Which one of the following is not expected to undergo iodoform reaction $ ? $
(A) Propan $ - 2 - $ ol
(B) $ 1 - $ Phenyl ethanol
(C) $ 2 - $ Butanol
(D) Ethanol
(E) Diphenyl methanol
Answer
555.6k+ views
Hint :Iodoform is an iodine containing organic compound with molecular formula $ CH{I_3} $ . It is a crystalline solid that has a characteristic pale yellow colour and distinct odour. Iodoform test is used to determine the presence of $ C{H_3}CO $ or $ C{H_3}CH(OH) $ groups in the given organic compound. It is a test given by certain aldehydes, ketones, and secondary alcohols.
Complete Step By Step Answer:
Iodoform test is given by aldehydes, ketones, secondary alcohols that have an alpha methyl group attached to the carbon containing functional group. It tests the presence of $ C{H_3}CO $ and $ C{H_3}CH(OH) $ . When iodine reacts with a base (NaOH) and the carbonyl group of the organic compound, it forms a yellow precipitate which is iodoform. Below is an example of an iodoform test.
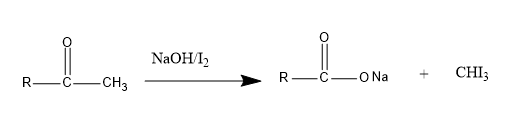
Now, let us examine the given compounds.
(A)
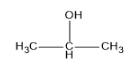
It is a secondary alcohol and undergoes iodoform test since it contains $ C{H_3}CH(OH) $ group.
(B)
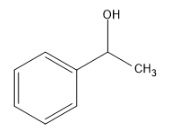
Compound contains the $ C{H_3}CH(OH) $ group and therefore gives positive results for the iodoform test.
(C)
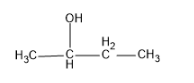
$ 2 - $ Butanol also contains $ C{H_3}CH(OH) $ group and hence a yellow precipitate of iodoform is formed.
(D)
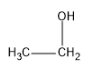
Ethanol is the only primary alcohol that gives an iodoform test.
(E)
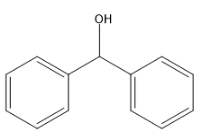
It does not contain any alpha methyl group. Iodoform is not formed when the given compound reacts with NaOH and $ {I_2} $ . There is no $ C{H_3}CO $ or $ C{H_3}CH(OH) $ group.
Therefore, the right option is (E) Diphenyl methanol.
Note :
Iodoform also known as triiodomethane or carbon triiodide has a distinct odour that is similar to chloroform. It has antimicrobial properties and is used as disinfectant. It is also used as an antiseptic.
Acetaldehyde is the only aldehyde that gives positive iodoform test.
Iodoform test is an important reaction used for the production of iodoform.
Complete Step By Step Answer:
Iodoform test is given by aldehydes, ketones, secondary alcohols that have an alpha methyl group attached to the carbon containing functional group. It tests the presence of $ C{H_3}CO $ and $ C{H_3}CH(OH) $ . When iodine reacts with a base (NaOH) and the carbonyl group of the organic compound, it forms a yellow precipitate which is iodoform. Below is an example of an iodoform test.

Now, let us examine the given compounds.
(A)

It is a secondary alcohol and undergoes iodoform test since it contains $ C{H_3}CH(OH) $ group.
(B)

Compound contains the $ C{H_3}CH(OH) $ group and therefore gives positive results for the iodoform test.
(C)

$ 2 - $ Butanol also contains $ C{H_3}CH(OH) $ group and hence a yellow precipitate of iodoform is formed.
(D)

Ethanol is the only primary alcohol that gives an iodoform test.
(E)

It does not contain any alpha methyl group. Iodoform is not formed when the given compound reacts with NaOH and $ {I_2} $ . There is no $ C{H_3}CO $ or $ C{H_3}CH(OH) $ group.
Therefore, the right option is (E) Diphenyl methanol.
Note :
Iodoform also known as triiodomethane or carbon triiodide has a distinct odour that is similar to chloroform. It has antimicrobial properties and is used as disinfectant. It is also used as an antiseptic.
Acetaldehyde is the only aldehyde that gives positive iodoform test.
Iodoform test is an important reaction used for the production of iodoform.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




