The structure of ${H_2}{O_2}$ is:
A.Planar
B.Non-planar
C.Spherical
D.Linear
Answer
601.8k+ views
Hint: First we should be aware with the term of Hydrogen peroxide which is a compound with peroxy linkage among the molecule. It is a very significant compound with both the oxidising and reducing capabilities. Its structure in not plane due to lone pair repulsion thus possess a non-planar structure.
Complete Step by step answer: Here in the given question statement the question is regarding the compound of Hydrogen peroxide. It is a very important compound in the field of chemistry and is used in multiple occasions as an oxidising as well as reducing agent in both inorganic and organic chemistry. It plays a great role in many processes.
Here are some facts regarding the nature and the molecular structure of ${H_2}{O_2}$ :
Hydrogen peroxide is a compoud which a peroxy linkage made up of oxygen and hydrogen.
The chemical formula of it is ${H_2}{O_2}$ .
In its pure form, hydrogen peroxide is very pale blue and in liquid state. It slightly more viscous than water.
Hydrogen peroxide is one of the simplest derivable and stable peroxide. Peroxide refers to a compound with an oxygen–oxygen single bond.
It is used in multiple tasks but most prominently as an oxidizer, bleaching agent, and antiseptic.
The structure of ${H_2}{O_2}$ is not a normal kind of structure, perhaps a unique one.open book shaped, and thus non-planar.
The structure of ${H_2}{O_2}$ looks like this:
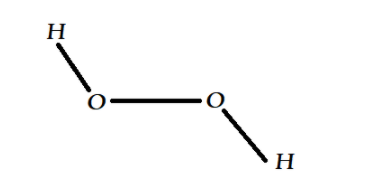
So that provides us the corret option as option B, Non-planar.
Note: Hydrogen peroxide is unstable and slowly decomposes in the presence of light. Because of its instability, hydrogen peroxide is typically stored with a stabilizer in a weakly acidic solution in a dark colored bottle. Hydrogen peroxide is found in biological systems including the human body. Enzymes that use or decompose hydrogen peroxide are classified as peroxidases.
Complete Step by step answer: Here in the given question statement the question is regarding the compound of Hydrogen peroxide. It is a very important compound in the field of chemistry and is used in multiple occasions as an oxidising as well as reducing agent in both inorganic and organic chemistry. It plays a great role in many processes.
Here are some facts regarding the nature and the molecular structure of ${H_2}{O_2}$ :
Hydrogen peroxide is a compoud which a peroxy linkage made up of oxygen and hydrogen.
The chemical formula of it is ${H_2}{O_2}$ .
In its pure form, hydrogen peroxide is very pale blue and in liquid state. It slightly more viscous than water.
Hydrogen peroxide is one of the simplest derivable and stable peroxide. Peroxide refers to a compound with an oxygen–oxygen single bond.
It is used in multiple tasks but most prominently as an oxidizer, bleaching agent, and antiseptic.
The structure of ${H_2}{O_2}$ is not a normal kind of structure, perhaps a unique one.open book shaped, and thus non-planar.
The structure of ${H_2}{O_2}$ looks like this:

So that provides us the corret option as option B, Non-planar.
Note: Hydrogen peroxide is unstable and slowly decomposes in the presence of light. Because of its instability, hydrogen peroxide is typically stored with a stabilizer in a weakly acidic solution in a dark colored bottle. Hydrogen peroxide is found in biological systems including the human body. Enzymes that use or decompose hydrogen peroxide are classified as peroxidases.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Explain zero factorial class 11 maths CBSE




