Which of the following is a transition element?
A. $Al$
B.$As$
C.$Ni$
D.$Rb$
Answer
603.3k+ views
Hint: Transition elements are those elements which are placed between metals and non - metals in periodic table. They are also called d-block elements.
Complete step by step answer:
Let us first explain the periodic table.
According to modern periodic table given by Moslay:
Non - Metals are placed in group \[13\] to group \[17.\]Inert gases are placed in group 18. And transition elements are placed in group 3 to group 12.
Aluminum$(Al)$ belongs to group \[3.\] Therefore, it is metal.
Arsenic$(As)$ belongs to the group \[15\] therefore it is metalloid.
Rubidium $({R_b})$ belongs to group $1$. Therefore, it is metal.
Nickel $(Ni)$ belongs to group $10$, therefore it is transition metal.
So, the correct answer is “Option C”.
Additional information:
The Atomic Number of $Ni$ is $28.$
It’s electronic configuration is $1{s^2}2{s^2}2{p^6}3{s^2}3{p^6}3{d^8}4{s^2}.$
$ = [Ne]3{s^2}3{p^6}3{d^8}4{s^2}$.
Since Electronic configuration of $Ne$ is $1{s^2}2{s^2}2{p^6}$. Therefore, we can write $Ne$ in place of $1{s^2}2{s^2}2{p^6}$.
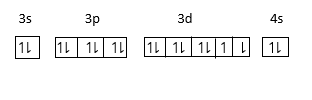
The energy of $4s$ orbital is less than $3d$ orbital therefore electron enters in $4s$ orbital first and then $3d$ orbital therefore electron enters in us first and then $3d.$ Thus last electron in Nickel enters in $d - $ orbital. Therefore, it is known as $d - block$ elements.
Note:
There are four transition series called as first (4d), second (5d), third (6d) and Fourth (7d). Nickel belongs to the first transition series.
Transition element can be identified by filling electrons in the orbital. If the last electron enters in $d - $orbital it is a transition element. Electrons should be filled according to Aufbau principle.
Complete step by step answer:
Let us first explain the periodic table.
According to modern periodic table given by Moslay:
Non - Metals are placed in group \[13\] to group \[17.\]Inert gases are placed in group 18. And transition elements are placed in group 3 to group 12.
Aluminum$(Al)$ belongs to group \[3.\] Therefore, it is metal.
Arsenic$(As)$ belongs to the group \[15\] therefore it is metalloid.
Rubidium $({R_b})$ belongs to group $1$. Therefore, it is metal.
Nickel $(Ni)$ belongs to group $10$, therefore it is transition metal.
So, the correct answer is “Option C”.
Additional information:
The Atomic Number of $Ni$ is $28.$
It’s electronic configuration is $1{s^2}2{s^2}2{p^6}3{s^2}3{p^6}3{d^8}4{s^2}.$
$ = [Ne]3{s^2}3{p^6}3{d^8}4{s^2}$.
Since Electronic configuration of $Ne$ is $1{s^2}2{s^2}2{p^6}$. Therefore, we can write $Ne$ in place of $1{s^2}2{s^2}2{p^6}$.

The energy of $4s$ orbital is less than $3d$ orbital therefore electron enters in $4s$ orbital first and then $3d$ orbital therefore electron enters in us first and then $3d.$ Thus last electron in Nickel enters in $d - $ orbital. Therefore, it is known as $d - block$ elements.
Note:
There are four transition series called as first (4d), second (5d), third (6d) and Fourth (7d). Nickel belongs to the first transition series.
Transition element can be identified by filling electrons in the orbital. If the last electron enters in $d - $orbital it is a transition element. Electrons should be filled according to Aufbau principle.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




