Write the chemical reaction for the preparation of phenol from chlorobenzene.
Answer
615.3k+ views
Hint: Chlorobenzene has partial double bond character. Removal of chlorine atom from benzene rings requires high temperature and high pressure.
Complete answer:
Phenol is an organic compound containing a benzene ring bonded to the hydroxyl group.Phenol are weak acids and generally form phenoxide ions by losing one positive hydrogen ion from the hydroxyl group.
Chlorobenzene is formed by monochlorination of the benzene ring. To form chloro benzene, the Benzene ring is reacted with chlorine gas and $FeC{{l}_{3}}$. The lone pair of electrons present on chlorine atoms are in conjugation with the pie electrons of the benzene ring, therefore, Chlorobenzene is having a double bond character therefore, removal of chlorine atom from benzene is not easy. Removal of chlorine from benzene atoms requires high pressure and temperature,because chlorobenzene is less reactive and does not undergo nucleophilic substitution reaction easily. Therefore, for removal of chlorine atom, Chlorobenzene is reacted with Sodium Hydroxide at high pressure of 300 atm and high temperature of 623K. This results in the formation of sodium phenoxide. Sodium phenoxide on acidification with HCl results in the formation of Phenol.
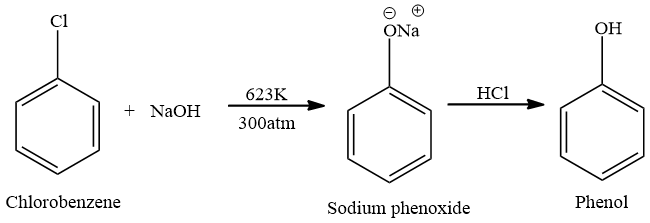
Note: When the electron withdrawing group is attached to the benzene ring, the removal of chlorine group becomes easy. When an electron withdrawing group like Nitro group, cyano group is attached to the ortho or para position of chlorobenzene. Trinitro chlorobenzene is highly reactive and gets hydrolysed at room temperature.
Complete answer:
Phenol is an organic compound containing a benzene ring bonded to the hydroxyl group.Phenol are weak acids and generally form phenoxide ions by losing one positive hydrogen ion from the hydroxyl group.
Chlorobenzene is formed by monochlorination of the benzene ring. To form chloro benzene, the Benzene ring is reacted with chlorine gas and $FeC{{l}_{3}}$. The lone pair of electrons present on chlorine atoms are in conjugation with the pie electrons of the benzene ring, therefore, Chlorobenzene is having a double bond character therefore, removal of chlorine atom from benzene is not easy. Removal of chlorine from benzene atoms requires high pressure and temperature,because chlorobenzene is less reactive and does not undergo nucleophilic substitution reaction easily. Therefore, for removal of chlorine atom, Chlorobenzene is reacted with Sodium Hydroxide at high pressure of 300 atm and high temperature of 623K. This results in the formation of sodium phenoxide. Sodium phenoxide on acidification with HCl results in the formation of Phenol.

Note: When the electron withdrawing group is attached to the benzene ring, the removal of chlorine group becomes easy. When an electron withdrawing group like Nitro group, cyano group is attached to the ortho or para position of chlorobenzene. Trinitro chlorobenzene is highly reactive and gets hydrolysed at room temperature.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




