The major product of the above reaction is:
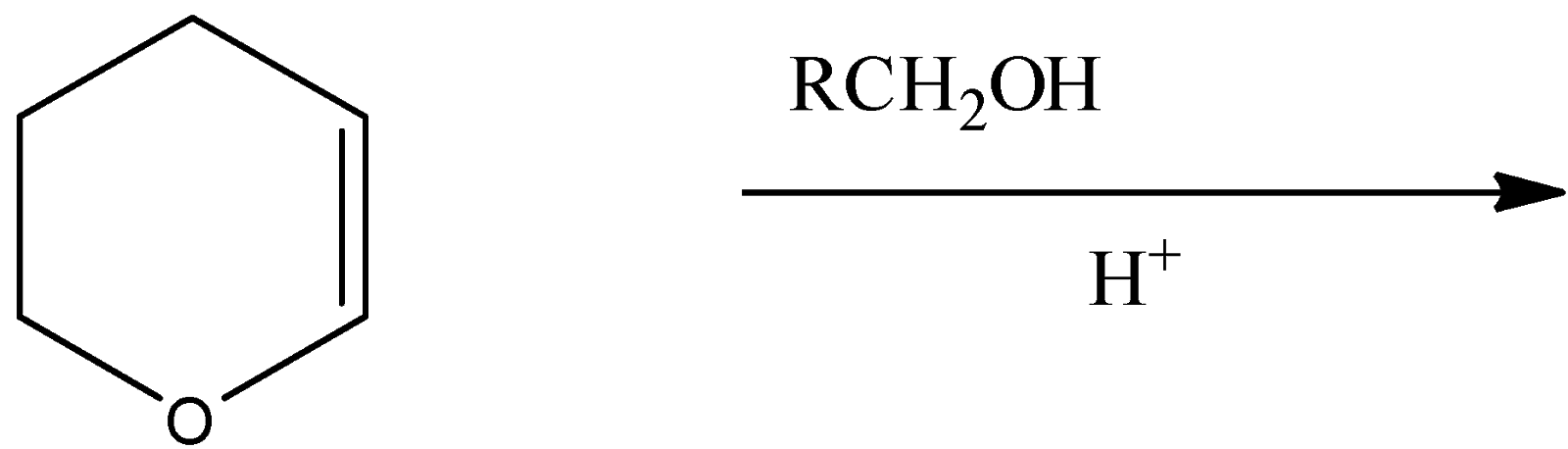
(A) A hemiacetal
(B) An acetal
(C) An ether
(D) An ester

Answer
597.9k+ views
Hint: A hemiacetal is a compound in which the same carbon has an alcohol group and $-OR$ group. An acetal is a compound in which the same carbon atom has two $-OR$ groups. In the above reaction double bond will be replaced with $-OC{{H}_{2}}R$ group.
Complete step by step solution:
So, in the reactant, there is an oxygen atom that has two lone pairs present. Now the lone pair will transfer to the next bond-forming a double bond, the double bond will be attacked by the hydrogen ion because the reaction is taking place in acid. This will form a negative charge on the carbon atom next to the oxygen atom.
Now the reactant $HOC{{H}_{2}}R$, the oxygen atom has two lone pairs. So the lone pair of the oxygen atom will attack the negative charge of the carbon and the $-OC{{H}_{2}}R$ group will be attached.
The reaction is given below:
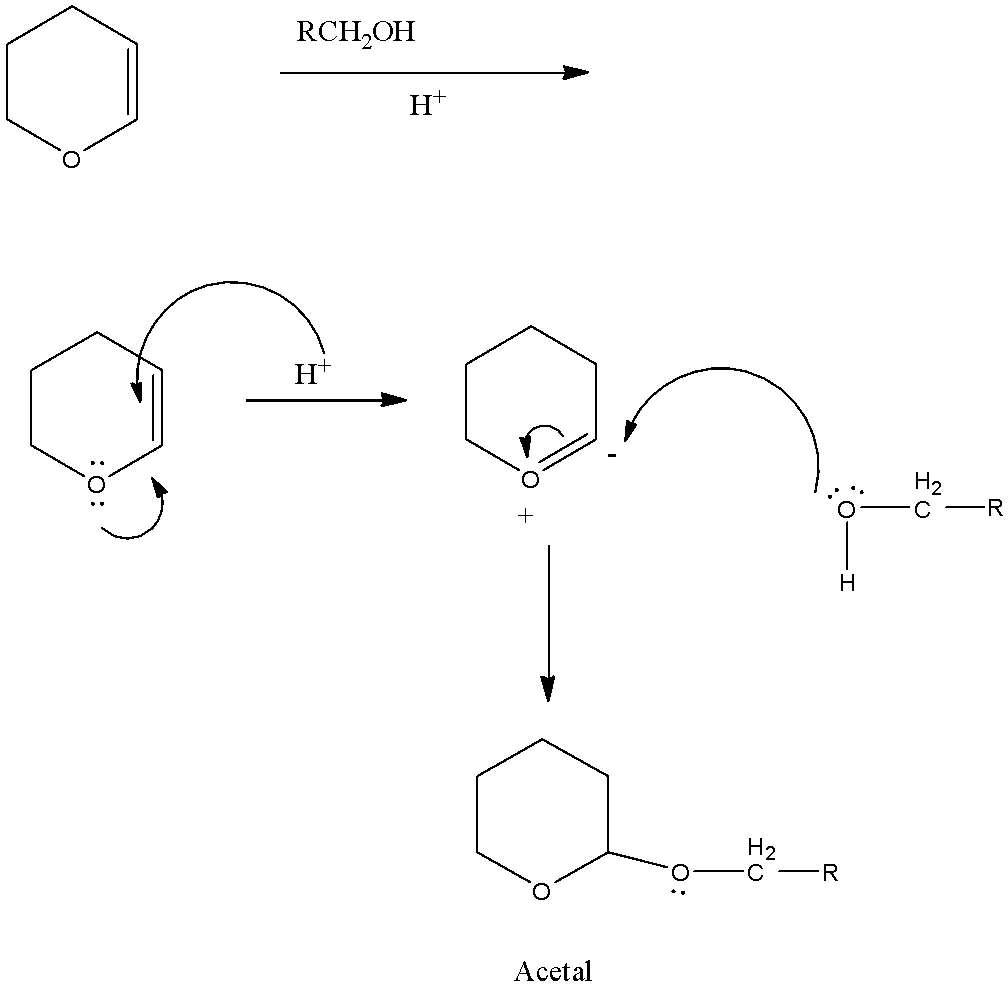
Here in this compound, the same carbon atom has two $-OR$ groups, therefore, it is an acetal.
Therefore, the correct answer is an option (B) An acetal.
Additional information:
A hemiacetal is a compound in which the same carbon has an alcohol group and $-OR$ group. Ether is a compound in which two alkyl groups are joined by one oxygen atom. An ester is a compound in which the functional group between two alkyl groups is $-COO-$.
Note: In an acetal compound the R group can be the same or different, but it must be on the same carbon atom. The above reaction can only take place in the presence of acid.
Complete step by step solution:
So, in the reactant, there is an oxygen atom that has two lone pairs present. Now the lone pair will transfer to the next bond-forming a double bond, the double bond will be attacked by the hydrogen ion because the reaction is taking place in acid. This will form a negative charge on the carbon atom next to the oxygen atom.
Now the reactant $HOC{{H}_{2}}R$, the oxygen atom has two lone pairs. So the lone pair of the oxygen atom will attack the negative charge of the carbon and the $-OC{{H}_{2}}R$ group will be attached.
The reaction is given below:

Here in this compound, the same carbon atom has two $-OR$ groups, therefore, it is an acetal.
Therefore, the correct answer is an option (B) An acetal.
Additional information:
A hemiacetal is a compound in which the same carbon has an alcohol group and $-OR$ group. Ether is a compound in which two alkyl groups are joined by one oxygen atom. An ester is a compound in which the functional group between two alkyl groups is $-COO-$.
Note: In an acetal compound the R group can be the same or different, but it must be on the same carbon atom. The above reaction can only take place in the presence of acid.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




