Which of the molecules is trigonal bipyramidal?
A. $B{{F}_{3}}$
B. $C{{H}_{4}}$
C. $PC{{l}_{5}}$
D. $S{{F}_{6}}$
Answer
583.2k+ views
Hint: The number of bonds, lone pair of electrons, and the hybridization of the central atom explain the shape of the molecules. If there is a presence of a lone pair of electrons on the central atom the shape of the molecule is going to change.
Complete step by step answer:
- In the question, it is asked which molecules have the shape of trigonal bipyramidal among the given options.
- First, we should know the number of lone pairs on the central atom, the number of bonds, and the hybridization of the given molecules to find the shape of the given molecules.
- Coming to option A, $B{{F}_{3}}$. The boron atom does not contain any lone pair of electrons on the central atom, the hybridization of the molecule is $s{{p}^{2}}$ and the number of bonds is three then the structure of $B{{F}_{3}}$ is trigonal planar and it is as follows.
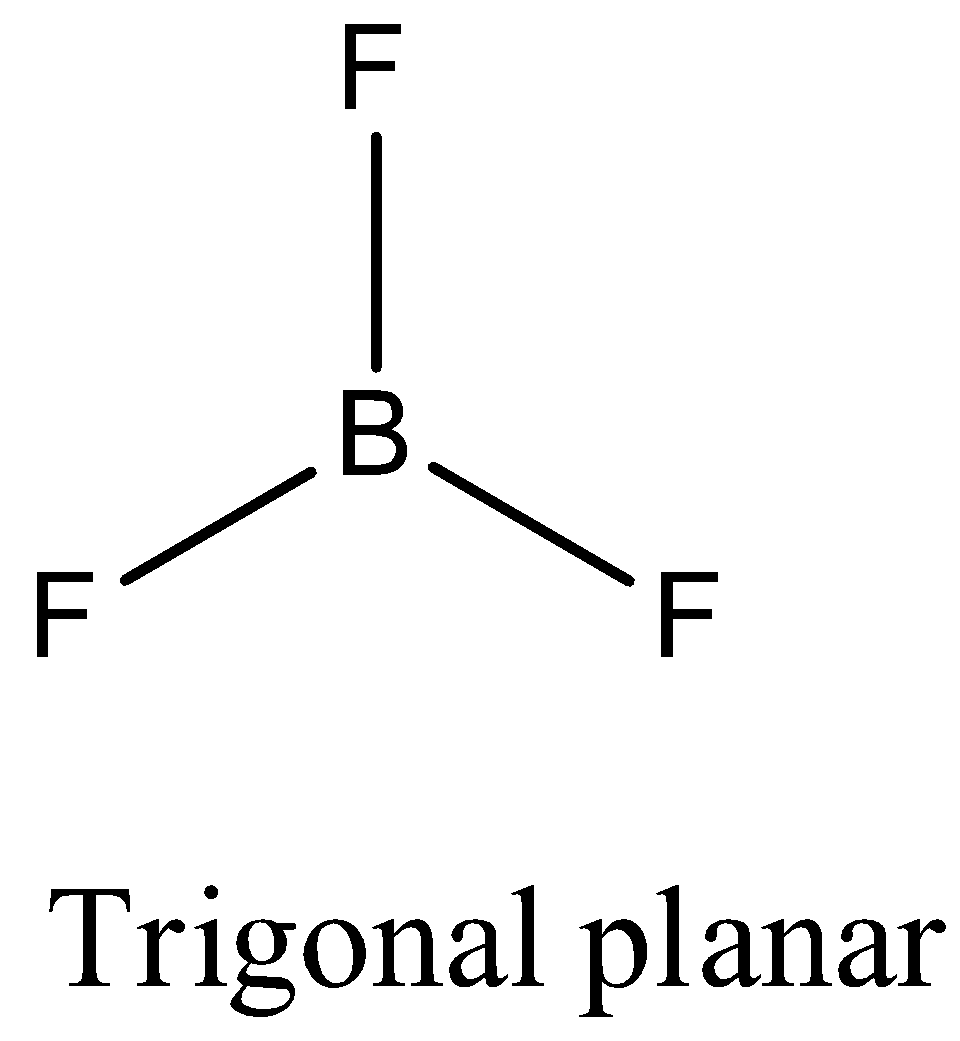
- Coming to option B, $C{{H}_{4}}$ . The carbon atom does not contain any lone pair of electrons on the central atom, the hybridization of the molecule is $s{{p}^{3}}$ and the numbers of bonds are four then the structure of $C{{H}_{4}}$ is tetrahedral and it is as follows.
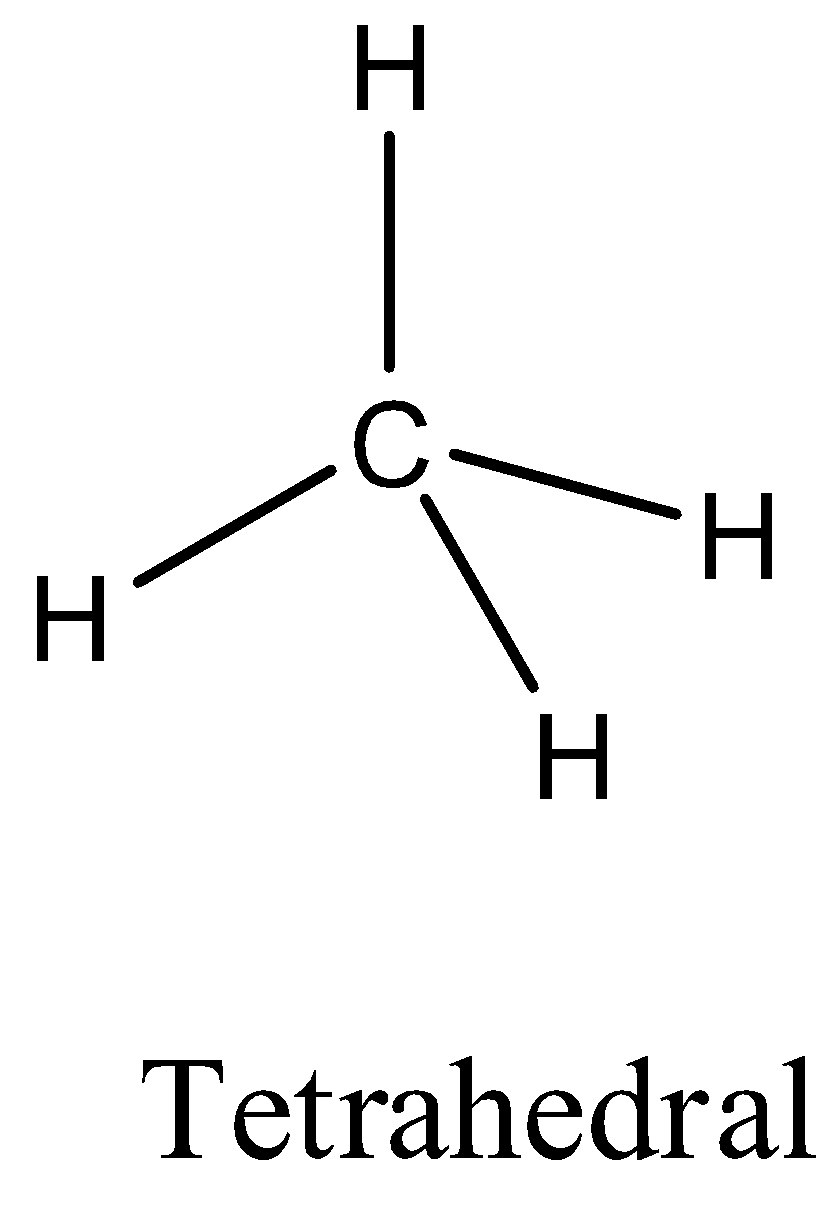
- Coming to the option C, $PC{{l}_{5}}$ . The phosphorus atom does not contain any lone pair of electrons on the central atom, the hybridization of the molecule is $s{{p}^{3}}d$and the numbers of bonds are five then the structure of is trigonal bipyramidal and it is as follows.
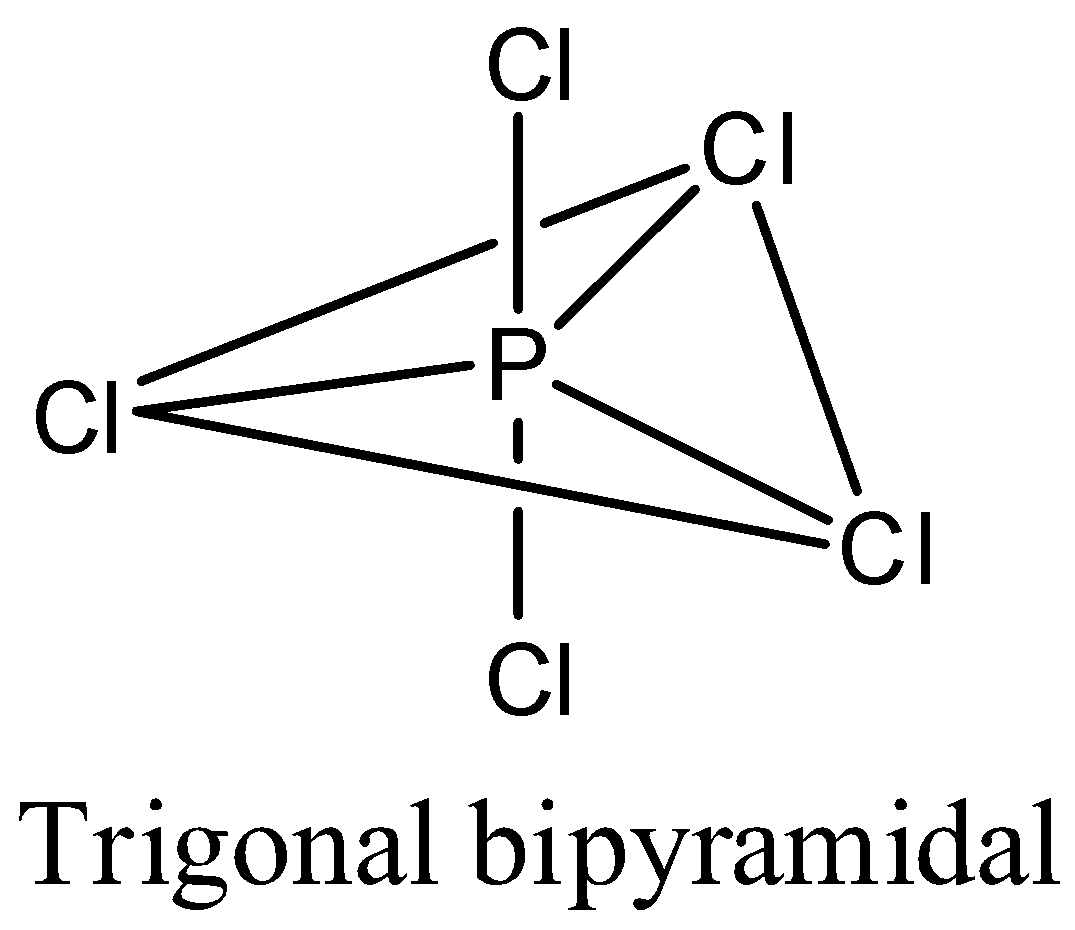
- Coming to the option D, $S{{F}_{6}}$. The sulfur atom does not contain any lone pair of electrons on the central atom, the hybridization of the molecule is $s{{p}^{3}}{{d}^{2}}$ and the numbers of bonds are six then the structure of is octahedral and it is as follows.
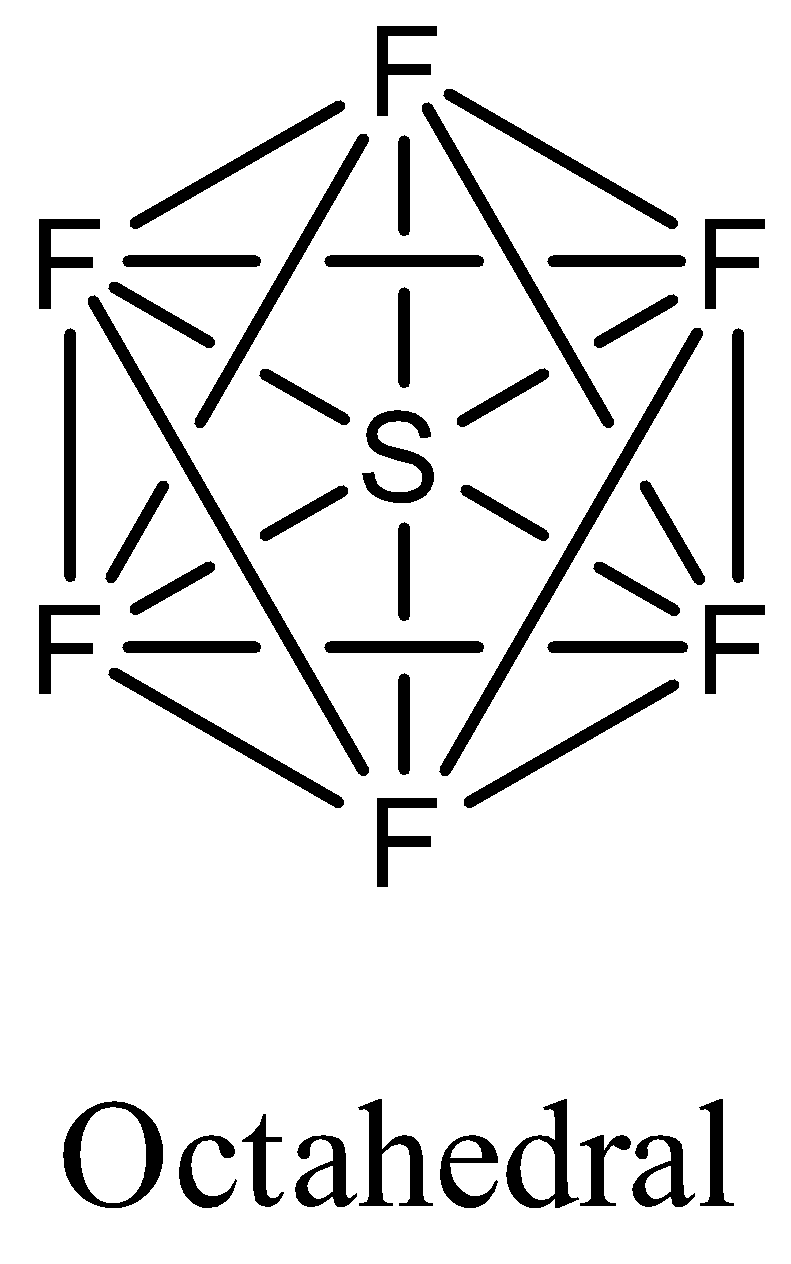
- Therefore the molecule which has trigonal bipyramidal structure among the given options is $PC{{l}_{5}}$. So, the correct answer is “Option C”.
Note: If a lone pair of electrons are present on the central atom in the molecule then the shape of the molecule is going to change because of the repulsions between lone pair and bond pair electrons in the molecule.
Complete step by step answer:
- In the question, it is asked which molecules have the shape of trigonal bipyramidal among the given options.
- First, we should know the number of lone pairs on the central atom, the number of bonds, and the hybridization of the given molecules to find the shape of the given molecules.
- Coming to option A, $B{{F}_{3}}$. The boron atom does not contain any lone pair of electrons on the central atom, the hybridization of the molecule is $s{{p}^{2}}$ and the number of bonds is three then the structure of $B{{F}_{3}}$ is trigonal planar and it is as follows.

- Coming to option B, $C{{H}_{4}}$ . The carbon atom does not contain any lone pair of electrons on the central atom, the hybridization of the molecule is $s{{p}^{3}}$ and the numbers of bonds are four then the structure of $C{{H}_{4}}$ is tetrahedral and it is as follows.

- Coming to the option C, $PC{{l}_{5}}$ . The phosphorus atom does not contain any lone pair of electrons on the central atom, the hybridization of the molecule is $s{{p}^{3}}d$and the numbers of bonds are five then the structure of is trigonal bipyramidal and it is as follows.

- Coming to the option D, $S{{F}_{6}}$. The sulfur atom does not contain any lone pair of electrons on the central atom, the hybridization of the molecule is $s{{p}^{3}}{{d}^{2}}$ and the numbers of bonds are six then the structure of is octahedral and it is as follows.

- Therefore the molecule which has trigonal bipyramidal structure among the given options is $PC{{l}_{5}}$. So, the correct answer is “Option C”.
Note: If a lone pair of electrons are present on the central atom in the molecule then the shape of the molecule is going to change because of the repulsions between lone pair and bond pair electrons in the molecule.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




