What is the action of chlorine on ${{C}}{{{S}}_{{2}}}$
Answer
581.7k+ views
Hint: We know that ${{C}}{{{S}}_{{2}}}$ is a volatile colourless liquid. It is called carbon disulphide. The structure of ${{C}}{{{S}}_{{2}}}$ is
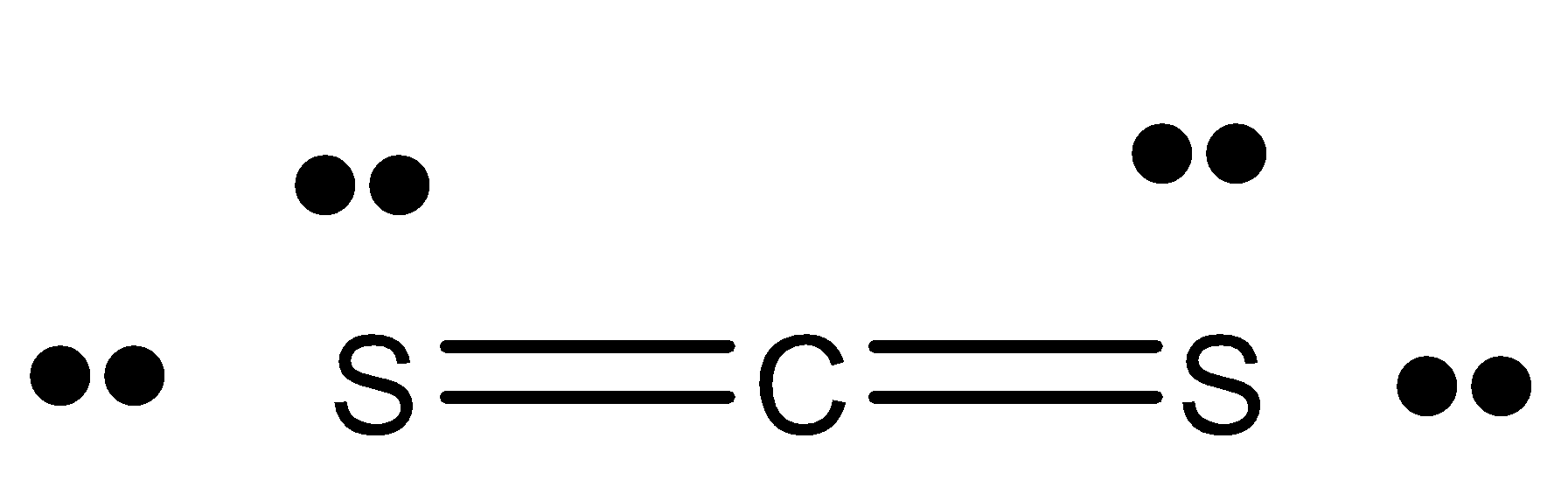 .
.
(Carbon has 4 valence electrons, Sulphur has 6 valence electrons. In ${{C}}{{{S}}_{{2}}}$ , there are 2 Sulphur atoms, so 12 valence electrons. Thus, the total electrons is 16). Now, there is a double bond in ${{C}}{{{S}}_{{2}}}$ and 2 lone pairs on each Sulphur atom. It is used as a chemical non-polar solvent. It belongs to a class of inorganic compounds known as non-metallic sulphides. It is used as a building block in organic chemistry.
Complete step by step answer:
We know that chlorine is a halogen. ${{C}}{{{l}}_{{2}}}$ molecules when reacts with sodium atom produces sodium chloride
When sulphur and carbon are combined at high temperatures, carbon disulphide or carbon disulfide is formed.
\[C + 2S \to C{S_2}\]
Carbon disulphide can be produced from volcanic eruptions also.
We know that chlorine molecule is used as a chlorinating agent, to add chlorine to molecules.
When chlorine is treated with carbon disulphide, we get carbon tetrachloride.
${{C}}{{{S}}_{{2}}}{{ + 3C}}{{{l}}_{{2}}} \to {{CC}}{{{l}}_{{4}}}{{ + }}{{{S}}_{{2}}}{{C}}{{{l}}_{{2}}}$
We could see that chlorine replaces the sulphur in carbon disulphide.
Now, on adding ${{C}}{{{l}}_{{2}}}$ , it gets chlorinated and all the lone pairs on chlorine are satisfied by a chlorine atom, forming carbon tetrachloride.
Thus, the action of chlorine on ${{C}}{{{S}}_{{2}}}$ is the formation of carbon tetrachloride ${{CC}}{{{l}}_{{4}}}$
Note:
We know the uses of the product Carbon Tetrachloride of this reaction. Chlorine is a better leaving group. It is used as a chlorine source in many reactions. Freons which are used in AC and refrigerators are made using ${{CC}}{{{l}}_{{4}}}$ . It is used in fire extinguishers as it has a high melting point and low boiling point. Prolonged exposure can cause so much damage to humans.

(Carbon has 4 valence electrons, Sulphur has 6 valence electrons. In ${{C}}{{{S}}_{{2}}}$ , there are 2 Sulphur atoms, so 12 valence electrons. Thus, the total electrons is 16). Now, there is a double bond in ${{C}}{{{S}}_{{2}}}$ and 2 lone pairs on each Sulphur atom. It is used as a chemical non-polar solvent. It belongs to a class of inorganic compounds known as non-metallic sulphides. It is used as a building block in organic chemistry.
Complete step by step answer:
We know that chlorine is a halogen. ${{C}}{{{l}}_{{2}}}$ molecules when reacts with sodium atom produces sodium chloride
When sulphur and carbon are combined at high temperatures, carbon disulphide or carbon disulfide is formed.
\[C + 2S \to C{S_2}\]
Carbon disulphide can be produced from volcanic eruptions also.
We know that chlorine molecule is used as a chlorinating agent, to add chlorine to molecules.
When chlorine is treated with carbon disulphide, we get carbon tetrachloride.
${{C}}{{{S}}_{{2}}}{{ + 3C}}{{{l}}_{{2}}} \to {{CC}}{{{l}}_{{4}}}{{ + }}{{{S}}_{{2}}}{{C}}{{{l}}_{{2}}}$
We could see that chlorine replaces the sulphur in carbon disulphide.
Now, on adding ${{C}}{{{l}}_{{2}}}$ , it gets chlorinated and all the lone pairs on chlorine are satisfied by a chlorine atom, forming carbon tetrachloride.
Thus, the action of chlorine on ${{C}}{{{S}}_{{2}}}$ is the formation of carbon tetrachloride ${{CC}}{{{l}}_{{4}}}$
Note:
We know the uses of the product Carbon Tetrachloride of this reaction. Chlorine is a better leaving group. It is used as a chlorine source in many reactions. Freons which are used in AC and refrigerators are made using ${{CC}}{{{l}}_{{4}}}$ . It is used in fire extinguishers as it has a high melting point and low boiling point. Prolonged exposure can cause so much damage to humans.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




