



What is Matter?
If you look around yourself, you will see many different objects such as pencils, books, boys, girls, desks, doors, etc. All these things are called matter. Matter is made up of many substances. Matter is defined as anything that has weight and volume and occupies space. Air, sand, water are some matter examples. Different types of matter can be described by their weight or volume. Weight is just how heavy something or an object is. For example- a boy lifting a school bag.

Volume is something that occupies space. For example- a ball takes up a small volume while the sun occupies a large volume.

States of Matter
Matter is made of particles. It exists in three states and these are solid, liquid and gas. There are three states of matter that are caused by the different arrangements of matter particles.
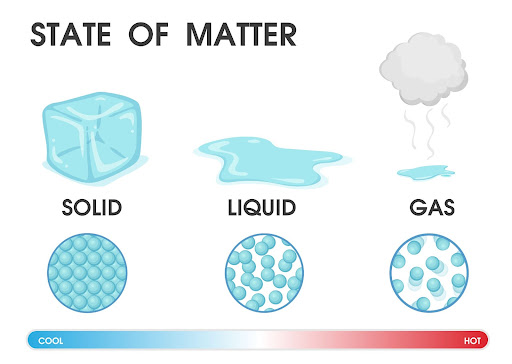
Solid State
In solid state, the particles are very tightly packed. Therefore, the particles hold together very tightly, that is, there is a strong force of attraction between them. As a result, solids consist of definite shape and volume. They have very little space for particles to move.

Examples of Solid State
(image will be uploaded soon)
Liquid State
The particles in the liquid state are not so closely packed, as in solids. Thus, it results in a lesser force of attraction among the particles than in solids.

Therefore, liquids have a specific volume, but it does not have a specific shape. They take the form of a shape in which they are kept.
For example- Milk takes the shape of glass when it is poured into it. If you drop milk on the floor, it will quickly spread because it will adopt the shape of the floor.

Examples of Liquid State

Examples of liquid state - milk and water
Gaseous State
In gases, the particles of matter are very far away from each other and so the force of attraction between them is very weak.

As a result, gases have neither a definite shape nor a definite volume. They can fill an entire space or volume of a container regardless of the size of the container.
Examples of Gaseous State are air and steam

Difference Between Solid, Liquid, and Gas
The difference between solid, liquid, and gas is given below
What is Material?
Objects are made of materials, which are the matter or substance from which they are produced. Every day, we work with a variety of materials, these might include:
Metal
Plastic
Wood
Glass

Each material has a large range of applications. For example, wood may be used to construct pencils, tables, doors, floors, and a variety of other items. A chair, for example, can be built of metal, wood, or plastic, or it might be composed of a combination of materials.
Four Properties of Materials
Different materials have different characteristics and behaviours. These are called properties. Four properties of a material are:
1. Hardness: Hardness is defined as the property in which an object can withstand scratches and force to some extent. Example- wood.
2. Malleability: Malleability is defined as the property of a material in which an object can be drawn into thin sheets. Example- Silver
3. Ductility: Ductility is defined as the property of a material in which an object can be drawn into thin wires. Example- Gold
4. Good Conductors of Heat - The materials that allow heat to pass through them are called good heat conductors. Example- Gold, Silver, Iron, etc.
Solved Questions
1. Why do solids have a definite shape and volume?
Ans. Solids have a definite shape and volume due to the close packing and fixed positions of their molecules. The molecules vibrate but do not move, maintaining the structure and volume of the object.
2. Differentiate between matter and materials.
Ans. The difference between matter and materials is as follows.
Fun Facts
1. Only water can exist in three forms naturally.

2. The states of matter are interchangeable i.e solid, liquid and gases are interconvertible.
3. Gases are invisible and take on the shape and volume of the container in which they are contained.
Summary
Matter is defined as anything that has weight and volume and occupies space. The various states of matter are caused by the different arrangements of matter particles. In a solid state, the particles of the object are very close together. The particles in the liquid state are packed densely together, yet they are slightly far spread than the particles in solids. In gases, particles are far away from each other.
Objects are made of materials, which are the matter or substance from which they are produced. The four properties of material are hardness, malleability, ductility and good conductors of heat and electricity.
Learning By Doing
Match the following
FAQs on Matter and Materials
1. What is matter?
Anything that has mass (weight) and takes up space (volume) is called matter. Everything around us, like your book, the water you drink, and even the air you breathe, is a form of matter.
2. What are the three common states of matter?
The three most common states of matter found on Earth are:
- Solid: Has a fixed shape and volume (e.g., a rock, a pencil).
- Liquid: Has a fixed volume but takes the shape of its container (e.g., water, milk).
- Gas: Has no fixed shape or volume; it spreads out to fill any container completely (e.g., air, steam).
3. Why can liquids flow but solids cannot?
This difference is because of how their tiny particles, called molecules, are arranged. In solids, particles are packed very tightly together and cannot move much, which gives them a fixed shape. In liquids, the particles are close but can slide past one another, allowing the liquid to flow and take the shape of its container.
4. What happens to matter when it changes state, for example, when water boils?
When matter changes its state, it is not lost or destroyed. For instance, when you boil water (a liquid), it turns into steam or water vapour (a gas). The water may seem to disappear, but it has only changed its form. The amount of matter remains the same. This is an example of a physical change.
5. What is all matter made of?
All matter is made up of extremely tiny particles called atoms. These atoms are so small that we cannot see them with our naked eyes. Atoms are the basic building blocks of everything, and they combine in different ways to form all the materials we see around us.
6. Can you give some examples of solids, liquids, and gases from our daily life?
Certainly! Here are some common examples:
- Solids: Ice, wood, stone, your chair, and your book.
- Liquids: Water, juice, oil, and milk.
- Gases: The air we breathe (which is a mix of gases like oxygen and nitrogen), steam from a kettle, and the helium inside a balloon.
7. How can we change a solid into a liquid or a liquid into a gas?
We can change the state of matter by adding or removing heat. For example, if you heat a solid like ice, it melts into a liquid (water). This process is called melting. If you continue to heat the water, it will boil and turn into a gas (steam). This is called evaporation or boiling.










