence of acidic solution. Red litmus turns to blue in the presence of a basic solution. Both litmuses turn to purple in the presence of a neutral solution. Litmus has 2 different structures in acidic and basic solutions.
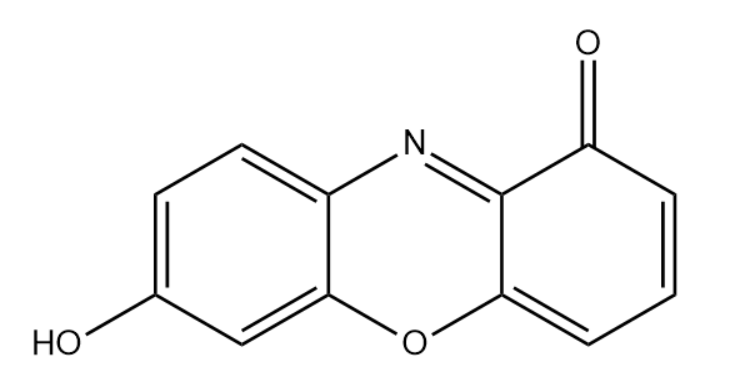
Note: Litmus paper test is quick but we cannot determine the pH of the solution by this test. We can reuse litmus paper. Litmus paper changes color only when the pH of the solution is in a given range. Otherwise, the nature of the solution cannot be determined.
Complete step by step answer:
We know that litmus is an organic compound prepared from a mixture of dyes which are extracted from lichens, cellulose compounds, and some other organic compounds. Impurities are removed from the litmus solution to prepare litmus paper. Litmus preparation requires several varieties of lichens. Cellulose treated with some solvents is used in the preparation of litmus paper. Litmus paper preparation is almost similar to the normal preparation but this litmus paper is infused with lichens.
We use litmus paper to test whether a substance is acidic or basic or neutral. For testing solutions whether it is acid or base, we need two colors of litmus papers. Blue litmus turns to red in the presence of acidic solution. Red litmus turns to blue in the presence of a basic solution. Both litmuses turn to purple in the presence of a neutral solution. Litmus has 2 different structures in acidic and basic solutions.

Note: Litmus paper test is quick but we cannot determine the pH of the solution by this test. We can reuse litmus paper. Litmus paper changes color only when the pH of the solution is in a given range. Otherwise, the nature of the solution cannot be determined.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




