Molecular formula of oxygen is ${{O}_{2}}$ while sulphur is ${{S}_{8}}$, why ?
Answer
624.9k+ views
Hint: Write the electronic configuration of oxygen and sulphur. Think about what happens to atomic size when you go down the group. Also, think about physical states of oxygen and sulphur.
Complete step by step answer:
- Oxygen and sulphur belongs to the Group 16 family of chalcogens. Oxygen has the electronic configuration as follows,
${}^{8}O=1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}$
- Oxygen is a gas while all other chalcogens are solids at room temperature.
- Oxygen has the potential to form a $p\pi -p\pi $ bond. This $p\pi -p\pi $ overlapping between two oxygen atoms leads to the formation of a diatomic stable ${{O}_{2}}$ molecule having a linear structure (O=O). Moreover, these ${{O}_{2}}$ molecules have only weak Van der Waals’ forces of interaction between them and so it exists in a gaseous state.
- Now, let’s have a look at sulphur.
The electronic configuration of sulphur is,
\[{{S}_{2}}=1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{4}}\]
- As we move down the group, atomic size increases due to addition of a greater number of orbitals. Due to the large size of sulphur and also the presence of vacant 3d orbitals, sulphur is not able to form $p\pi -p\pi $ bonds.
- The p-orbitals of adjacent sulphur overlap axially only giving rise to $\text{ }\!\!\sigma\!\!\text{ -bonds}$. But one atom of sulphur needs to form at least two such bonds to attain noble gas configuration. Similarly, each sulphur needs to form a stable configuration. This can only be achieved by formation of a cyclic ring.
So, eight sulphur atoms join together to form a polyatomic crown shaped puckered ring.
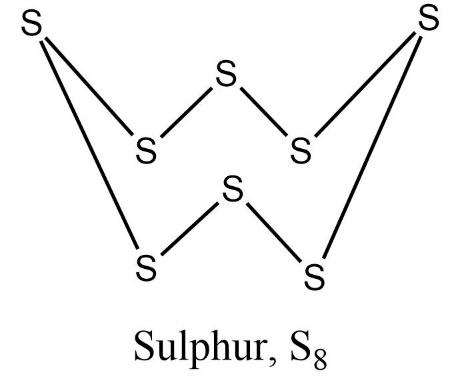
Therefore, we can justify that, the molecular formula of oxygen is ${{O}_{2}}$ while sulphur is ${{S}_{8}}$.
Note: Like sulphur, selenium also forms a puckered ring structure. Also, polyatomic molecules have strong Van der Waals’ forces of interaction between them and so they exist as solids at room temperature. Remember atomic radii increases down the group and decreases as we move from left to right in the periodic table.
Complete step by step answer:
- Oxygen and sulphur belongs to the Group 16 family of chalcogens. Oxygen has the electronic configuration as follows,
${}^{8}O=1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}$
- Oxygen is a gas while all other chalcogens are solids at room temperature.
- Oxygen has the potential to form a $p\pi -p\pi $ bond. This $p\pi -p\pi $ overlapping between two oxygen atoms leads to the formation of a diatomic stable ${{O}_{2}}$ molecule having a linear structure (O=O). Moreover, these ${{O}_{2}}$ molecules have only weak Van der Waals’ forces of interaction between them and so it exists in a gaseous state.
- Now, let’s have a look at sulphur.
The electronic configuration of sulphur is,
\[{{S}_{2}}=1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{4}}\]
- As we move down the group, atomic size increases due to addition of a greater number of orbitals. Due to the large size of sulphur and also the presence of vacant 3d orbitals, sulphur is not able to form $p\pi -p\pi $ bonds.
- The p-orbitals of adjacent sulphur overlap axially only giving rise to $\text{ }\!\!\sigma\!\!\text{ -bonds}$. But one atom of sulphur needs to form at least two such bonds to attain noble gas configuration. Similarly, each sulphur needs to form a stable configuration. This can only be achieved by formation of a cyclic ring.
So, eight sulphur atoms join together to form a polyatomic crown shaped puckered ring.

Therefore, we can justify that, the molecular formula of oxygen is ${{O}_{2}}$ while sulphur is ${{S}_{8}}$.
Note: Like sulphur, selenium also forms a puckered ring structure. Also, polyatomic molecules have strong Van der Waals’ forces of interaction between them and so they exist as solids at room temperature. Remember atomic radii increases down the group and decreases as we move from left to right in the periodic table.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




