Which of the following is not correctly matched?
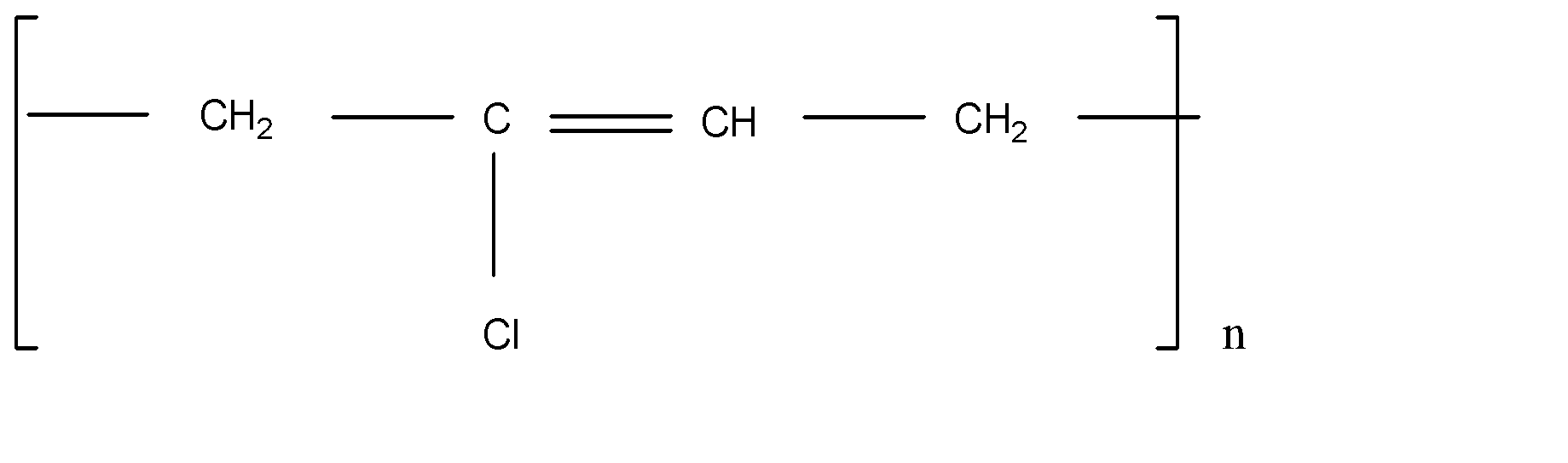
A.Neoprene
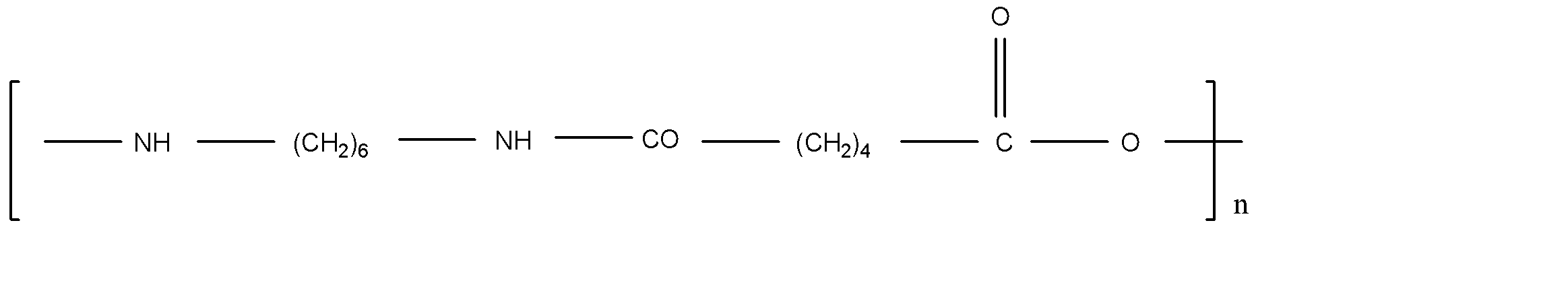
B.Nylon 6,6
 C.Terylene
C.Terylene
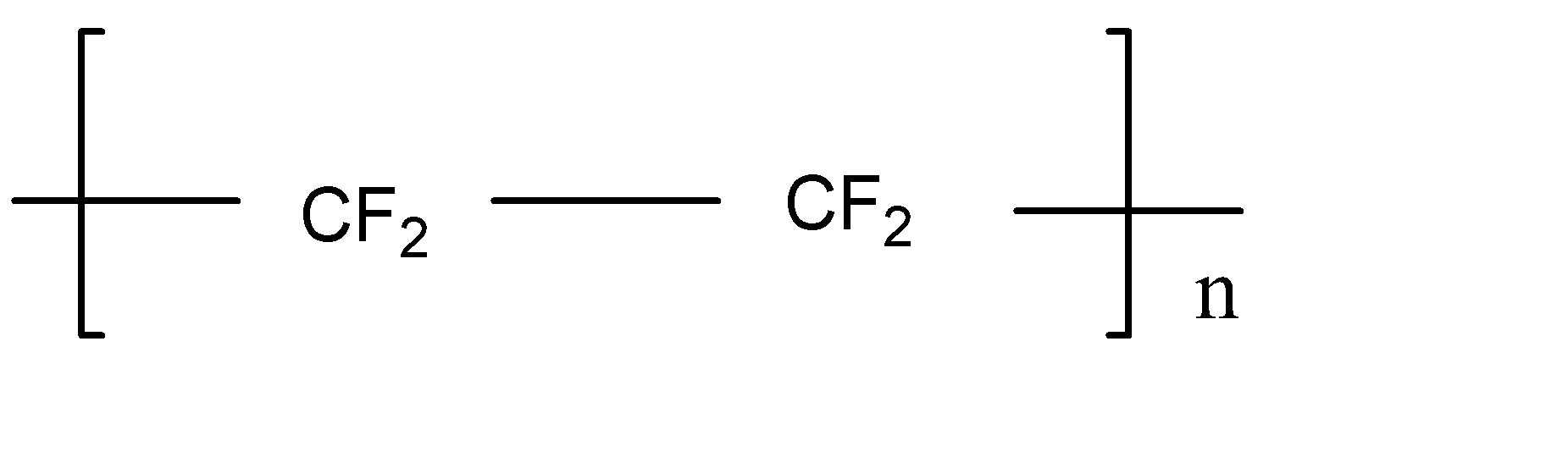
D.Teflon



Answer
605.7k+ views
Hint: Polymers are long chain compounds, which are repeating chains of molecules. Polymers are a large number of molecules which are made by chemical bonding. Here we see the examples of polymers and find the option which is not correctly matched by repeat molecules and form polymers. Neoprene, Nylon 6,6 and Teflon are given in the long chain. In these polymers, molecules are repeating and form long chain molecules.
Complete step by step answer:
Here we see the example of polymers, we find the polymer which is not correctly matched. The examples of polymers are given by structure in this option. Here Terylene is not described by the structure.
Nylon 6,6 is the polyamide polymer. The monomer in nylon 6,6 is polyamide. Neoprene is the polychloroprene and it exhibits good chemical stability and maintains flexibility. Ethylene glycol and terephthalic acid react and form Terylene by polymerization. Teflon is a polytetrafluoroethylene polymer.
Hence, option (C) is the correct answer.
Additional information:
Polymer is a useful chemical made of many repeating units. It is made up of many of the materials in living organisms, including proteins, cellulose and nucleic acids. Many important polymers have nitrogen or oxygen atoms. The nylon and polyester in jackets or sneakers. Polymers are large molecules made by bonding.
Note: Here Terylene is the polymer between two molecules, but other polymers are the long chain of single molecules. It forms by the single molecule by repeat. Nylon 6,6, Neoprene and Teflon is formed by the single molecule and Terylene is the formation of two different molecules. That’s why Terylene is different from others.
Complete step by step answer:
Here we see the example of polymers, we find the polymer which is not correctly matched. The examples of polymers are given by structure in this option. Here Terylene is not described by the structure.
Nylon 6,6 is the polyamide polymer. The monomer in nylon 6,6 is polyamide. Neoprene is the polychloroprene and it exhibits good chemical stability and maintains flexibility. Ethylene glycol and terephthalic acid react and form Terylene by polymerization. Teflon is a polytetrafluoroethylene polymer.
Hence, option (C) is the correct answer.
Additional information:
Polymer is a useful chemical made of many repeating units. It is made up of many of the materials in living organisms, including proteins, cellulose and nucleic acids. Many important polymers have nitrogen or oxygen atoms. The nylon and polyester in jackets or sneakers. Polymers are large molecules made by bonding.
Note: Here Terylene is the polymer between two molecules, but other polymers are the long chain of single molecules. It forms by the single molecule by repeat. Nylon 6,6, Neoprene and Teflon is formed by the single molecule and Terylene is the formation of two different molecules. That’s why Terylene is different from others.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




