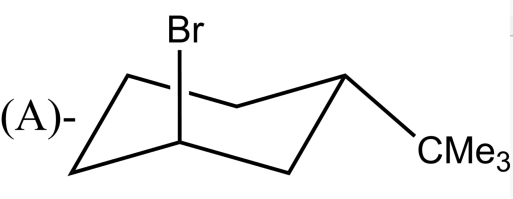
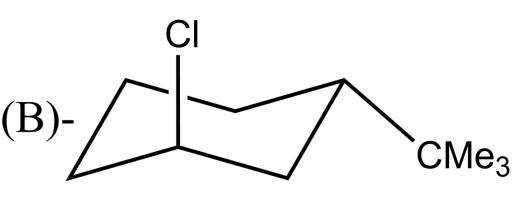
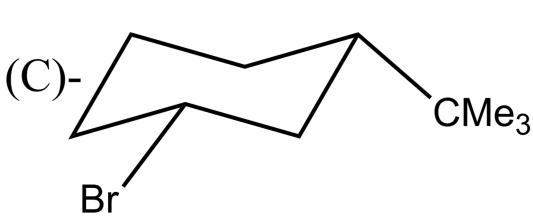
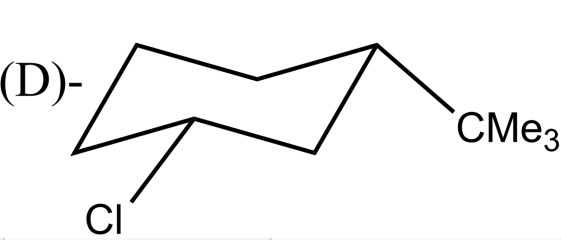
Which alkyl halide undergoes ${{E}_{2}}$ elimination?




Answer
615.9k+ views
Hint: ${{E}_{2}}$ reactions are a single step concerted reaction in which there is one transition state. ${{E}_{2}}$ reactions are seen typically with secondary and tertiary alkyl halides, but in the case of primary halide, there is a necessity of a hindered base.
Complete answer:
-${{E}_{2}}$ reactions, in order to proceed, must follow the following requirements-
(i) Secondary and tertiary alkyl halides will undergo ${{E}_{2}}$ reactions in the presence of a base like OH-, RO-, ${{R}_{2}}N-$ .
(ii) Both leaving groups must be present on the same plane. This will allow the formation of double bond giving stability to the complex.
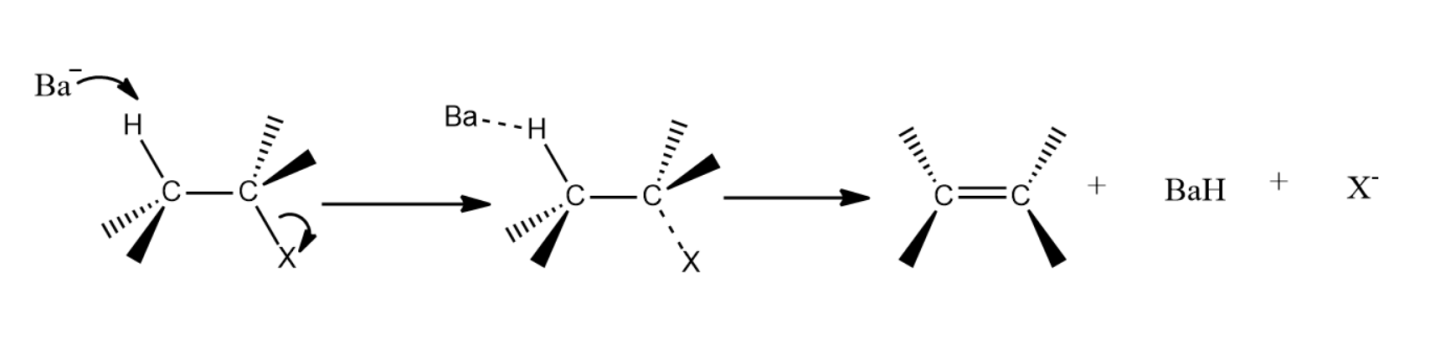
(iii) The complex must follow Zaitsev’s rule, which is a good predictor for simple elimination reactions of alkyl halide. The most substituted alkene is usually the major product at the end.
(iv) The complex must follow Hoffman rule, which says that when an elimination reaction takes place which can produce two or more alkenes (or alkyne) products, the product containing the less highly substituted pi bond is the major one.
-${{E}_{2}}$ reactions are anti elimination which means that there must be a proton in the beta position of the leaving group. In complexes (A) and (B) there Hydrogen atom is present in the anti position of the leaving group. After the removal of the leaving group, i.e Br and Cl respectively in option (A) and (B) an alkene will be formed. Whereas the complexes in (C) and (D) do not have Hydrogen atoms in the anti position of the leaving group.
So, the correct answer is “Option A and B”.
Note: ${{E}_{2}}$ elimination reactions of alkyl halides will dominate with most of the nucleophiles, even if they are weak bases. In high dielectric ionizing solvents, the products formed maybe ${{S}_{N}}1$ and ${{E}_{1}}$ .
Complete answer:
-${{E}_{2}}$ reactions, in order to proceed, must follow the following requirements-
(i) Secondary and tertiary alkyl halides will undergo ${{E}_{2}}$ reactions in the presence of a base like OH-, RO-, ${{R}_{2}}N-$ .
(ii) Both leaving groups must be present on the same plane. This will allow the formation of double bond giving stability to the complex.

(iii) The complex must follow Zaitsev’s rule, which is a good predictor for simple elimination reactions of alkyl halide. The most substituted alkene is usually the major product at the end.
(iv) The complex must follow Hoffman rule, which says that when an elimination reaction takes place which can produce two or more alkenes (or alkyne) products, the product containing the less highly substituted pi bond is the major one.
-${{E}_{2}}$ reactions are anti elimination which means that there must be a proton in the beta position of the leaving group. In complexes (A) and (B) there Hydrogen atom is present in the anti position of the leaving group. After the removal of the leaving group, i.e Br and Cl respectively in option (A) and (B) an alkene will be formed. Whereas the complexes in (C) and (D) do not have Hydrogen atoms in the anti position of the leaving group.
So, the correct answer is “Option A and B”.
Note: ${{E}_{2}}$ elimination reactions of alkyl halides will dominate with most of the nucleophiles, even if they are weak bases. In high dielectric ionizing solvents, the products formed maybe ${{S}_{N}}1$ and ${{E}_{1}}$ .
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




