The pure form of carbon is found in:
A. Jute
B. Diamond
C. Methane Gas
D. All of the above
Answer
620.1k+ views
Hint: We should be having an idea about the allotropes of carbon. Knowing the occurrence of the allotropes will help us to find the purest form of carbon. Think about the composition, chemical formulae and the sources of all the materials given in the options.
Complete answer:
Allotropy is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Allotropes are different structural modifications of an element, the atoms of the element are bonded together in a different manner.
There are three main allotrope of carbon, which are graphite, diamond and buckminsterfullerene. Among these allotropes, carbon is found in the purest form in diamond. Diamond is arranged in consecutive tetrahedral unit structures. A carbon is present at the canter as well as the corners of every tetrahedral unit. These carbon atoms at the vertices are a part of other tetrahedral units. This continues in a never-ending fashion. The structure of diamond is as follows:
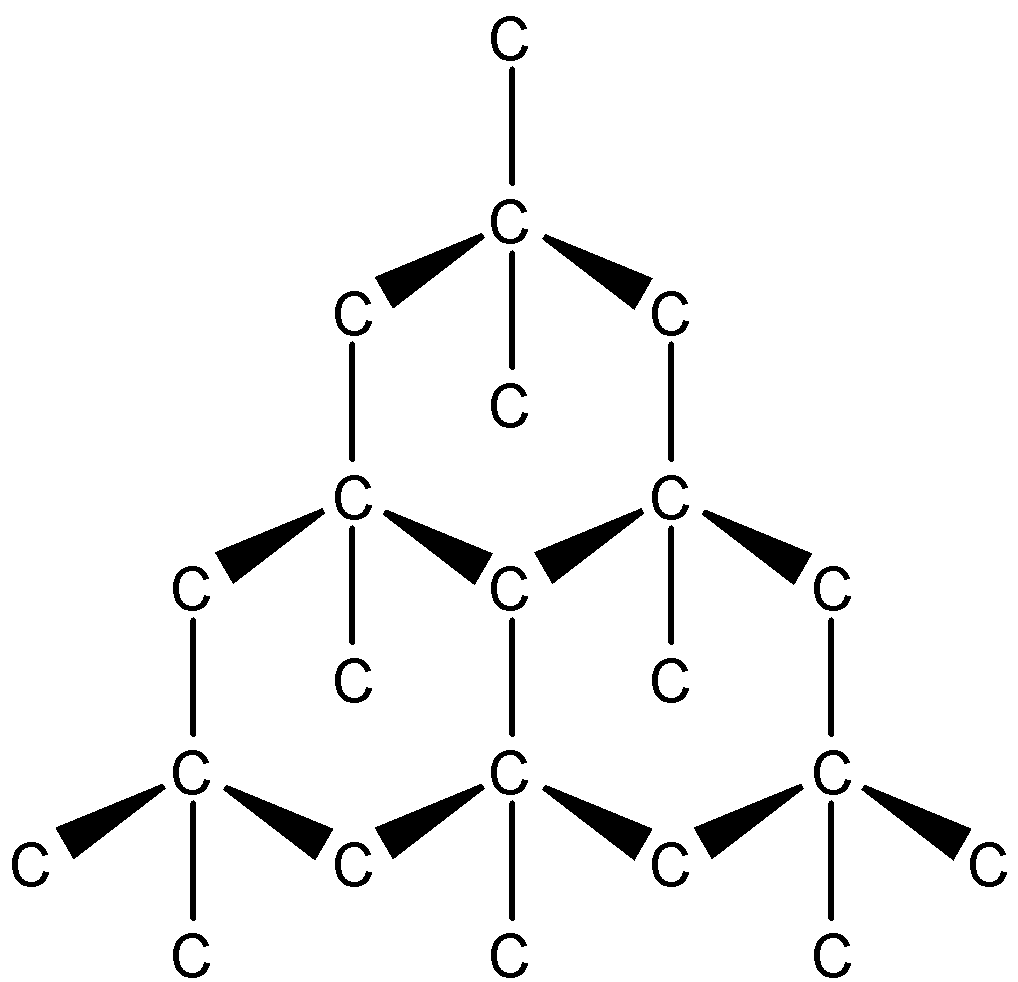
The other answers are not correct. While they do consist of carbon atoms, they contain many other atoms and are far from what we can call pure. So, the correct answer is “Option B”.
Additional Information: Here are some properties of diamond which we should know:
- Diamond consists of carbon atoms which are arranged in the specific type of cubic lattice, known as diamond cubic.
- Diamond is an optically anisotropic crystal that is transparent to opaque.
- Diamond is the hardest naturally known material.
- There are no weak bonds in diamond, so it takes a lot of energy to break the material, giving rise to the high strength of diamond.
- Diamond does not conduct electricity since there are no delocalized electrons in the structure.
Note: Although methane does contain carbon atoms, the chemical formula of methane is $C{{H}_{4}}$, thus you can see that it contains 4 hydrogen atoms per carbon atom. Jute is a type of fibre that is made from the plant materials cellulose and lignin, which are complex compounds of carbon as well as other atoms.
Complete answer:
Allotropy is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Allotropes are different structural modifications of an element, the atoms of the element are bonded together in a different manner.
There are three main allotrope of carbon, which are graphite, diamond and buckminsterfullerene. Among these allotropes, carbon is found in the purest form in diamond. Diamond is arranged in consecutive tetrahedral unit structures. A carbon is present at the canter as well as the corners of every tetrahedral unit. These carbon atoms at the vertices are a part of other tetrahedral units. This continues in a never-ending fashion. The structure of diamond is as follows:

The other answers are not correct. While they do consist of carbon atoms, they contain many other atoms and are far from what we can call pure. So, the correct answer is “Option B”.
Additional Information: Here are some properties of diamond which we should know:
- Diamond consists of carbon atoms which are arranged in the specific type of cubic lattice, known as diamond cubic.
- Diamond is an optically anisotropic crystal that is transparent to opaque.
- Diamond is the hardest naturally known material.
- There are no weak bonds in diamond, so it takes a lot of energy to break the material, giving rise to the high strength of diamond.
- Diamond does not conduct electricity since there are no delocalized electrons in the structure.
Note: Although methane does contain carbon atoms, the chemical formula of methane is $C{{H}_{4}}$, thus you can see that it contains 4 hydrogen atoms per carbon atom. Jute is a type of fibre that is made from the plant materials cellulose and lignin, which are complex compounds of carbon as well as other atoms.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




