
Which of the following statements regarding the structure of $SOC{l_2}$ is not correct ?
( A ) The sulphur is $s{p^3}$ hybridised and it has a tetrahedral shape .
( B ) The sulphur is $s{p^3}$ hybridised and it has a trigonal pyramid shape .
( C ) The oxygen – sulphur bond is p$\pi $ – d$\pi $ bond .
( D ) It contains one lone pair of electrons in the $s{p^3}$ hybrid orbital of sulphur .
Answer
585k+ views
Hint: As three atoms are bonded to the sulphur atom, we can say that the sulphur atom is $s{p^3}$ hybridized.And by referring to the shapes of atoms of different hybridization, it’s a trigonal pyramid shape. The shape and geometry of the molecule are defined by minimum repulsion.
For 3 electron pairs, the shape is trigonal pyramidal.
Complete step by step solution:
First, we consider, option ( A )
The $SOC{l_2}$ molecule is $s{p^3}$ hybridized.
The formula for finding hybridization of molecules is
Hybridization $ = \dfrac{1}{2}\left( {V + H - C + A} \right)$
Here, V $ = $ VALENCE ELECTRONS IN CENTRAL METAL ATOM
H $ = $ Number of monovalent atom attached to the central metal atom
C $ = $ Cation charge
A $ = $ Anion charge
Substituting the values for $SOC{l_2}$,
$\Rightarrow $Hybridization $ = $ $\dfrac{1}{2}$( 6 $ + $2 $ + $0 $ + $0 )
$ = $$\dfrac{1}{2}$( 8 )
$ = $4
Hence, the hybridization of $SOC{l_2}$is $s{p^3}$
Now, the structure of $SOC{l_2}$ is a trigonal planar because of the electron densities of 2 chlorine atoms and 1 oxygen atom, as given in the image.
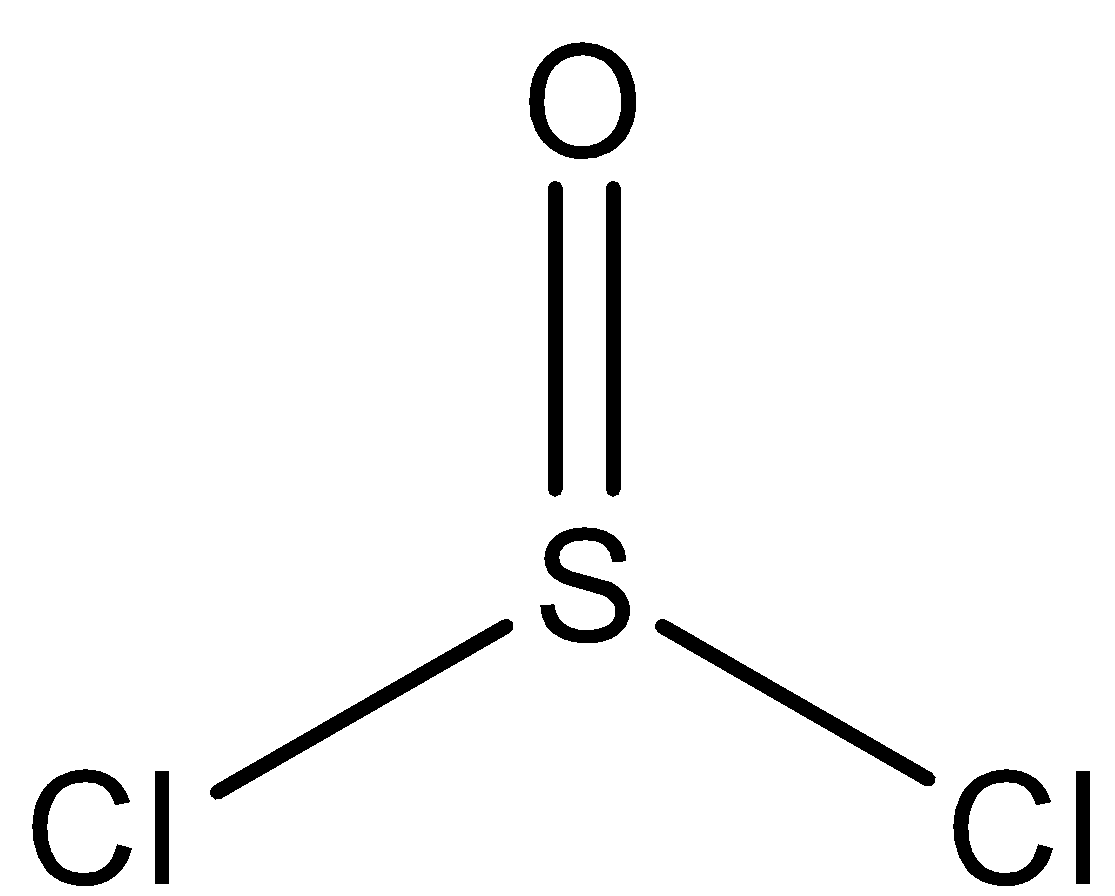
Hence, option ( A ) is incorrect
For option ( B ),
The statement is true, as proved above that the sulphur is $s{p^3}$ hybridized and it has a trigonal pyramid shape.
Hence, option ( B ) is correct
For option ( C ),
The lone pair of p orbital back bonds with that of d orbital of central atom sulphur.
Hence, option ( C ) is correct
For option ( D ),
The electronic configuration of the sulphur atom is
1${s^2}$ 2${s^2}$ 2${p^6}$ 3${s^2}$ 3${p^4}$
The 4 valence electrons of sulphur bond with 2 chlorine atoms and 1 oxygen atom leaving behind 2 valence electrons out of 6
Hence, the sulphur atom contains 1 lone pair of electrons
Hence, option ( D ) is correct
So, The correct option is ( A ) - The sulphur is $s{p^3}$ hybridized and it has a tetrahedral shape.
Note: The geometry of $SOC{l_2}$ is due to the lone pair effect. The VSEPR Theory ( Valence Shell Electron Pair Repulsion ) gives the geometry and shape of molecules.
According to the VSEPR Theory the bond angle between oxygen, sulphur and chlorine are $106^\circ $and between chlorine, sulphur, chlorine is $96^\circ $ respectively.
For 3 electron pairs, the shape is trigonal pyramidal.
Complete step by step solution:
First, we consider, option ( A )
The $SOC{l_2}$ molecule is $s{p^3}$ hybridized.
The formula for finding hybridization of molecules is
Hybridization $ = \dfrac{1}{2}\left( {V + H - C + A} \right)$
Here, V $ = $ VALENCE ELECTRONS IN CENTRAL METAL ATOM
H $ = $ Number of monovalent atom attached to the central metal atom
C $ = $ Cation charge
A $ = $ Anion charge
Substituting the values for $SOC{l_2}$,
$\Rightarrow $Hybridization $ = $ $\dfrac{1}{2}$( 6 $ + $2 $ + $0 $ + $0 )
$ = $$\dfrac{1}{2}$( 8 )
$ = $4
Hence, the hybridization of $SOC{l_2}$is $s{p^3}$
Now, the structure of $SOC{l_2}$ is a trigonal planar because of the electron densities of 2 chlorine atoms and 1 oxygen atom, as given in the image.

Hence, option ( A ) is incorrect
For option ( B ),
The statement is true, as proved above that the sulphur is $s{p^3}$ hybridized and it has a trigonal pyramid shape.
Hence, option ( B ) is correct
For option ( C ),
The lone pair of p orbital back bonds with that of d orbital of central atom sulphur.
Hence, option ( C ) is correct
For option ( D ),
The electronic configuration of the sulphur atom is
1${s^2}$ 2${s^2}$ 2${p^6}$ 3${s^2}$ 3${p^4}$
The 4 valence electrons of sulphur bond with 2 chlorine atoms and 1 oxygen atom leaving behind 2 valence electrons out of 6
Hence, the sulphur atom contains 1 lone pair of electrons
Hence, option ( D ) is correct
So, The correct option is ( A ) - The sulphur is $s{p^3}$ hybridized and it has a tetrahedral shape.
Note: The geometry of $SOC{l_2}$ is due to the lone pair effect. The VSEPR Theory ( Valence Shell Electron Pair Repulsion ) gives the geometry and shape of molecules.
According to the VSEPR Theory the bond angle between oxygen, sulphur and chlorine are $106^\circ $and between chlorine, sulphur, chlorine is $96^\circ $ respectively.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

The largest wind power cluster is located in the state class 11 social science CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE

Can anyone list 10 advantages and disadvantages of friction




