The number of equivalent \[Cl - O\] bonds in $C{l_2}{O_7}$ is:
A. $6$
B. $4$
C. $8$
D. $2$
Answer
607.8k+ views
Hint: A chemical substance composed from the atoms of different elements which are bonded together by chemical bonds is known as chemical compound. Dichlorine heptoxide is a chemical compound, chlorine and oxygen elements have contributed to this compound.
Complete step by step answer:
A chemical substance composed of many molecules made up of atoms of more than one element which are held together by chemical bonds is known as a chemical compound. If it is made up of a single type of element then it is not called as a compound rather it is a molecule.
Dichlorine heptoxide is a chemical compound composed of oxygen and chlorine element atoms. The chemical formula of dichlorine heptoxide is $C{l_2}{O_7}$. Dichlorine heptoxide is basically the anhydride of perchloric acid. When maximum possible water molecules are removed from the perchloric acid, dichlorine heptoxide is produced. The dehydrating agent used in this distillation process is phosphorus pentoxide.
Structure of dichlorine heptoxide ($C{l_2}{O_7}$) is:
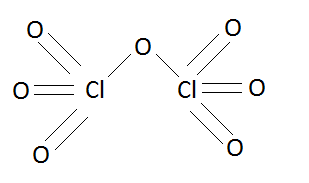
In the above structure we can see that one oxygen atom is shared by both of the chlorine atoms and rest oxygen atoms are distributed equally. This sharing of oxygen atoms or the linkage between three atoms is known as peroxy linkage.
So in this structure dichlorine heptoxide has two chlorine oxygen bonds (\[Cl - O\]) which are situated in the middle of this chemical substance.
Hence the number of equivalent \[Cl - O\] bonds in $C{l_2}{O_7}$ are two.
So, the correct answer is Option D .
Note:
Basically dichlorine heptaoxide is an unstable compound or we can say it is an endergonic material which means intrinsic unstable. Easily it decomposes into its constituent elements with release of energy.
Complete step by step answer:
A chemical substance composed of many molecules made up of atoms of more than one element which are held together by chemical bonds is known as a chemical compound. If it is made up of a single type of element then it is not called as a compound rather it is a molecule.
Dichlorine heptoxide is a chemical compound composed of oxygen and chlorine element atoms. The chemical formula of dichlorine heptoxide is $C{l_2}{O_7}$. Dichlorine heptoxide is basically the anhydride of perchloric acid. When maximum possible water molecules are removed from the perchloric acid, dichlorine heptoxide is produced. The dehydrating agent used in this distillation process is phosphorus pentoxide.
Structure of dichlorine heptoxide ($C{l_2}{O_7}$) is:

In the above structure we can see that one oxygen atom is shared by both of the chlorine atoms and rest oxygen atoms are distributed equally. This sharing of oxygen atoms or the linkage between three atoms is known as peroxy linkage.
So in this structure dichlorine heptoxide has two chlorine oxygen bonds (\[Cl - O\]) which are situated in the middle of this chemical substance.
Hence the number of equivalent \[Cl - O\] bonds in $C{l_2}{O_7}$ are two.
So, the correct answer is Option D .
Note:
Basically dichlorine heptaoxide is an unstable compound or we can say it is an endergonic material which means intrinsic unstable. Easily it decomposes into its constituent elements with release of energy.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE




