Explain how Hope’s apparatus can be used to demonstrate the anomalous behavior of water?
Answer
560.9k+ views
Hint: Hope’s apparatus is a vertical vessel containing water surrounded around the middle with a trough which is filled with ice. Two thermometers are placed above and below the trough to measure the temperature of the water as shown below:
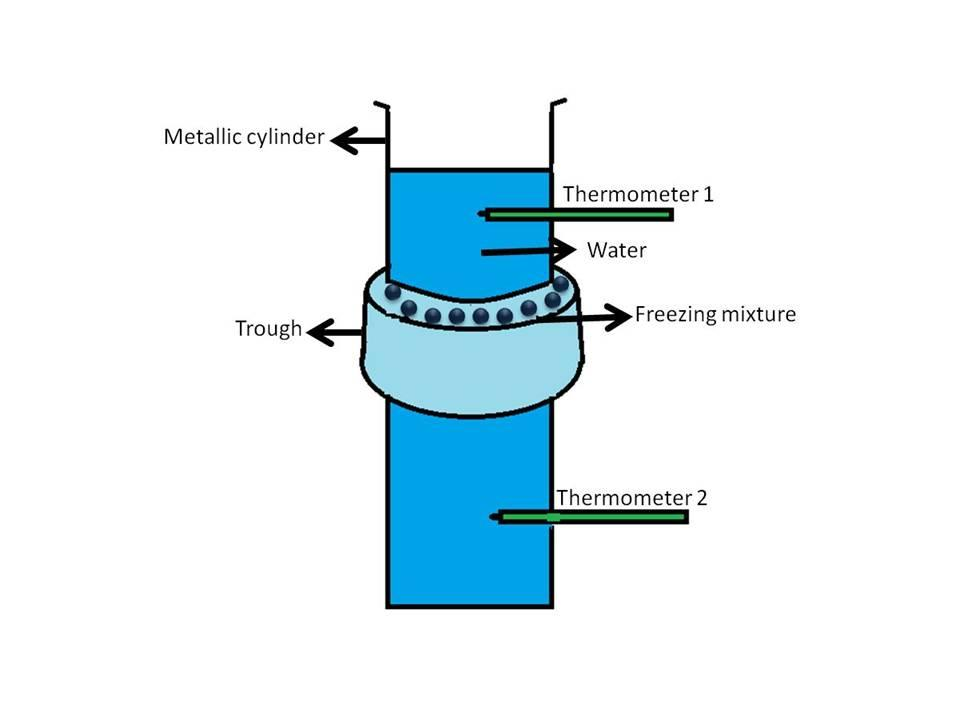
Complete answer:We all know that the density of solids is greater than liquids so it is expected that when a liquid becomes solid its density increases and it contracts. But water on cooling shows contraction till $4^oC$ and after that expands and converts into ice which has a lower density than water (clear from the fact that ice floats on water). This anomalous (different) behavior of water can be demonstrated by the use of an instrument called Hope’s apparatus. In this experiment the temperature of the water-filled in a cylinder vessel is varied. It is conducted in two sets:
$1.$ In the first set, the water is maintained at a temperature higher than $4^oC$, and the cylindrical vessel is filled with this water. The reading of the two thermometers is noted (it is observed to be the same in the beginning). Then freezing mixture containing ice and salt id added to the trough and after some time (say $30$ minutes) temperature is noted. In this case, the temperature of the water in the lower part of the vessel below the trough is found to be less as compared to the upper part. It can be explained by keeping in mind that the addition of freezing mixture decreased the temperature of the water and due to an increase in density (above $4^oC$) the water at lower temperature sinks down resulting in a lowering of the temperature of the thermometer $2$ as compared to the temperature of thermometer $1$.
$2.$ In the second set, the water is maintained to a temperature near to $4^oC$ and filled in the cylinder. After the addition of the freezing mixture, the temperature of water drops below $4^oC$ and it begins to expand thus comes to the upper part (remember, ice floats on water). Thus, the temperature of the upper part of the vessel decreases resulting in a lowering of the temperature of the thermometer $1$ as compared to the temperature of the thermometer $2$.
Note:
During the experiment, the temperature of the water should vary between $0^oC$ to higher than $4^oC$ otherwise the change will not be observed. Both water to be filled in the vessel and the apparatus itself is maintained at the same temperature.

Complete answer:We all know that the density of solids is greater than liquids so it is expected that when a liquid becomes solid its density increases and it contracts. But water on cooling shows contraction till $4^oC$ and after that expands and converts into ice which has a lower density than water (clear from the fact that ice floats on water). This anomalous (different) behavior of water can be demonstrated by the use of an instrument called Hope’s apparatus. In this experiment the temperature of the water-filled in a cylinder vessel is varied. It is conducted in two sets:
$1.$ In the first set, the water is maintained at a temperature higher than $4^oC$, and the cylindrical vessel is filled with this water. The reading of the two thermometers is noted (it is observed to be the same in the beginning). Then freezing mixture containing ice and salt id added to the trough and after some time (say $30$ minutes) temperature is noted. In this case, the temperature of the water in the lower part of the vessel below the trough is found to be less as compared to the upper part. It can be explained by keeping in mind that the addition of freezing mixture decreased the temperature of the water and due to an increase in density (above $4^oC$) the water at lower temperature sinks down resulting in a lowering of the temperature of the thermometer $2$ as compared to the temperature of thermometer $1$.
$2.$ In the second set, the water is maintained to a temperature near to $4^oC$ and filled in the cylinder. After the addition of the freezing mixture, the temperature of water drops below $4^oC$ and it begins to expand thus comes to the upper part (remember, ice floats on water). Thus, the temperature of the upper part of the vessel decreases resulting in a lowering of the temperature of the thermometer $1$ as compared to the temperature of the thermometer $2$.
Note:
During the experiment, the temperature of the water should vary between $0^oC$ to higher than $4^oC$ otherwise the change will not be observed. Both water to be filled in the vessel and the apparatus itself is maintained at the same temperature.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Find the value of the expression given below sin 30circ class 11 maths CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Difference between physical and chemical change class 11 chemistry CBSE

Which of the following is the hottest month AMay BJune class 11 social science CBSE

What is the percentage of photosynthetically active class 11 biology CBSE




