Explain the formation of a chemical bond.
Answer
628.2k+ views
Hint: A chemical bond is formed as a result of forces of attraction due to electrostatic forces. This brings the reacting species together and helps their orbital with equal energies and proper orientation to overlap and form molecular orbitals.
Complete answer:
A chemical bond is a formation of bond due to the electrostatic forces of attraction due to sharing of electrons or donating electrons. The attractive force which holds up the constituent particles (atoms, ions or molecules) together in a chemical species is known as chemical bond.
Atoms either share or gain or lose electrons to attain stable electronic configuration. Due to this, a state of minimum energy is obtained and a chemical bond is formed. This results in maximum stability. When two atoms share electrons, a covalent bond is formed and when atoms lose or gain electrons, ionic bond is formed.
Chemical bond are of three type which are as follows:
Ionic bonds, covalent bonds and metallic bonds.
Ionic bonds:
Ionic bond is formed when a metal cation is bonded to anion of a non-metal that is metal donates electron to anion and in this case both cation and anion complete their octet.
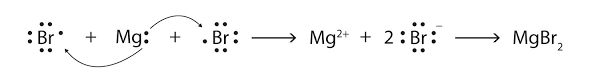
Covalent bond:
A covalent bond occurs when electrons are shared between the two or more bonding atoms to complete - an octet.
Example given:

Electrons are shared as per the requirement of each atom involved.
Note: There are exceptions to octet rule. For example in the case of Hydrogen and helium a duet configuration is stable.
Complete answer:
A chemical bond is a formation of bond due to the electrostatic forces of attraction due to sharing of electrons or donating electrons. The attractive force which holds up the constituent particles (atoms, ions or molecules) together in a chemical species is known as chemical bond.
Atoms either share or gain or lose electrons to attain stable electronic configuration. Due to this, a state of minimum energy is obtained and a chemical bond is formed. This results in maximum stability. When two atoms share electrons, a covalent bond is formed and when atoms lose or gain electrons, ionic bond is formed.
Chemical bond are of three type which are as follows:
Ionic bonds, covalent bonds and metallic bonds.
Ionic bonds:
Ionic bond is formed when a metal cation is bonded to anion of a non-metal that is metal donates electron to anion and in this case both cation and anion complete their octet.
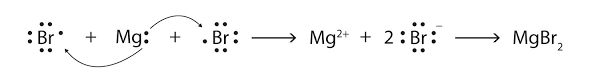
Covalent bond:
A covalent bond occurs when electrons are shared between the two or more bonding atoms to complete - an octet.
Example given:

Electrons are shared as per the requirement of each atom involved.
Note: There are exceptions to octet rule. For example in the case of Hydrogen and helium a duet configuration is stable.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is the full form of CNG A Complete Natural Gas class 10 social science CBSE

In cricket, what is a "Yorker" designed to do?

What is the full form of POSCO class 10 social science CBSE

Define Potential, Developed, Stock and Reserved resources




