Write the structural formula of ethene (Ethylene)?
Answer
602.1k+ views
Hint: To answer this question we should be aware of alkene. Ethylene is widely used as a plant growth regulator that helps the process of senescence, ripening, and abscission in plants. It is marketed under the trade name serafin.
Complete Solution :
- Alkenes are the hydrocarbons with general formula ${{C}_{n}}{{H}_{2n}}$ where n is the number of carbon atoms. It is an unsaturated hydrocarbon as alkene contains c-c double bond in their molecules. Alkenes also known as Olefins where, Oleum means oil and fines means to care. The lower members of alkene form oily products on treating it with bromine or chlorine. They are produced by cracking of petroleum.
- The first member of the alkene series is ethylene, the number of carbon atoms in ethylene is 2.
General formula = ${{C}_{n}}{{H}_{2n}}$
n = 2 ( for ethylene)
Substituting n in general formula,
\[{{C}_{2}}{{H}_{4}}\]
- The chemical formula of ethylene is \[{{C}_{2}}{{H}_{4}}\].
- In ethylene the carbon atoms are of $s{{p}^{2}}$ hybridized . They are attached to each other by one $\sigma $ bond and a $\sigma $ bond. The $\sigma $ bond is obtained by the overlap of $s{{p}^{2}}$ hybrid orbital and the $\pi $ bond is obtained by the overlap of unhybridized p orbitals.
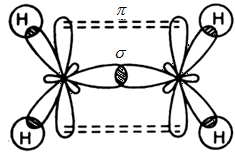
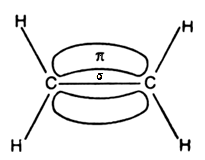
These two above mentioned diagrams are orbital structures of ethylene.
- The structural formula is:
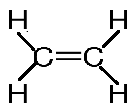
Note: The bond angle $\angle HCC$ and $\angle HCH$ are ${{120}^{\circ }}$. The bond angle of c = c is 1.34 $\overset{\circ }{\mathop{A}}\,$. In ethylene the carbon is $s{{p}^{2}}$ hybridized. In case of alkane the carbon will be $s{{p}^{3}}$ and in case of alkyne the carbon is sp hybridized. Ethylene and ethene are the same. Ethene is the IUPAC name and ethylene is a common name.
Complete Solution :
- Alkenes are the hydrocarbons with general formula ${{C}_{n}}{{H}_{2n}}$ where n is the number of carbon atoms. It is an unsaturated hydrocarbon as alkene contains c-c double bond in their molecules. Alkenes also known as Olefins where, Oleum means oil and fines means to care. The lower members of alkene form oily products on treating it with bromine or chlorine. They are produced by cracking of petroleum.
- The first member of the alkene series is ethylene, the number of carbon atoms in ethylene is 2.
General formula = ${{C}_{n}}{{H}_{2n}}$
n = 2 ( for ethylene)
Substituting n in general formula,
\[{{C}_{2}}{{H}_{4}}\]
- The chemical formula of ethylene is \[{{C}_{2}}{{H}_{4}}\].
- In ethylene the carbon atoms are of $s{{p}^{2}}$ hybridized . They are attached to each other by one $\sigma $ bond and a $\sigma $ bond. The $\sigma $ bond is obtained by the overlap of $s{{p}^{2}}$ hybrid orbital and the $\pi $ bond is obtained by the overlap of unhybridized p orbitals.


These two above mentioned diagrams are orbital structures of ethylene.
- The structural formula is:

Note: The bond angle $\angle HCC$ and $\angle HCH$ are ${{120}^{\circ }}$. The bond angle of c = c is 1.34 $\overset{\circ }{\mathop{A}}\,$. In ethylene the carbon is $s{{p}^{2}}$ hybridized. In case of alkane the carbon will be $s{{p}^{3}}$ and in case of alkyne the carbon is sp hybridized. Ethylene and ethene are the same. Ethene is the IUPAC name and ethylene is a common name.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




