The type of hybridization of boron in diborane is?
A- $sp$ – hybridization
B- $sp^{ 3 }$ – hybridization
C- $sp^{ 2 }$ – hybridization
D- ${ sp }^{ 3 }{ d }^{ 2 }$- hybridization
Answer
623.1k+ views
Hint: Try to figure out hybridization based on the number of neighboring atoms around boron atoms. Diborane consists of special bonds to satisfy the octet configuration of boron atoms.
Complete step by step answer:
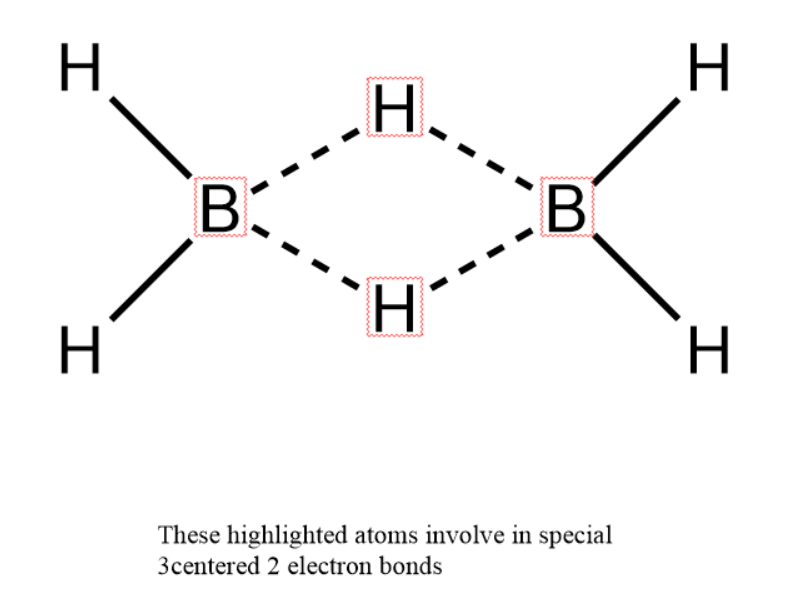
- We know that diborane consists of 2 boron atoms and 6 hydrogen atoms. Out of these 6 hydrogen atoms 4 hydrogen atoms are attached to only one boron atom remaining two hydrogen atoms are attached to both borons through a special type of bond.
-Boron has 3 electrons in valence shell; it cannot complete octet directly through covalent bonds. In the excited state of boron, it has 3 unpaired electrons with 4 orbitals which can involve in bonding. In diborane 2 hydrogen atoms in between two boron atoms are connected to both atoms through a special 3 centered 2 electron bond. In 3 centered 2 electron bonds, 2 electrons are shared in between 3 centers; those are 2 boron atoms and 1 hydrogen atom to satisfy the octet of boron. This type of bond is also considered as regular while calculating hybridization. Since each boron atom has 4 bonds so hybridization will $sp^{ 3 }$ – hybridization.
Therefore, option B is correct.
Note:
has only 3 electrons in the outer shell and can form only 3 hybridized orbitals. So both boron atoms are connected whether it is through direct or indirect bonding.
Complete step by step answer:
- We know that diborane consists of 2 boron atoms and 6 hydrogen atoms. Out of these 6 hydrogen atoms 4 hydrogen atoms are attached to only one boron atom remaining two hydrogen atoms are attached to both borons through a special type of bond.
-Boron has 3 electrons in valence shell; it cannot complete octet directly through covalent bonds. In the excited state of boron, it has 3 unpaired electrons with 4 orbitals which can involve in bonding. In diborane 2 hydrogen atoms in between two boron atoms are connected to both atoms through a special 3 centered 2 electron bond. In 3 centered 2 electron bonds, 2 electrons are shared in between 3 centers; those are 2 boron atoms and 1 hydrogen atom to satisfy the octet of boron. This type of bond is also considered as regular while calculating hybridization. Since each boron atom has 4 bonds so hybridization will $sp^{ 3 }$ – hybridization.

Therefore, option B is correct.
Note:
has only 3 electrons in the outer shell and can form only 3 hybridized orbitals. So both boron atoms are connected whether it is through direct or indirect bonding.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE




