Which C-H bond has minimum bond dissociation energy for homolysis
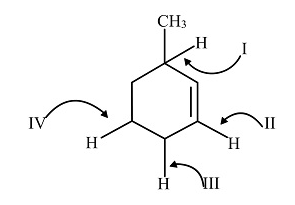
A. IV
B. III
C. II
D. I

Answer
590.1k+ views
Hint: The bond dissociation energy is defined as the amount of energy required to break the chemical bond formed between the two atoms in a chemical compound. The compound which forms a stable intermediate will be strong and high energy will be required to break the bond.
Complete step by step answer:
The covalent bond present between the two atoms is broken by two ways. It can be done either by heterolytic cleavage or by homolytic cleavage.
In homolytic cleavage, the product formed are radicals and the energy required for breaking the covalent bond is known as bond dissociation energy.
The homolytic bond dissociation energy is defined as the amount of energy required to break one mole of gases which are covalently bonded into radical pairs. The SI unit of bond dissociation energy is kJ/mol.
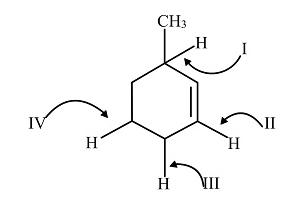
In the given compound, there are four hydrogen atoms bonded to carbon atoms by a covalent bond. But the C-H bond present at first position forms a stable intermediate by resonance formed by the radical due to the presence of methyl group which shows positive inductive effect, the double bond and the hyperconjugation due to five hydrogen atoms present in the ring.
Therefore, the C-H bond at (I) position requires minimum bond-dissociation energy to break the bond.
So, the correct answer is Option A.
Note: On comparing the primary C-H bond, secondary C-H Bond and tertiary C-H bond, the tertiary C-H bond requires less bond dissociation energy.
Complete step by step answer:
The covalent bond present between the two atoms is broken by two ways. It can be done either by heterolytic cleavage or by homolytic cleavage.
In homolytic cleavage, the product formed are radicals and the energy required for breaking the covalent bond is known as bond dissociation energy.
The homolytic bond dissociation energy is defined as the amount of energy required to break one mole of gases which are covalently bonded into radical pairs. The SI unit of bond dissociation energy is kJ/mol.

In the given compound, there are four hydrogen atoms bonded to carbon atoms by a covalent bond. But the C-H bond present at first position forms a stable intermediate by resonance formed by the radical due to the presence of methyl group which shows positive inductive effect, the double bond and the hyperconjugation due to five hydrogen atoms present in the ring.
Therefore, the C-H bond at (I) position requires minimum bond-dissociation energy to break the bond.
So, the correct answer is Option A.
Note: On comparing the primary C-H bond, secondary C-H Bond and tertiary C-H bond, the tertiary C-H bond requires less bond dissociation energy.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




