Which of the following have –M effect ( \[{e^ - }\] withdrawing effect)?
A.
B.\[ - S{O_3}H\]
C.\[ - OR\]
D.\[ - Br\]

Answer
612.9k+ views
Hint: The electronegativity of an element will have capability to withdraw the electron from the neighbouring atom towards itself.
Complete Step by step answer:
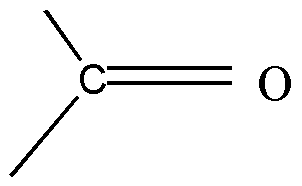
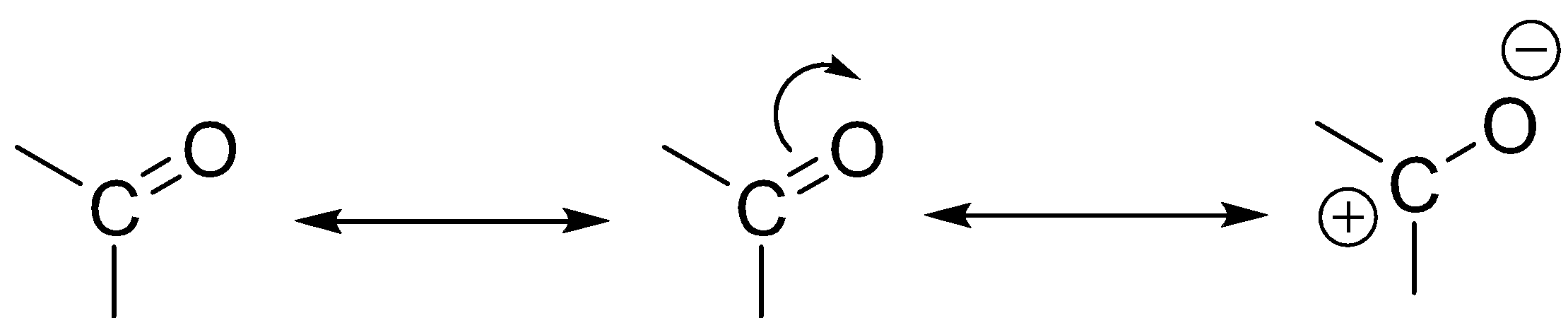
Carbonyl:
The \[C = O\] functional group available in a system will have –M effect as the oxygen atom which is highly electronegative in nature will attract the electron from the \[C - O\] bond and make the carbon electron deficient. They will lead to the withdrawal of electrons from the system where the \[C = O\] is attached with.
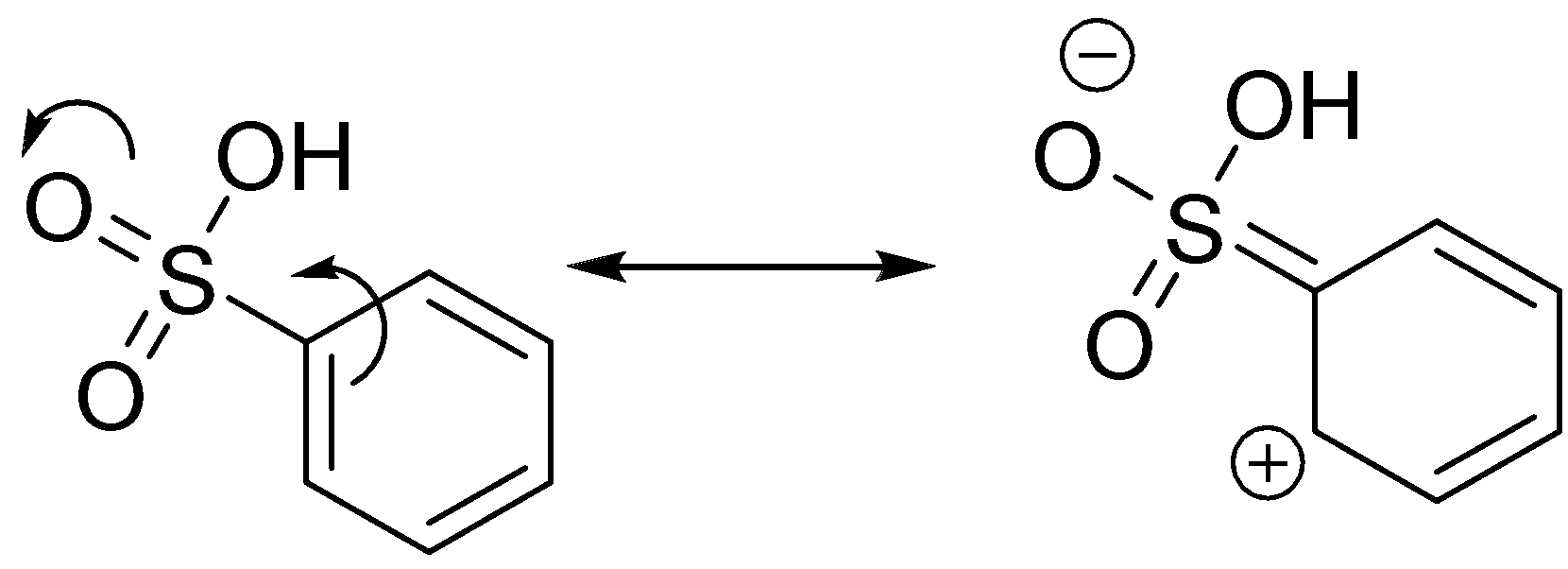
Sulfonic acid:
The sulphur is relatively higher in size than carbon. Similar electron withdrawing effect was observed as the presence of two oxygen atoms connected with sulphur through the pi bond.
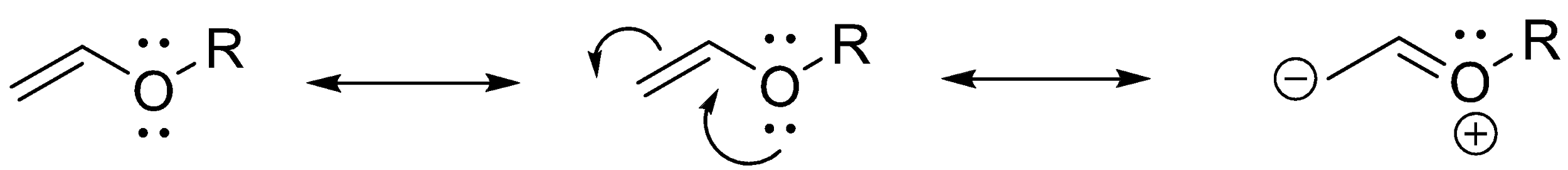
\[ - OR\] :
Alkyl and alkoxides are ready to donate electrons into the system where it is connected and hence, they show +M effect. The lone pair of electrons present in the oxygen will be moved towards the pi bond present in the molecule.
\[ - Br\] :
In general, the halogens are electronegative in nature but the electronegativity reduces when going from top to bottom in the periodic table. Bromine is bigger in size and the effect was very less. Bromine is the least electronegative halogen atom.
Note: Groups like \[C = O\] , \[ - N{O_2}\] , \[ - COOH\] , \[ - COOR\] , \[ - S{O_3}H\] when present adjacent to a multiple bond in molecules are conjugated with the multiple bonds. They bring resonance into the molecule by withdrawing the π electrons and cause electron displacement towards themselves.
When a group showing -M effect is present as a substituent in benzene, it makes the benzene ring less reactive towards electrophilic substitution.
Complete Step by step answer:
Carbonyl:
The \[C = O\] functional group available in a system will have –M effect as the oxygen atom which is highly electronegative in nature will attract the electron from the \[C - O\] bond and make the carbon electron deficient. They will lead to the withdrawal of electrons from the system where the \[C = O\] is attached with.

Sulfonic acid:
The sulphur is relatively higher in size than carbon. Similar electron withdrawing effect was observed as the presence of two oxygen atoms connected with sulphur through the pi bond.

\[ - OR\] :
Alkyl and alkoxides are ready to donate electrons into the system where it is connected and hence, they show +M effect. The lone pair of electrons present in the oxygen will be moved towards the pi bond present in the molecule.

\[ - Br\] :
In general, the halogens are electronegative in nature but the electronegativity reduces when going from top to bottom in the periodic table. Bromine is bigger in size and the effect was very less. Bromine is the least electronegative halogen atom.
Note: Groups like \[C = O\] , \[ - N{O_2}\] , \[ - COOH\] , \[ - COOR\] , \[ - S{O_3}H\] when present adjacent to a multiple bond in molecules are conjugated with the multiple bonds. They bring resonance into the molecule by withdrawing the π electrons and cause electron displacement towards themselves.
When a group showing -M effect is present as a substituent in benzene, it makes the benzene ring less reactive towards electrophilic substitution.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




