Which among the following is a non-benzenoid aromatic compound?
A. o-xylene
B. Phenanthrene
C. Indole
D. Thiophene
Answer
618.9k+ views
Hint:Aromaticity means a conjugated system made of alternating single and double bonds in a ring. This conjugation allows the electrons in the molecule to be delocalized around the ring thus, increasing the molecule's stability.
Complete step by step answer:
To find the answer to this question, we should know what non-benzenoid compounds are, these are aromatic compounds which have highly unsaturated rings other than benzene rings. Aromatic compounds are cyclic and planar structures with a ring of resonance bonds. Aromatic molecules are very stable. Let us discuss the options one by one to find non-benzenoid aromatic compound:
A. o-xylene: It is also known as ortho-xylene is an aromatic compound with the formula ${{\text{C}}_{6}}{{\text{H}}_{4}}{{\left( \text{C}{{\text{H}}_{3}} \right)}_{2}}$. The two methyl groups bonded to adjacent carbon atoms of a benzene ring. It is an isomer of m-xylene and p-xylene and their mixture is called xylene or xylenes. Its structure is
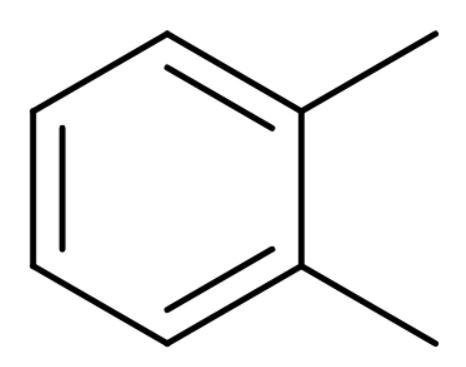
By structure it is clear that it is a benzenoid compound.
B. Phenanthrene: A polycyclic aromatic compound composed of three fused benzene rings. It is made up of phenyl and anthracene. Its structure is

As it is a fusion of benzene rings then, it is a benzenoid compound.
C. Indole: Its molecular formula is ${{\text{C}}_{8}}{{\text{H}}_{7}}\text{N}$ and it is an aromatic heterocyclic organic compound. Its structure is bicyclic consisting of a six-membered benzene ring attached to a five-membered pyrrole ring. Its structure is

The lone pair on nitrogen is in resonance with the rings and satisfies Huckel’s Rule (the compound should have $\left( 4\text{n}+\text{2} \right)\pi \text{ }{{\text{e}}^{-}}$ in it). This compound has 10 $\pi {{\text{e}}^{-}}$in it which satisfies the rule. It has benzene attached to it. So, it is a benzenoid aromatic compound.
D. Thiophene: Thiophene is aromatic, heterocyclic compound with molecular formula ${{\text{C}}_{4}}{{\text{H}}_{4}}\text{S}$. Consisting of a planar five-membered ring. It resembles benzene. The lone pair on sulphur is delocalized in the pi electron system. Its structure is

It has 6 $\pi {{\text{e}}^{-}}$ inside the ring. It is aromatic but it is not attached to any benzene ring. So, it is a non-benzenoid compound.
The correct answer to this question is option ‘d’ (Thiophene).
Note:
By using Huckel’s Rule, we can check whether a compound is aromatic or not. The conditions of aromaticity are (i) should have $\left( 4\text{n}+\text{2} \right)\pi \text{ }{{\text{e}}^{-}}$in it. (ii) should have cyclic and planar structure and (iii) the electrons present should be inside the ring only.
Complete step by step answer:
To find the answer to this question, we should know what non-benzenoid compounds are, these are aromatic compounds which have highly unsaturated rings other than benzene rings. Aromatic compounds are cyclic and planar structures with a ring of resonance bonds. Aromatic molecules are very stable. Let us discuss the options one by one to find non-benzenoid aromatic compound:
A. o-xylene: It is also known as ortho-xylene is an aromatic compound with the formula ${{\text{C}}_{6}}{{\text{H}}_{4}}{{\left( \text{C}{{\text{H}}_{3}} \right)}_{2}}$. The two methyl groups bonded to adjacent carbon atoms of a benzene ring. It is an isomer of m-xylene and p-xylene and their mixture is called xylene or xylenes. Its structure is

By structure it is clear that it is a benzenoid compound.
B. Phenanthrene: A polycyclic aromatic compound composed of three fused benzene rings. It is made up of phenyl and anthracene. Its structure is

As it is a fusion of benzene rings then, it is a benzenoid compound.
C. Indole: Its molecular formula is ${{\text{C}}_{8}}{{\text{H}}_{7}}\text{N}$ and it is an aromatic heterocyclic organic compound. Its structure is bicyclic consisting of a six-membered benzene ring attached to a five-membered pyrrole ring. Its structure is

The lone pair on nitrogen is in resonance with the rings and satisfies Huckel’s Rule (the compound should have $\left( 4\text{n}+\text{2} \right)\pi \text{ }{{\text{e}}^{-}}$ in it). This compound has 10 $\pi {{\text{e}}^{-}}$in it which satisfies the rule. It has benzene attached to it. So, it is a benzenoid aromatic compound.
D. Thiophene: Thiophene is aromatic, heterocyclic compound with molecular formula ${{\text{C}}_{4}}{{\text{H}}_{4}}\text{S}$. Consisting of a planar five-membered ring. It resembles benzene. The lone pair on sulphur is delocalized in the pi electron system. Its structure is

It has 6 $\pi {{\text{e}}^{-}}$ inside the ring. It is aromatic but it is not attached to any benzene ring. So, it is a non-benzenoid compound.
The correct answer to this question is option ‘d’ (Thiophene).
Note:
By using Huckel’s Rule, we can check whether a compound is aromatic or not. The conditions of aromaticity are (i) should have $\left( 4\text{n}+\text{2} \right)\pi \text{ }{{\text{e}}^{-}}$in it. (ii) should have cyclic and planar structure and (iii) the electrons present should be inside the ring only.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE

10 examples of friction in our daily life




